This is a 60-question, multiple-choice quiz on organic acids and bases covering the topics listed below.
It comes with a 1-hour video showing a detailed step-by-step solution to all the questions. It’s a must-watch, master course video!
Organic Acids and Bases
- The definition of organic acids and bases, as well as conjugate acids and bases.
Example question:
Identify the acid, base, conjugate acid, and conjugate base:

Conjugate Acids and Bases
Acid Strength and pKa
The pKa is the quantitative description of the acid strength. For the beginning, it is important to identify the more acidic proton in a compound and also determining the more acidic compound.
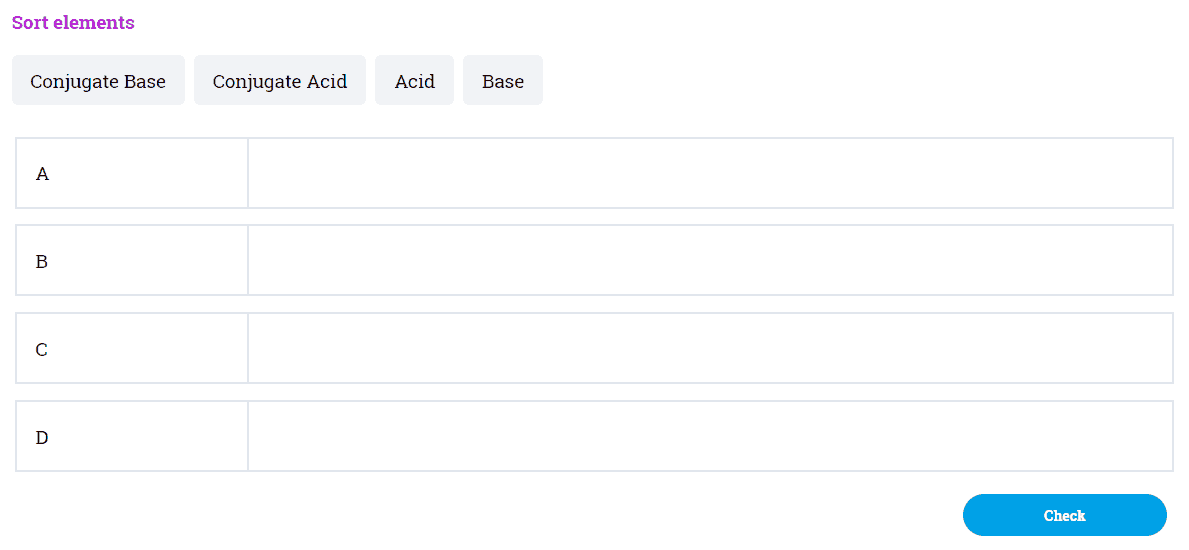
Identify, based on the pKa values, the more acidic compound:
The Position of Equilibrium for an Acid–Base Reaction
Remember that the equilibrium of an acid-base reaction is always shifted towards the formation of a weaker acid. Therefore, always choose the side where the acid (or the conjugate acid) has a higher pKa value.
Example question:
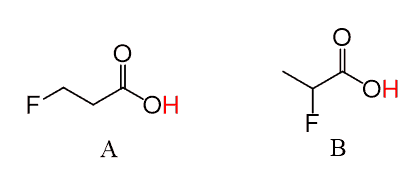
Using the pKa table, determine if the position of equilibrium for each of the following reactions will be on the left or on the right side:
Factors That Determine the pKa and Acid Strength (ARIO)
The acidity of a proton can be determined based on the Atom that it is connected to, the possible Resonance structures of the conjugate bases, the Inductive effect, and the Orbital based on the hybridization state of this atom.
Example question:
Identify, based on the pKa values, the more acidic compound:

How to Choose an Acid or a Base to Protonate or Deprotonate a Given Compound
Often, we are asked to pick an appropriate Acid or a Base to Protonate or Deprotonate a Given Compound. This is, again, based on the pKa values of the acid and the conjugate acid. The simple principle to remember there is that any acid base reaction favors formation of a weaker acid and base.
Example question:
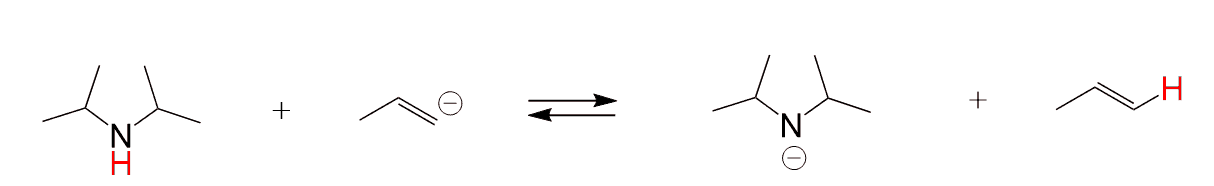
Choose a compound that can be used as a base (deprotonate the acid) in the following reaction:

Lewis Acids and Bases
The Lewis acid-base theory is the most comprehensive since it defines the acids and bases from the prospective of electron donor and acceptor. Many organic reaction go by this definition and, thereofore Lewis acid-base definition relates to most organic chemistry reactions. Recognizing Leweis acids and base is another important tool for understanding organic chemistry reactions.
Example question:
Identify the Lewis acid and the base in the following reaction:
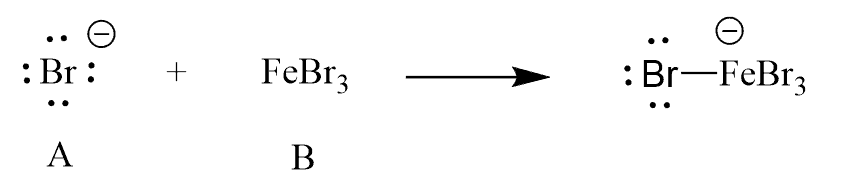


how to go back and review the quizzes if you got out one time?