Norbornane is a bicyclic compound (bicyclo[2.2.1]heptane) with a rigid, saturated framework. Norbornene is its corresponding alkene, containing a double bond that makes it reactive toward typical electrophilic addition reactions. Norbornanone is a ketone derivative, where the carbonyl group undergoes nucleophilic addition reactions.
Because of this rigidity, reactions of norbornane and norbornene derivatives are not only governed by the usual reactivity of functional groups, but also by stereochemical constraints imposed by the framework. This makes them excellent models for studying how steric effects and geometry control reaction pathways.
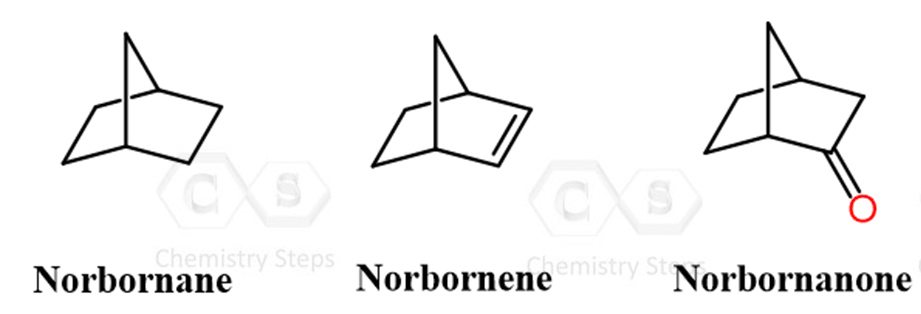
In particular, many reactions – such as additions to norbornene, nucleophilic additions to norbornanone, and eliminations in norbornane derivatives – show strong face selectivity (exo vs endo), often favoring attack from the less hindered exo face, unless substitution alters this preference.
Let’s start our discussion with the reactions of norbornene. We can see that it has a double bond, and as expected, most reactions of alkenes are characteristic of norbornene systems. There are, of course, some important differences we need to be familiar with.
This content is for registered users only.
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.
