Zaitsev’s Rule
Suppose you have this alkyl halide and need to determine all the products of an E2 reaction when it is treated with a strong base such as sodium ethoxide.
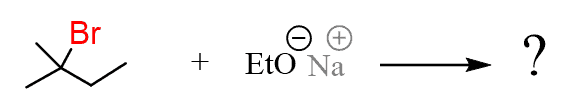
Remember in E2 reactions, we’re first going to identify all the β hydrogens and draw the products of elimination based on each set of these β hydrogens. In this case, we have β-hydrogens on the right side of the leaving group (Br) and on the two methyl groups.
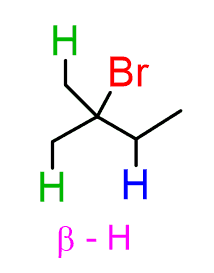
So, one product is going to be the alkene where the double bond is in the middle:
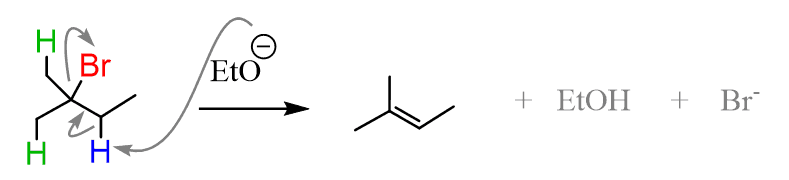
On the other hand, the hydrogens on each methyl group give the same alkene since they are equivalent, as confirmed by the symmetry plane and the structure of the alkenes:
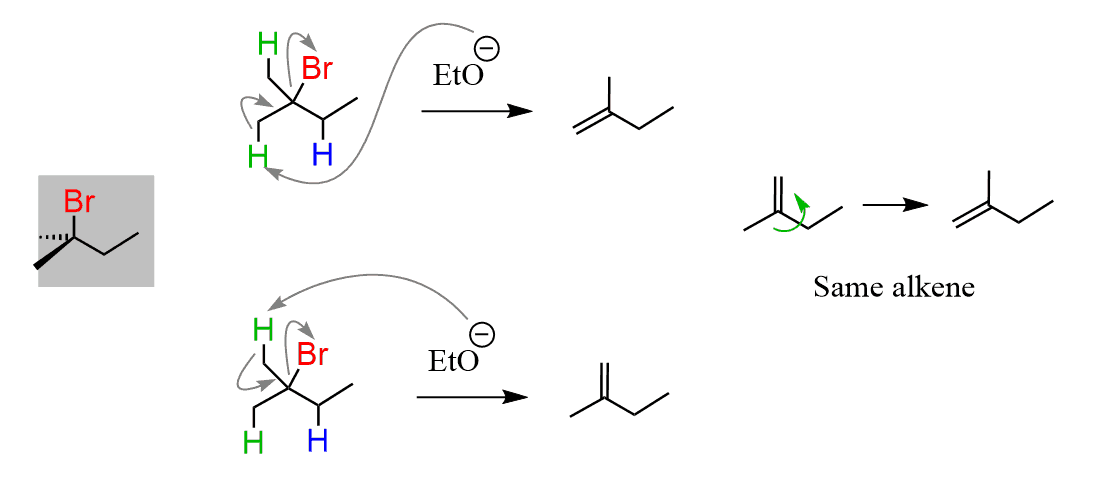
What is interesting is that it turns out that only one of these two alkenes is the major product. The ratio is about 80:20 in favor of the more substituted alkene:

The reason for this is the stability of the alkenes. Remember that the more substituted alkenes are more stable. Adding groups on the double bond increases the stability of the alkene:

Now, checking our products, we can see that it is a mixture of a trisubstituted alkene with a disubstituted alkene, and the trisubstituted alkene is the major product of this elimination reaction:
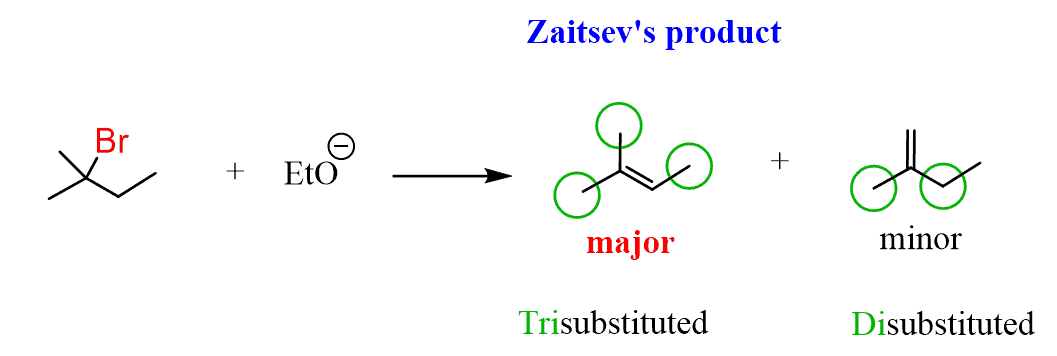
So, the more substituted alkene is the major product of E2 elimination because of its higher stability.
Russian chemist Alexander Zaitsev was the first who observed this pattern, and the reaction is named after him. Zaitsev’s rule states that in an elimination reaction, the more substituted alkene is the major product.
Hoffman Product
Zaitsev’s rule is not always followed in E2 reactions.
For example, if we treat the same alkyl halide with a sterically hindered base (large/bulky), such as, for example, the potassium tert-butoxide, we see the opposite trend. The less substituted alkene is the major product despite the fact that it is less stable.
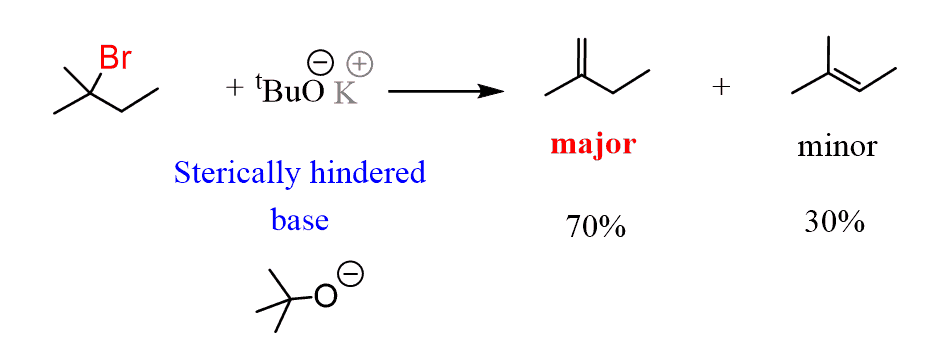
This is known as Hoffman’s rule, which states that if we treat the alkyl halide (or other suitable substrates) with a strong sterically hindered base, the major product is going to be the less substituted alkene.
The reason for this is that it is easier for the bulky base to access the protons that are not hindered by other carbons.
And because the protons of the methyl groups are more accessible than the protons of the CH2 group, the less substituted alkene forms faster and is the major product in this case.
So, to summarize, if you need a more substituted alkene, use a sterically non-hindered base.
If the lees substituted alkene is the target, then use a sterically hindered base.

The most common sterically hindered and non-hindered bases are shown above.
These two rules are very applicable in organic synthesis because they allow us to control what alkene we want to synthesize. And these alkenes can further be used for preparing other products.
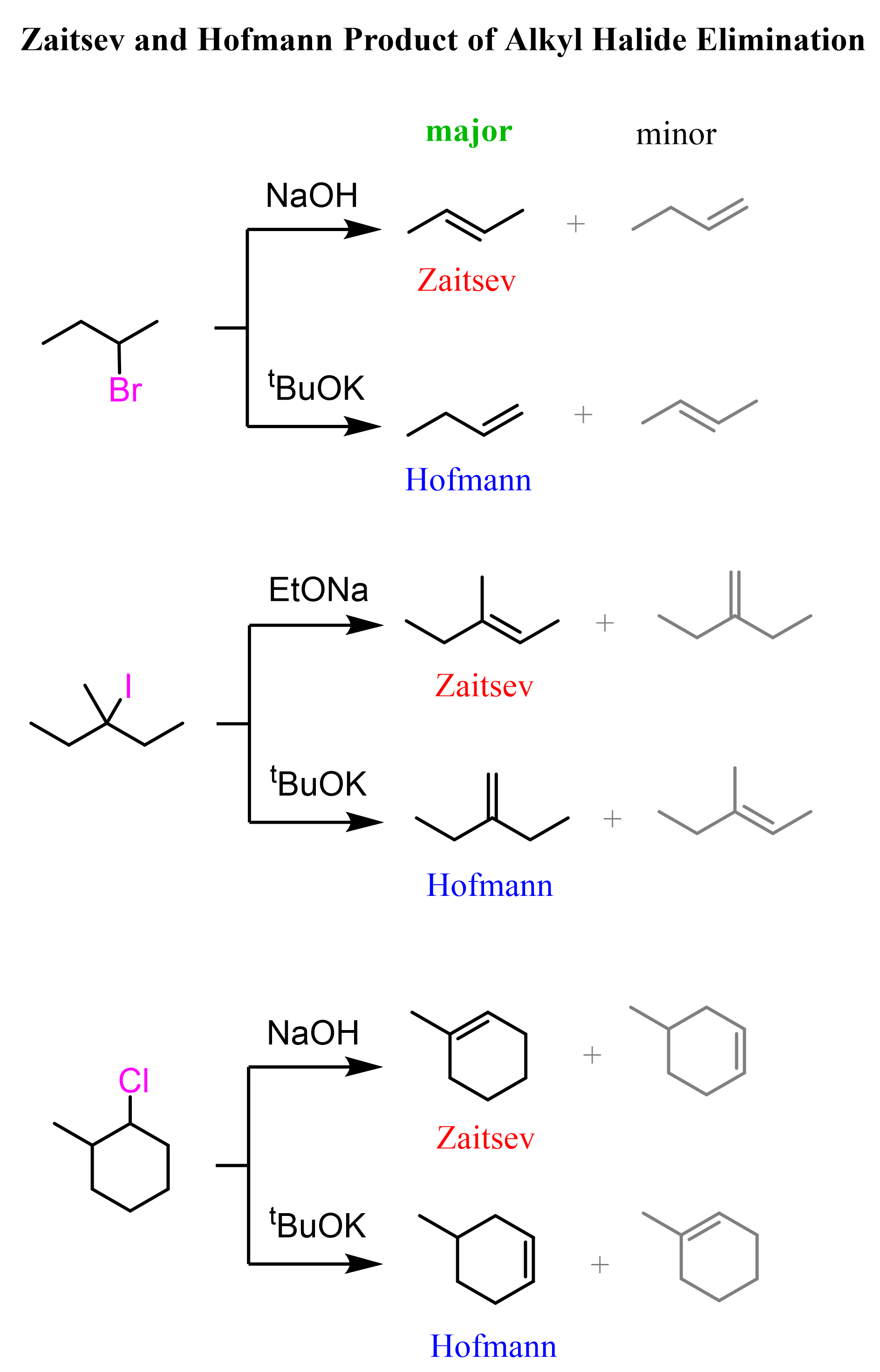
Here are some more examples of Zaitsev and Hofmann elimination achieved by sterically non-hindered and sterically hindered (bulky) bases such as EtO–, –OH, and tBuOK, respectively:
Regioselectivity of E2 Reaction
The Zaitsev’s and Hoffman’s rules demonstrate the principle of regioselectivity in elimination reactions.
Regioselective means that the reaction selectively produces one regioisomer as the major product. The two alkenes are regioisomers since the double bonds are in different regions. That’s one way to remember this, but also notice that the alkenes are constitutional isomers. So regioselectivity is the preferred formation of one constitutional isomer.




Sir I’m form india . Can I get the ORGANIC 1 and 2 guide book 📕. If yes
In what timings I can order it?
Hello,
Both the study guide and the reagent guide are delivered to your e-mail as PDF files and you can order them anytime 24/7.