Although amines are considered weak bases, in general, their presence in neutral organic molecules makes the basic sites in those structures because they are stronger bases and nucleophiles than other neutral organic compounds. For example, in DNA, there are aromatic rings known as nucleobases (nitrogenous bases or often simply bases) because of the presence of nitrogen atoms with lone pairs.
Nucleobases are the species that form the complementary hydrogen bonding (A:T, G:C) by which the genetic information is passed on:

So, let’s first understand what makes amines basic. We know from the Brønsted-Lowry acids and base theory that bases are proton acceptors, and in the Lewis acid-base theory, they are classified as electron donors. And therefore, the lone pair of the nitrogen qualifies amines as bases. Mixing an amine with any acid forms an ammonium ion as the nitrogen gets protonated:

The Base Strength of Amines
In any reaction where the amine works as a base, it donates the lone pair to capture a proton. We are talking about acid-base reactions where a proton source is present – in Lewis acid-base reactions, the acid does not have to be protic.
So, what makes amines stronger bases? The stronger the ability of the amine to donate the electron pair, the more basic it is. Therefore, the question is what groups attached to the niterigen can make it more basic by giving it some additional electron density. The first, and simplest trend, is the alkyl groups connected to the nitrogen.
For example, ethylamine is more basic than ammonia because the electron-donating effect of the alkyl group increases the electron density on the nitrogen, thus making it more basic.
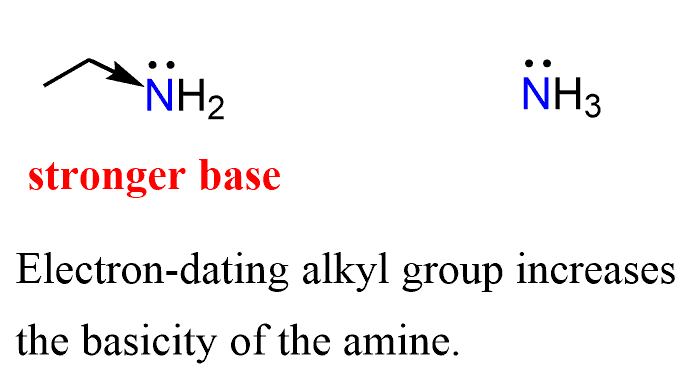
And this is a general trend: the more alkyl groups on the nitrogen, the stronger the basicity of the amine. The steric effects might start influencing this trend; however, for now, let’s remember that alkyl groups increase the basicity of the amine.
How do we quantify the base strength of amines?
We know from the acid-base chemistry that, at least in organic chemistry, the use of pKa is the standard approach when assessing the acid strength. However, because we are looking at the base strength here, the pKa of the conjugate acid is used to predict the basicity of the amine.
For example, going back to the comparison of ammonia and ethyl amine, we use the pKa values of their conjugate acids to assess their basicity. The pKa of the conjugate acid of ammonia is 9.3, and that of ethylamine is 10.8. These numbers tell us that the conjugate acid of ammonia is a stronger acid than that of ethylamine and, therefore, it is a weaker base.

Remember, the stronger the acid, the weaker its conjugate base, and vice versa, the stronger the base, the weaker its conjugate acid. Consequently, because the conjugate acid of ethylamine is a weaker acid, it is a stronger base than ammonia.
What Molecules can be Deprotonated by Amines?
So, when can the amine work as a base? In other words, what acids can be used to protonate an amine? In general, any inorganic acid, such as HCl and H2SO4, and also carboxylic acids, is strong enough to protonate an amine. If you are asked to pick a compound that can serve as an acid and protonate a given amine, you need to compare the pKa values. The pKa of many protonated amines is 10–11, and therefore, the pKa of the starting acid must be less than 10 for the equilibrium to favor the products.
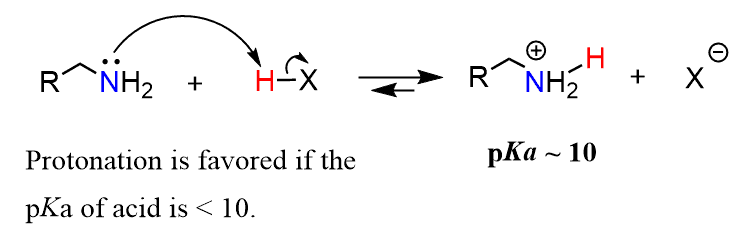
Remember that equilibrium favors the formation of a weaker acid, i.e., it must have a larger pKa than the starting acid. Again, for most amines, this falls in the range that is below 10.
Basicity of Alkyl and Aryl Amines
Let’s compare the base strength of cyclohexylamine and aniline and state from the beginning that the former is a stronger base. So, you can take a moment to try finding an explanation for this observation:
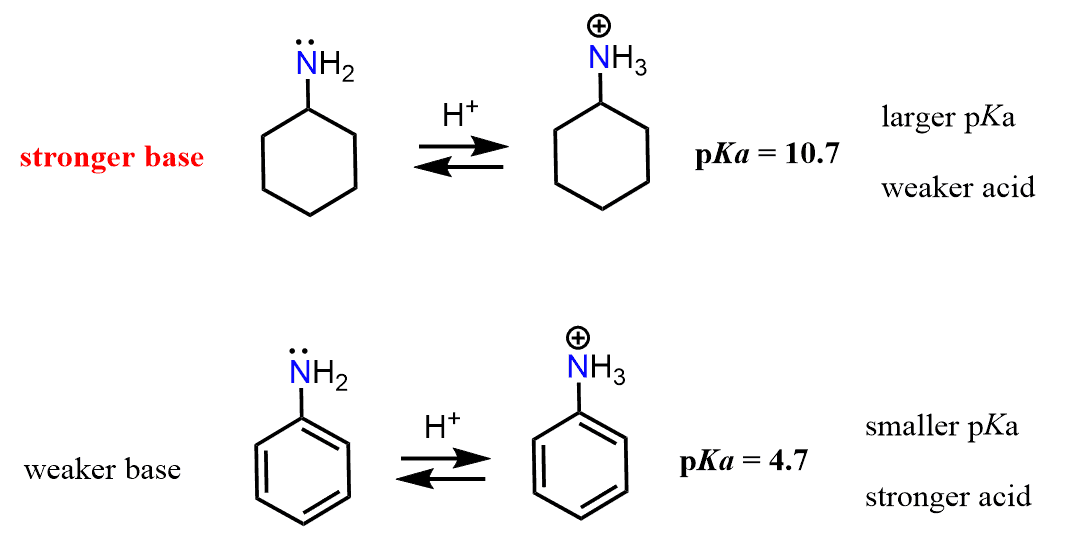
Keep in mind that the base strength of the amine will increase with its readiness to donate the electron pair. Now, the lone pair on the nitrogen in ethyl amine is “not doing anything” other than just sitting there, i.e., it is localized. The one in aniline, on the other hand, is next to an aromatic ring, which is a conjugated system, and if this lone pair can be part of the conjugate system, we can predict that it is not going to be as ready to accept a proton. And, in fact, the lone pair of nitrogen in aniline is delocalized on the benzene ring, and that is why aniline is less basic than ethyl amine or most alkyl amines for that comparison.

And here as well, the pKa values support this reasoning. Because the pKa of CH3CH2NH3+ is higher than the pKa of C6H5NH3+, CH3CH2NH2 is a stronger base than C6H5NH2.
Another factor contributing to the decreased basicity of aromatic amines is the hybridization of the carbon atoms in the aromatic ring. These are sp2 hybridized and sp2 hybridized carbon atoms, which are more electronegative than sp3 carbons because they have more s character.
As a result, they have an electron-withdrawing inductive effect while the aliphatic carbons are electron donors. In general, the more s character, the more electronegative the atom is. These are the percentages of the s orbital (s-character) in each hybrid orbital:

The relationship of the base strength of aniline and cyclohexyl amine is an analogy of comparing the basicity of phenoxide and alkoxide ions. Phenoxide ions are less basic than alkoxide ions for the same reasons mentioned above.
Summarizing this comparison, keep in mind that aromatic amines are considerably weaker bases than aliphatic amines.
What about the basicity of substituted anilines?
Whether a substituted aniline is more or less basic than aniline depends on the nature of the substituent.
Electron-donating groups increase the electron density of the benzene ring, making the arylamine more basic than aniline.
Electron-withdrawing groups decrease the electron density of the benzene ring, making the arylamine less basic than aniline.

As we learned in the ortho, meta, para directing chemistry, the overall effect of the substituent on the basicity of the amine is a combination of resonance and inductive effects.
For example, the decreased basicity of halogen-substituted aryl amines is the result of the electron-withdrawing inductive effect of the electronegative halogen. The closer the chlorine is to the amino group, the less basic it gets because the inductive effect is more pronounced:

Similarly, an alkyl group increases the basicity via an electron-donating inductive effect. For example, p-methyl aniline is more basic than aniline.

Interestingly, though, when the CH3 group is ortho to the amino group, it becomes a slightly weaker base than aniline (pKa 4.62 vs pKa 4.38).
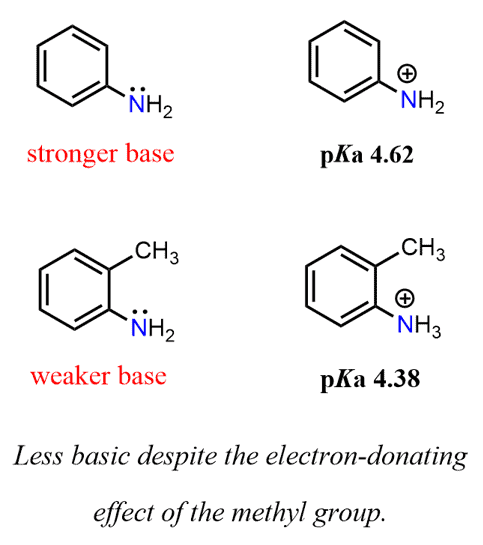
This is likely because of the steric effect, causing, in turn, an electronic effect. What might be happening is that because of the steric hindrance, the nitrogen adopts sp2 hybridization, which puts the lone pair in a p orbital delocalizing it over the aromatic ring and thus making the nitrogen less basic.
Even if this assumption of changing the hybridization is not very accurate, the problem of sterics is going to be decisive once the nitrogen gets protonated. The nitrogen must, in this case, be sp3-hybridized, which brings a lot of steric tension between the CH3 and NH3 groups:
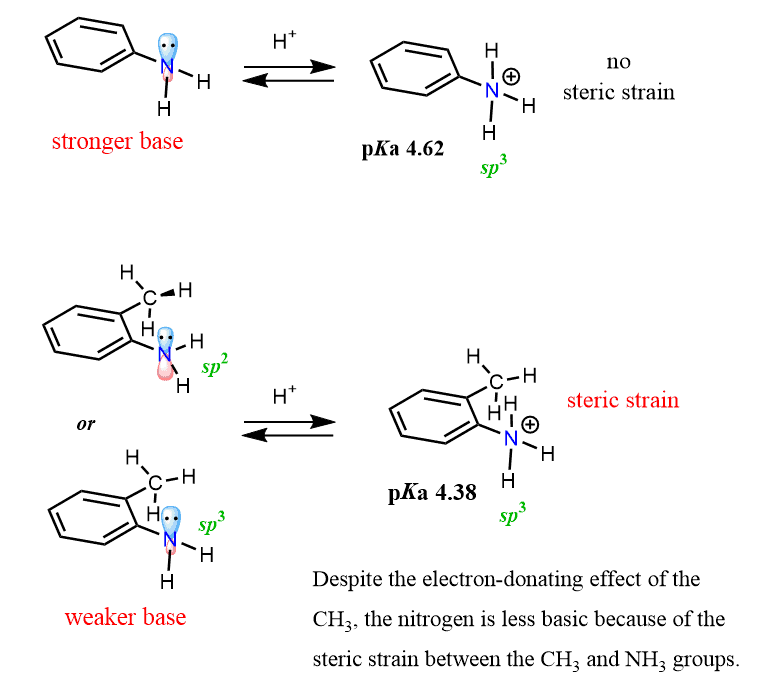
In addition, sp2 hybridized elements are more electronegative, which also makes them less available to capture a proton.
The nitro group decreases the basicity of the aryl amine regardless if it is in the ortho, meta, or para position. However, the effect is most pronounced when it is in the ortho position because, in addition to the resonance effect, which does not change much with the distance, the inductive effect is the strongest when the two groups are next to each other.

The basicity is least affected when the nitro group is in the meta position because, in this case, only the inductive effect influences the electron density in the ring. Check the meta-directing resonance effects of election withdrawing groups here.
Basicity of Heterocyclic Aromatic Amines
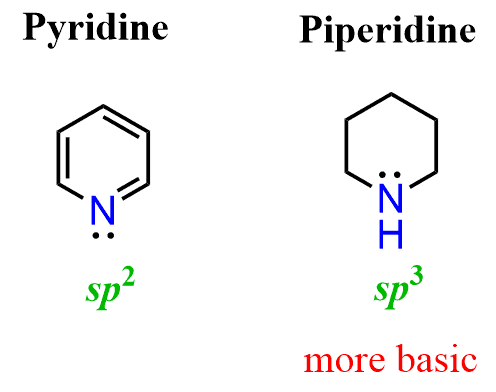
Let’s compare the basicity of pyridine and piperidine. One is an aromatic amine, and the other is a cyclic aliphatic amine.
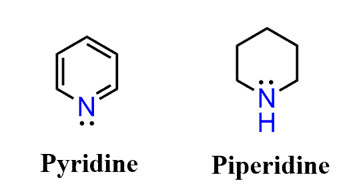
From what we have discussed so far, it should be clear to you that pyridine is going to be the weaker base because of the aromatic ring. And that prediction is in fact accurate – pyridine is a lot weaker a base than piperidine.
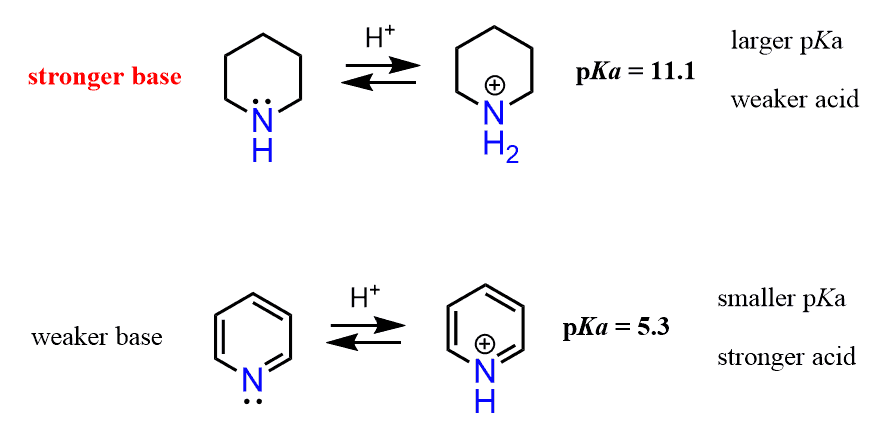
However, the effect of the aromatic ring here is not the same as in aniline. The big difference here, compared to the effect of the aryl ring on the basicity of aniline, is that the lone pair of pyridine is not delocalized over the aromatic system. It is localized since the electrons are in the sp2 orbital, and this orbital is at 90o to the p orbitals of the ring and cannot be part of the conjugated system.
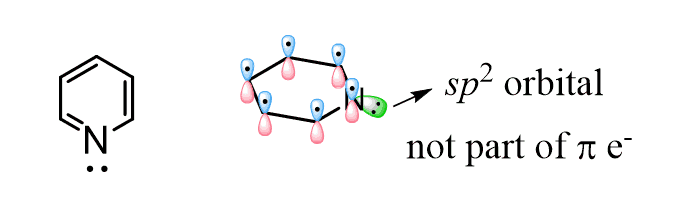
Remember, in order to be conjugated, the orbitals must be parallel and not perpendicular. Now, if the lone pair is localized, what makes pyridine less basic than the one in piperidine?
The answer to this question is the hybridization of the nitrogen. As we mentioned earlier, sp2 orbitals have more s character than sp3 orbitals and therefore, they are more electronegative, which in turn makes them less basic. Notice that the nitrogen in piperidine is sp3 hybridized and thus it is more basic:
The next scenario, then, would be an aromatic amine where the lone pair of the nitrogen is part of the aromatic system, i.e., it is delocalized.
One such good example is pyrrole.
The nitrogen here looks to be sp3 based on the steric number (s.n. = 4: 3 bonds and a lone pair) but because the molecule “wants” to be aromatic”, it changes the hybridization to sp2 thus placing the lone pair in a p orbital and forming a cyclic, planar, fully conjugated system of 6 electrons:
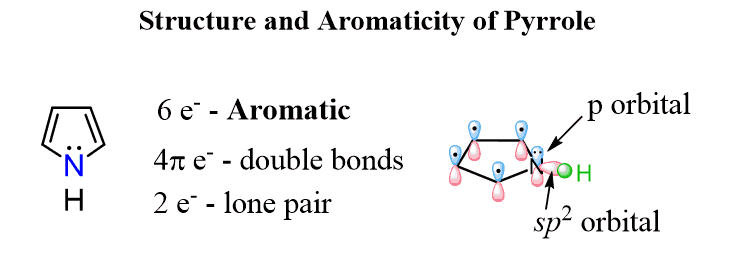
The lone pair on pyrrole, therefore, is delocalized, thus making a five-membered aromatic ring, and as expected, pyrrole is a much weaker base than pyridine.
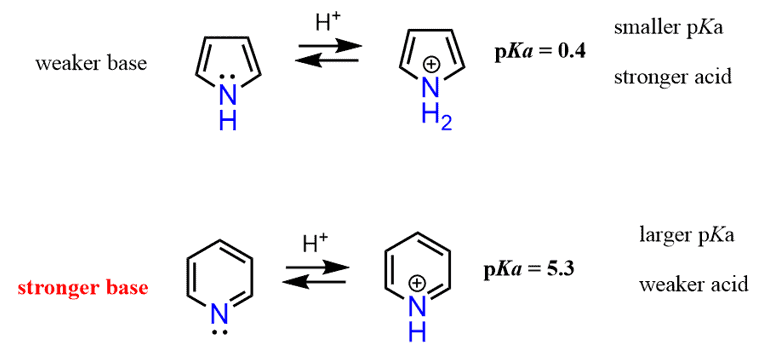
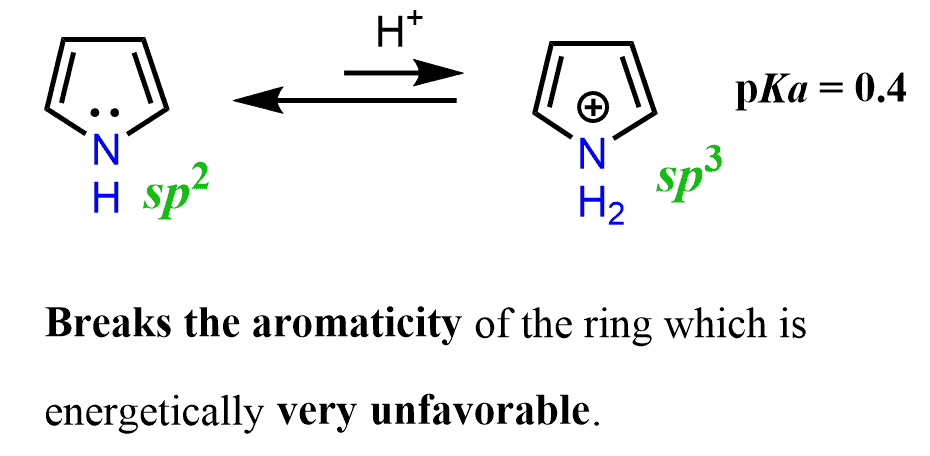
Not only does the protonation of pyrrole occur less readily, but the resulting structure breaks the aromaticity of the ring, which is energetically very unfavorable.
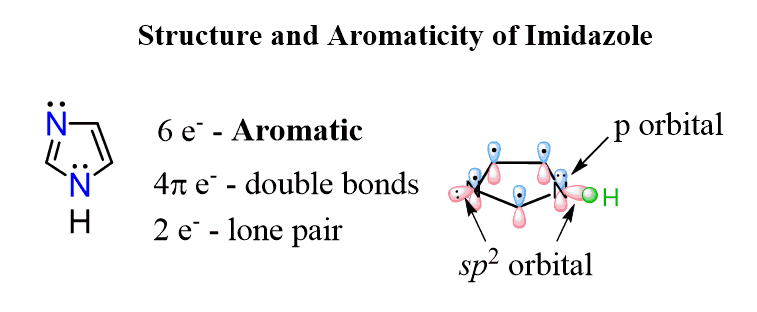
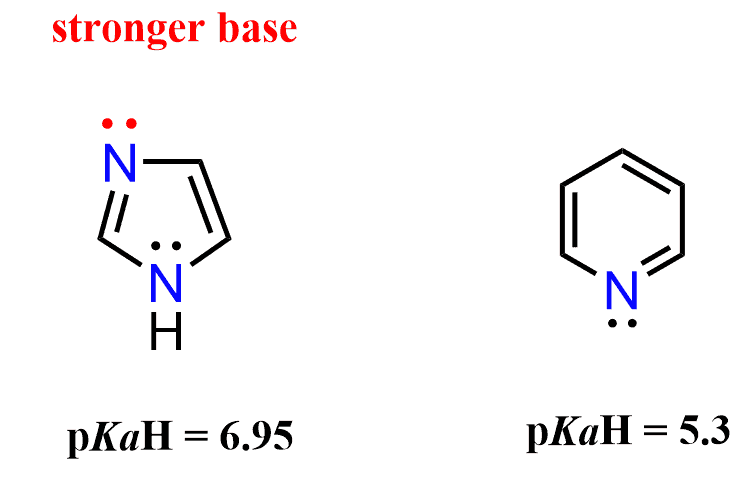
A great example, combining these two types of nitrogens (one with a localized and the other with a delocalized lone pair), is imidazole. It has one lone pair in a perpendicular to the conjugated system sp2 orbital, which is not part of the aromatic system, and another one in a p orbital, and thus part of the aromatic system, because the nitrogen with single bonds is sp2 hybridized, which favors the aromaticity:
Just like pyridine, when treated with an acid, the lone pair that is not part of the aromatic system is protonated, forming the imidazolium ion:
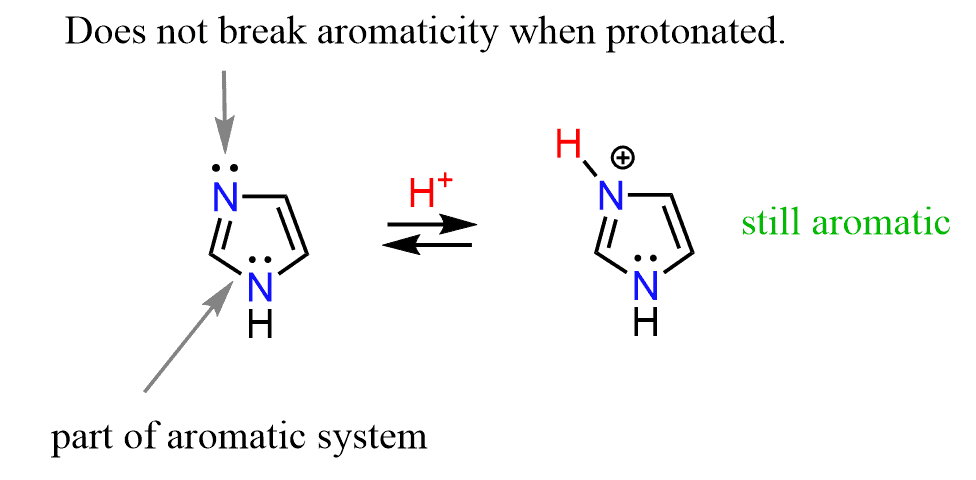
Imidazole is a stronger base than pyridine because the positive charge on the imidazolium ion is stabilized by the resonance effect of the lone pair on the other nitrogen. In other words, the charge is delocalized on both nitrogen atoms of the ring.
Basicity of Amines and Amides
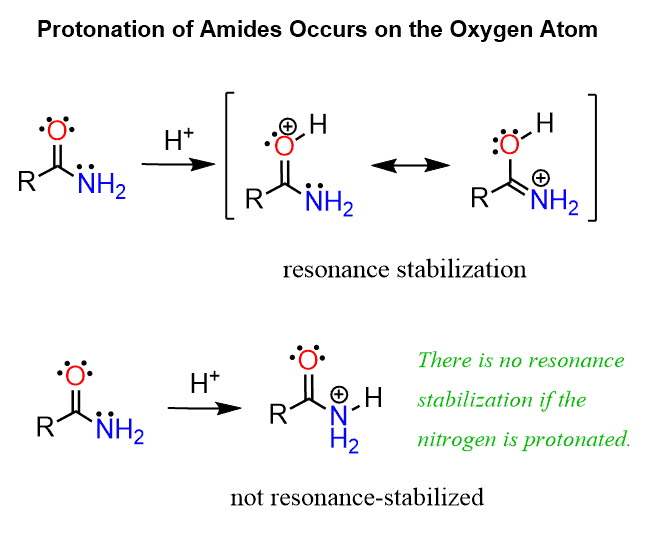
Amides are generally weaker bases than amines. The pKa of the conjugate acid of a typical amide is about zero, and there are two factors contributing to this decreased basicity. One is what we have already mentioned in a few examples, and that is the delocalized nature of the lone pair on nitrogen:

Because of these resonance configurations, the protonation of amides occurs on the oxygen rather than the nitrogen atom. Compare how the oxygen is resonance-stabilized by the lone pair of the nitrogen when it is protonated and the lack of this stabilization if the nitrogen were to be protonated:
The second factor decreasing the basicity going from amines to amides is the electron-withdrawing effect of the carbonyl group in the amide. Remember, the entire alpha carbon chemistry is based on this effect of the carbonyl group, which makes the ɑ proton significantly more acidic than in other hydrocarbons.
In a very brief summary, remember this: increasing the electron density on the nitrogen, in any way, increases its basicity. Therefore, the more electron-donating groups, such as alkyls, you have around the nitrogen, the more basic it gets. On the contrary, electron-withdrawing groups will decrease the basicity of the amine.
Check Also
- Naming Amines: Systematic and Common Nomenclature
- Preparation of Amines
- The Gabriel Synthesis of Primary Amines
- The Hofmann Elimination of Amines and Alkyl Fluorides
- Imines from Aldehydes and Ketones with Primary Amines
- Enamines from Aldehydes and Ketones with Secondary Amines
- The Reaction of Amines with Nitrous Acid
- The Cope elimination
- Reactions of Amines Practice Problems
- Acids and Bases – General Chemistry
- Organic Acids and Bases
- Organic acid-base mechanisms
- Acid Strength and pKa
- How to Determine the Position of Equilibrium for an Acid-Base Reaction
- Inductive and Resonance (Mesomeric) Effects
- Factors That Determine the pKa and Acid Strength
- How Resonance Affects Acidity and Basicity
- How to Choose an Acid or a Base to Protonate or Deprotonate a Given Compound
- Lewis Acids and Bases
- Basicity of Amines
- Organic Acids and Bases Practice Problems
- Organic Acids and Bases Quiz
