In this post, we will talk about the synthesis of amides using DCC and EDC coupling agents. Before getting into the details, let’s first discuss a couple of strategies for preparing amides without using any fancy coupling agent.
First, considering the nucleophilic addition-elimination mechanism, the reaction of an amine with a carboxylic acid may seem to be a suitable way of making an amide:

When comparing the –OH and RNH–, the hydroxide ion is a better leaving group. Recall, the pKa (H2O) = 15.7, and the pKa (RNH2) = 38. Therefore, the –OH is a weaker base and hence a better leaving group.
However, the problem with this is that carboxylic acids are strong organic acids, and when mixed with a base, the fast acid-base reaction occurs first, before any nucleophilic substitution reaction:
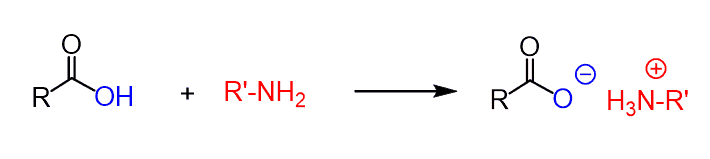
So, the protonation kills the nucleophilic properties of the amide since it no longer has a lone pair of electrons. If heated > 100 oC, the ammonium salts can be converted to amides:
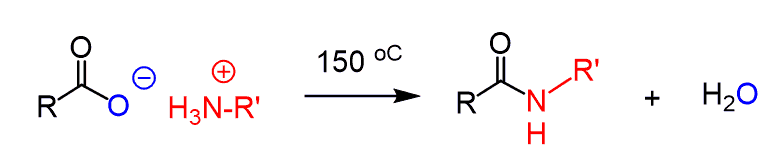
However, this is not an ideal condition for many compounds, and therefore, alternative ways of preparing amides were developed.
DCC coupling – Amides from Amines and Carboxylic Acids
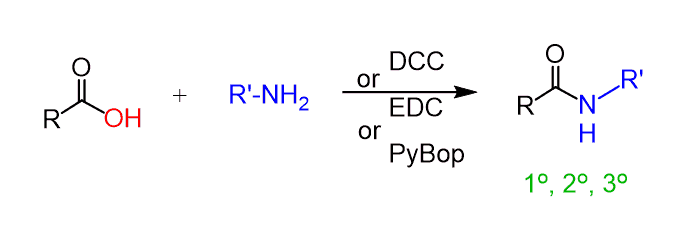
One of the strategies is to use a coupling agent such as DCC (N,N’-dicyclohexane carbodiimide) or EDC (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide. Both are carbamides (RN=C=NR), which prevent the acid-base reaction and make the carboxylic acid susceptible to a nucleophilic attack.
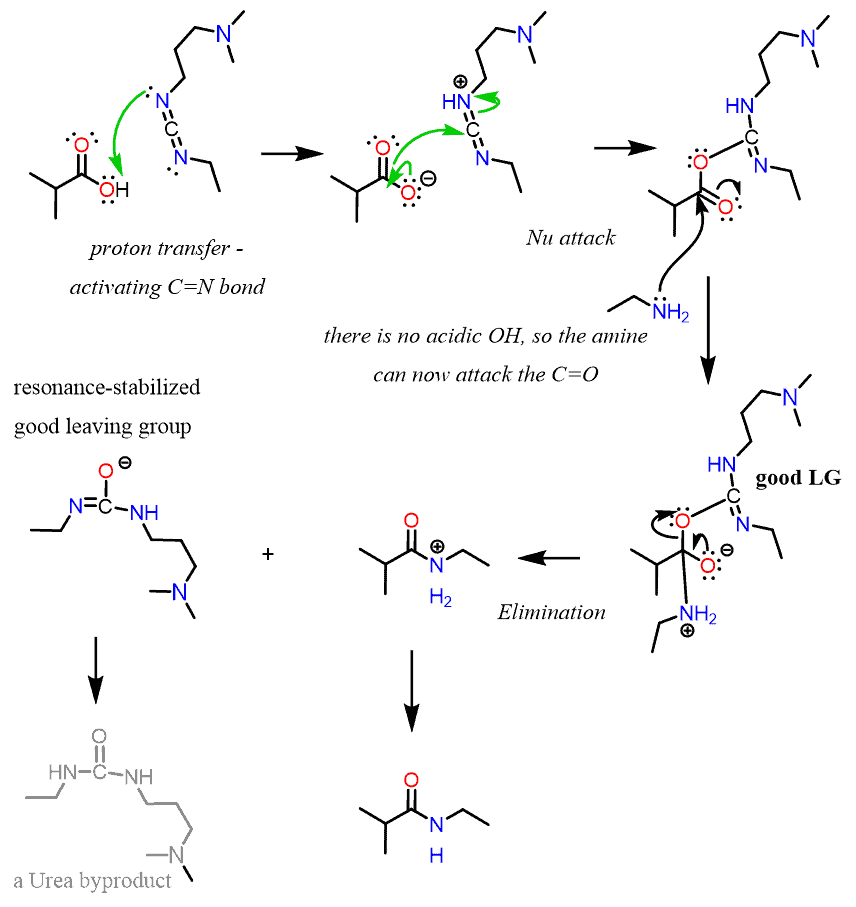
In the first step, the imine deprotonates the carboxylic acid and becomes highly electrophilic, which promotes a nucleophilic attack by the carboxylate group:

This forms an intermediate with the oxygen converted to a good leaving group, and after an addition-elimination, a protonated amide is formed. The side product of the reaction is a conjugate base of urea, which deprotonates the amide.
Without DCC, the amine deprotonates the carboxylic acid instead of a nucleophilic attack on the C=O. So, DCC has two main functions: 1) getting rid of the acidic proton, and 2) converting the oxygen into a good leaving group.
EDC coupling – Amides from Amines and Carboxylic Acids
In an identical reaction, EDC facilitates the formation of amides from carboxylic acids:
In both cases, the reaction works at room temperature with quite good yields (70-90 %).
Aside from this, remember, you can also use esters and especially acid chlorides to readily react with amines and get the target amide:
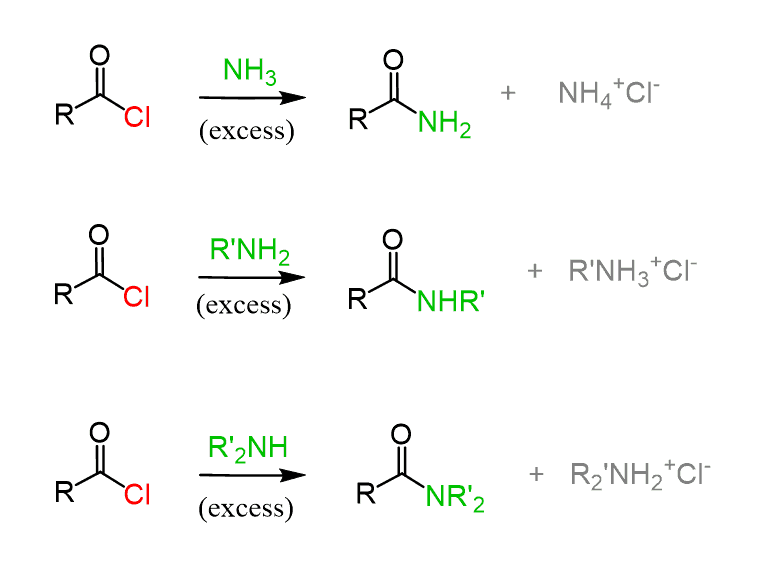
In general, using these coupling reagents is not the only way of preparing amides. Other common approaches include converting carboxylic acids to more reactive derivatives such as acid chlorides or anhydrides, which then react readily with amines, as well as direct condensation of esters with amines (aminolysis). Amides can also be formed from nitriles via hydrolysis or through rearrangement reactions such as the Hofmann and Beckmann rearrangements.
Below are some practice problems on preparing amides using different reagents and strategies.

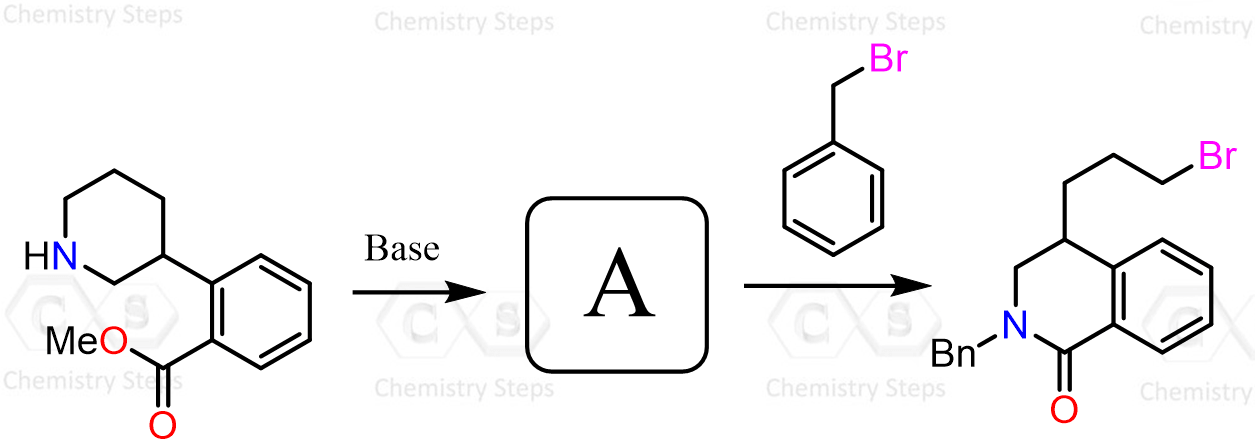
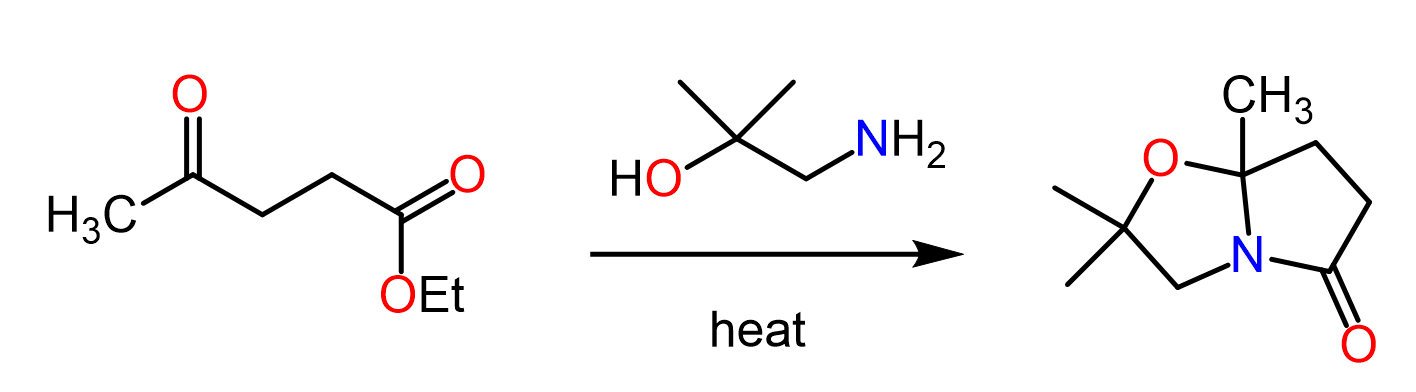
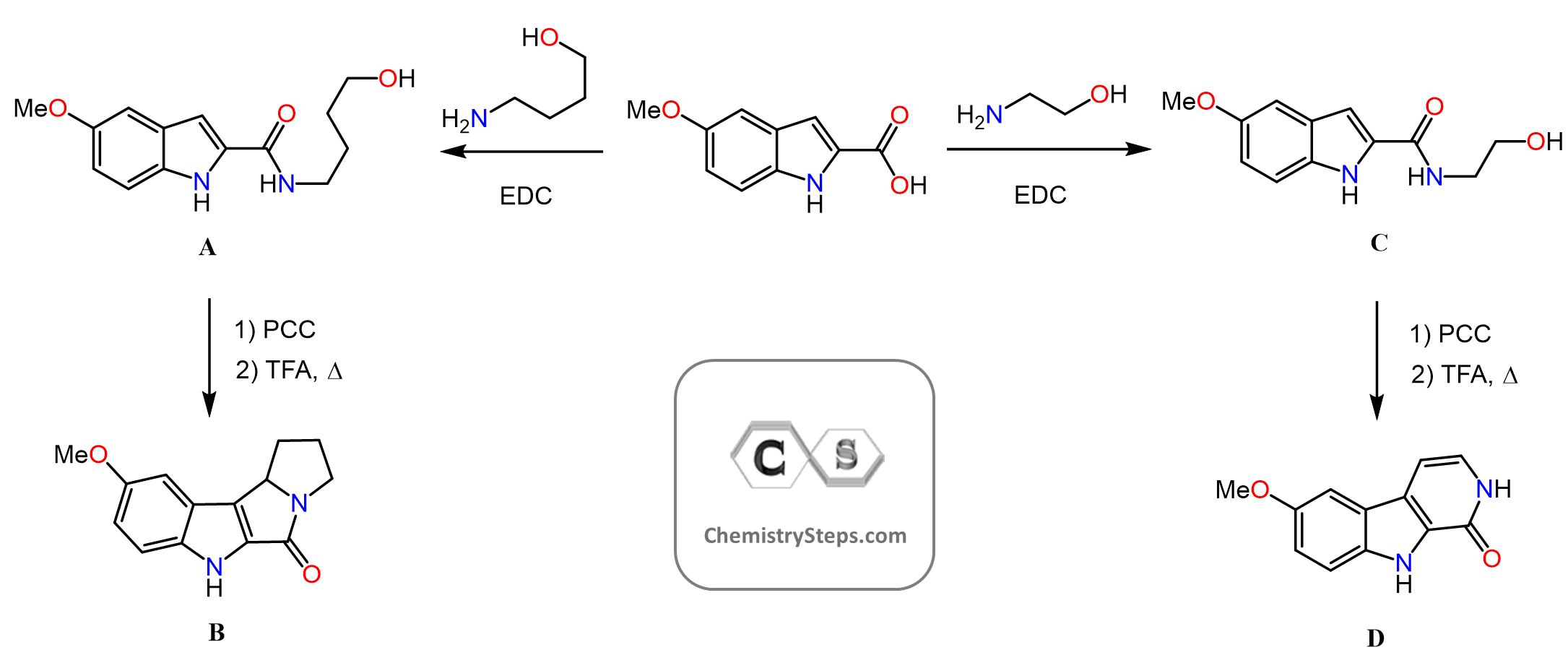
Amines are better nucleophiles than carboxylates. So, why amine do not add on DCC (or protonated form of DCC)…?
DCC is attacked by carboxylate in the protonated form of EDC. First, EDC takes a proton from Acid and forms carboxylate ion. Then EDC becomes an electrophilic center. Hence to attack EDC, it must be protonated. But amine does not use as a proton source. So, Amine-EDC complex does not form. But Acid-EDC complex forms.
I would say this: All the addition and elimination steps are reversible, and therefore, even if the amine adds to DCC, it can still be expelled by the lone pairs of the nitrogen in DCC. Although I ran this reaction several times, I only dedicated time to study the byproducts when the yield with a large molecule was very low.
Interestingly, the question asked below made me go back and retrieve some details about the reaction mechanism which still leaves the possibility of some other addition products to the coupling agents.
Hi. Thanks a lot for these amazing explanations, I don’t know why also NHS and DMAP would be added simultanieously into this type of reactions? and between DCC and PYBOP whis is better to use?
You are in luck asking this question as I worked with amide synthesis reactions using different coupling agents. They are all essentially doing the same thing – converting the OH into a good leaving group. In some reactions, when I used DMAP and EDC, the yield was about 30 percent, so I started analyzing the products and did some literature search. A common reason for this mentioned in papers was the rearrangement of the intermediate A via O-N acyl shift which apparently was faster the addition of the amine. The resulting derivative of urea is an amide, and these are pretty reluctant towards nucleophilic addition-elimination reactions. What PyBop does is it attacks the carbon before the rearrangement occurs forming a new intermediate that is stable but reactive enough to be attacked by the amine.
This does not, however, mean that EDC or DCC with DMAP will always be less effective than, let’s say EDC with PyBop or similar coupling agents. It just happened to be for the reaction that I was doing. At the end of the day, all you want is find a suitable combination of reagents to get your reaction going😊
In some reactions, when I used DMAP and EDC, the yield was about 30 percent, so I started analyzing the products and did some literature search.
–> I’m really curious about the paper you mentioned—would you mind sharing the reference?
Can’t find all, but this should be one of them: Carbodiimide chemistry: recent advances, Chem. Rev. 1981, 81, 6, 589–636
You can try adding PyBop or Hobt – they usually help.
Hi, I’m curious about the step involving the conversion of the acid–EDC intermediate to the HOBt species. How is HOBt formed from PyBOP? You mentioned that ‘PyBOP attacks the carbon,’ but it’s actually HOBt that acts as the nucleophile, not PyBOP itself. Right?
They are different agents that are used interchangeably.
But PyBOP Is reported to be a coupling agent itself, while HOBt is not, It must be used with DCC or EDC.
They are not the same, I agree with you that once PyBOP degrade somehow It frees HOBt and that Is the nucleophile.
The point Is how does HOBt forms from PyBOP?
Hey Daniele, PyBop is a derivative of Hydroxybenzotriazole (abbreviated HOBt). You’d need to do some research on how exactly the Acid-Hobt derivative is formed, but it is there, the formation of the amide should be a straightforward nucleophilic addition elimination to the the carbonyl group. More specifically, it is an amylolysis reaction where Hobt is the byproduct leaving group from the ester.
Is this a class material or you are doing a reaction in the lab?
Perchè è preferibile usare agenti di accoppiamento piuttosto che il cloruro??
Can this method be applicable to indole amide coupling with methacrylic acid?
It should work, though α,β-unsaturated acids can give competing 1,4-addition (Michael addition) with amines under certain conditions.