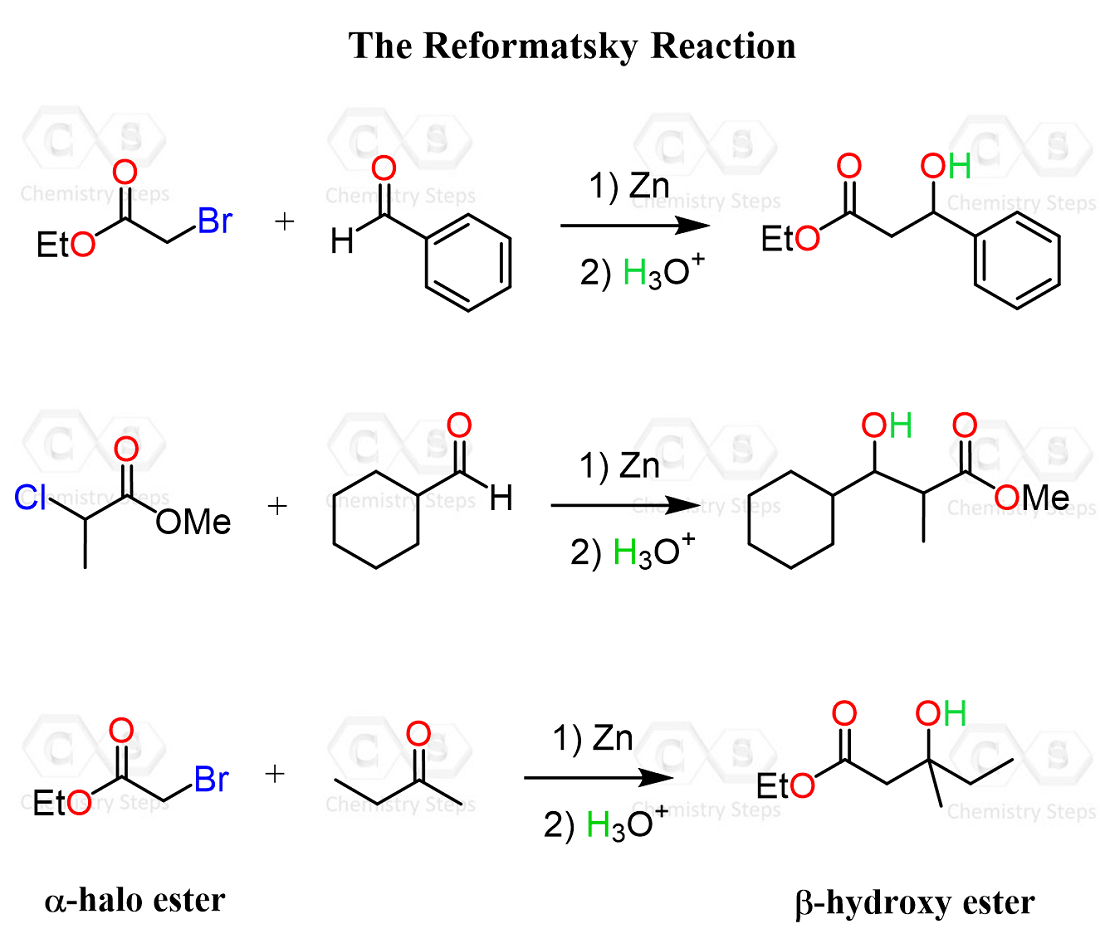
All the aldol reactions we discussed so far were between identical partners, meaning the same carbonyl compound served as an electrophile and a nucleophile in the form of an enolate:

An aldol reaction between two different carbonyl compounds is called a crossed aldol or mixed aldol reaction.
For example, if we mix acetaldehyde and propanal in the presence of sodium hydroxide, a crossed aldol reaction is expected to occur with four different products. This happens because each aldehyde has an ɑ hydrogen and therefore, can be converted into an enolate to react with the carbonyl form of itself or the other molecule.
The structure of the aldol addition product depends on which aldehyde served as the enolate and which one reacted as the electrophile:

Because of this, crossed aldol reactions are generally not useful in organic synthesis. One exception is the reaction of aldehydes with no alpha hydrogens.
A good example of these is benzaldehyde and formaldehyde:

The lack of the ɑ hydrogens prevents it from converting into an enolate. So, if we react it with propanal in the presence of a base, one product is expected since benzaldehyde can only serve as an electrophile:
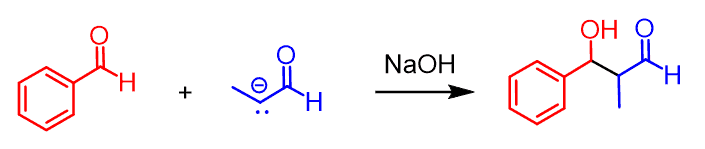
Sounds like a good workaround for the issues of crossed aldol reactions. However, there is one thing we have not considered here, and that is the fact that propanal can still react with itself. Its enolate does not have to react with benzaldehyde only:

To avoid this unwanted path, we also need to consider the order of addition in a crossed aldol reaction. The strategy is to mix the benzaldehyde with sodium hydroxide and add the acetaldehyde to this solution in a dropwise manner to keep its concentration very low. This allows for a huge excess of the benzaldehyde over the enolate that is forming, thus preventing the self-condensation reaction:

Self-condensation can be avoided by planning the order of addition for carbonyls with no a-hydrogens:
Directed Aldo Reaction
We talked about the crossed aldol reaction, its limitations, and strategies to overcome those by using carbonyl compounds with no ɑ hydrogens.
However, chemists wouldn’t tolerate such a limitation, and luckily, it was found that any crossed aldol reaction can be synthetically useful by selectively and irreversibly converting one of the carbonyl compounds into its enolate form.
For example, let’s go back to the reaction between acetaldehyde and propanal and assume the following is the only target product:

First, we identify, based on the target product, which of the aldehydes had served as the nucleophile by cleaving the bond between ɑ and β carbon atoms:

This indicates that propanal needs to be irreversibly converted into an enolate and then reacted with acetaldehyde. This deprotonation is achieved with lithium diisopropyl amide (LDA) – a strong, bulky base that you are going to see the most in alpha carbon chemistry.
The strength of LDA can be understood by comparing pKa values. The conjugate acid of LDA, diisopropylamine, has a pKa of about 36, while typical alpha hydrogens of carbonyl compounds have pKa values in the range of about 19 to 25. Because LDA is associated with a much weaker conjugate acid, it is strong enough to deprotonate these alpha carbons, making enolate formation both feasible and efficient.
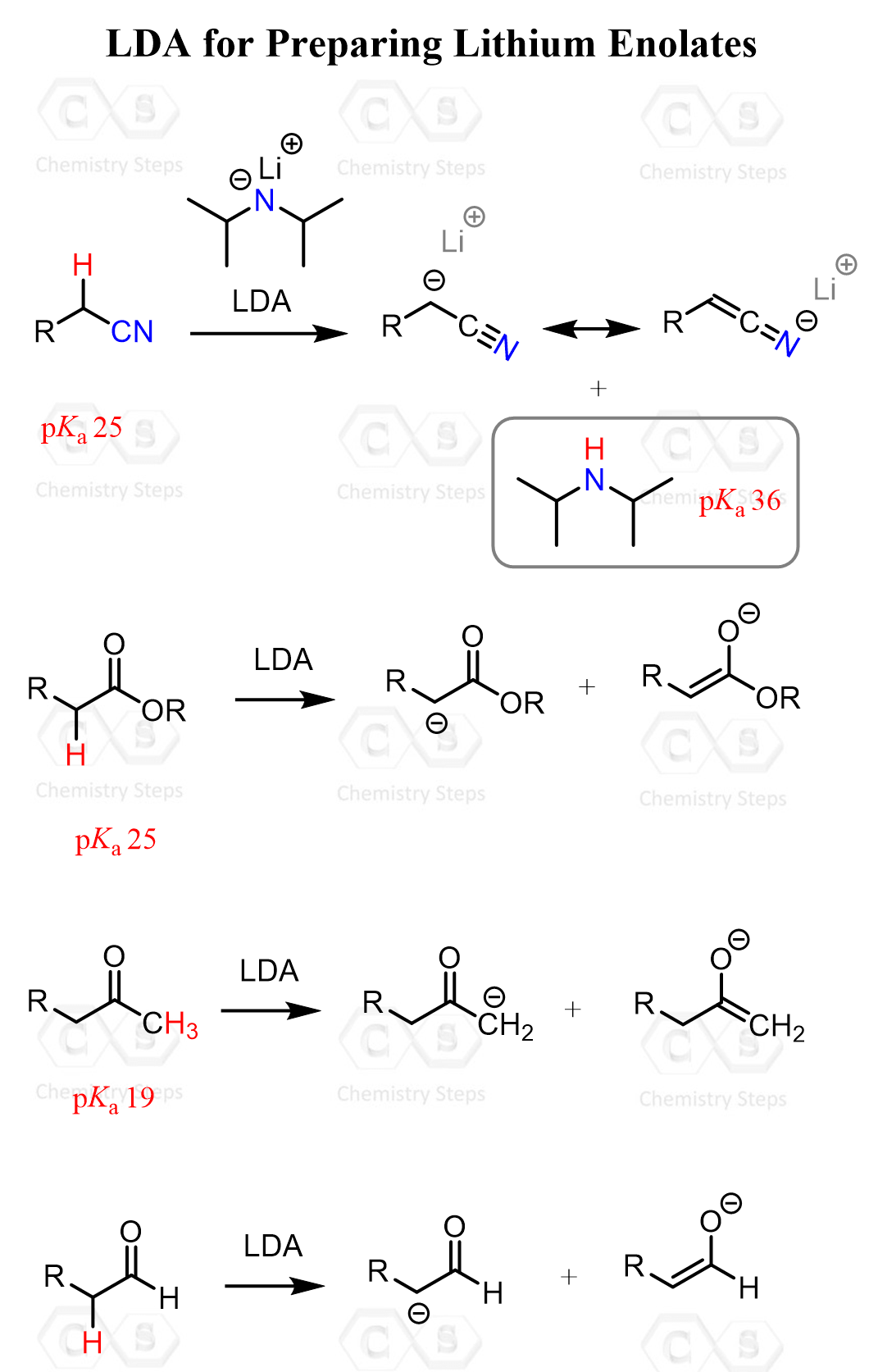
The deprotonation is carried out at low temperature, and -78 oC just happens to be the surface temperature of dry ice (CO2), which is the most common temperature for preparing an affordable ice bath.
Mechanistically, it is an attack of the nitrogen lone pair on the acidic alpha hydrogen, forming a new N-H bond while the C-H bond electrons remain on carbon to generate the enolate. At the same time, the lithium cation coordinates to the oxygen of the carbonyl, stabilizing the negative charge and organizing the transition state. This concerted deprotonation makes LDA a strong, non-nucleophilic base that cleanly forms enolates under kinetic control. Tetrahydrofuran (THF) is a common solvent used for these reactions.
Once the enolate is formed, we add the second carbonyl compound (acetaldehyde in this case, as the electrophile) to the solution of this enolate:
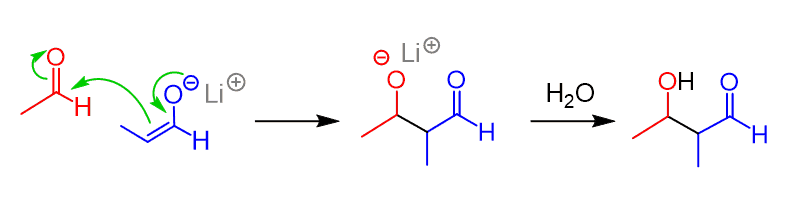
By using this strategy, we prevented the self-condensation of propanal, and the same strategy could be used to convert the acetaldehyde into an enolate and react it with propanal.
Regiochemistry of Crossed Aldol Reactions
LDA is a sterically hindered strong base, and therefore, it deprotonates the less substituted carbon of the ketone:

The directed aldol reaction works for other C-H acids such as esters and nitriles as well:

To react on the more substituted ɑ-carbon of the ketone, sodium hydride is often used as a strong, unhindered base:
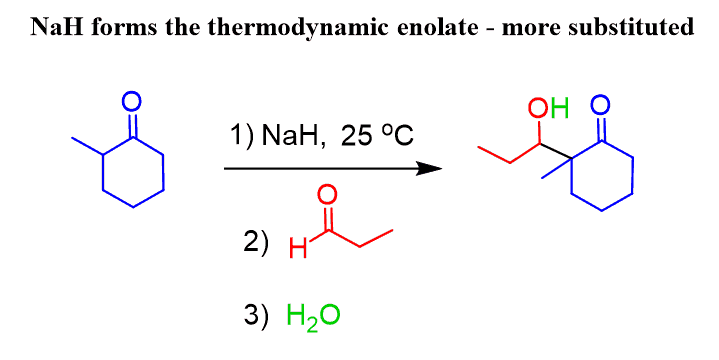
For a symmetrical ketone, it wouldn’t matter which of these bases is used.
The only thing is to make sure a strong base is used. Sodium hydroxide and ethoxides don’t work for directed aldol reactions because the enolate is not formed irreversibly, and self-condensation reactions can occur because there is still a lot of carbonyl present in the equilibrium mixture.
As always, if you are ready to test your skills, here are some practice problems on the crossed aldol reaction:
https://www.chemistrysteps.com/crossed-aldol-retrosynthesis-practice-problems/
Check Also
- Alpha Halogenation of Enols and Enolates
- The Haloform and Iodoform Reactions
- Alpha Halogenation of Carboxylic Acids
- Alpha Halogenation of Enols and Enolates Practice Problems
- Aldol Reaction – Principles and Mechanism
- Aldol Condensation – Dehydration of Aldol Addition Product
- Intramolecular Aldol Reactions
- Aldol Addition and Condensation Reactions – Practice Problems
- Crossed Aldol And Directed Aldol Reactions
- Crossed Aldol Condensation Practice Problems
- Alkylation of Enolates Alpha Position
- Enolate Alkylation Practice Problems
- Acetoacetic Ester Synthesis
- Acetoacetic Ester Enolates Practice Problems
- Malonic Ester Synthesis
- Michael Reaction: The Conjugate Addition of Enolates
- Robinson Annulation, Shortcut, and Retrosynthesis
- Claisen Condensation
- Dieckmann Condensation – An Intramolecular Claisen Reaction
- Crossed Claisen and Claisen Variation Reactions
- Claisen Condensation Practice Problems
- Stork Enamine Synthesis
- Enolates in Organic Synthesis – a Comprehensive Practice Problem