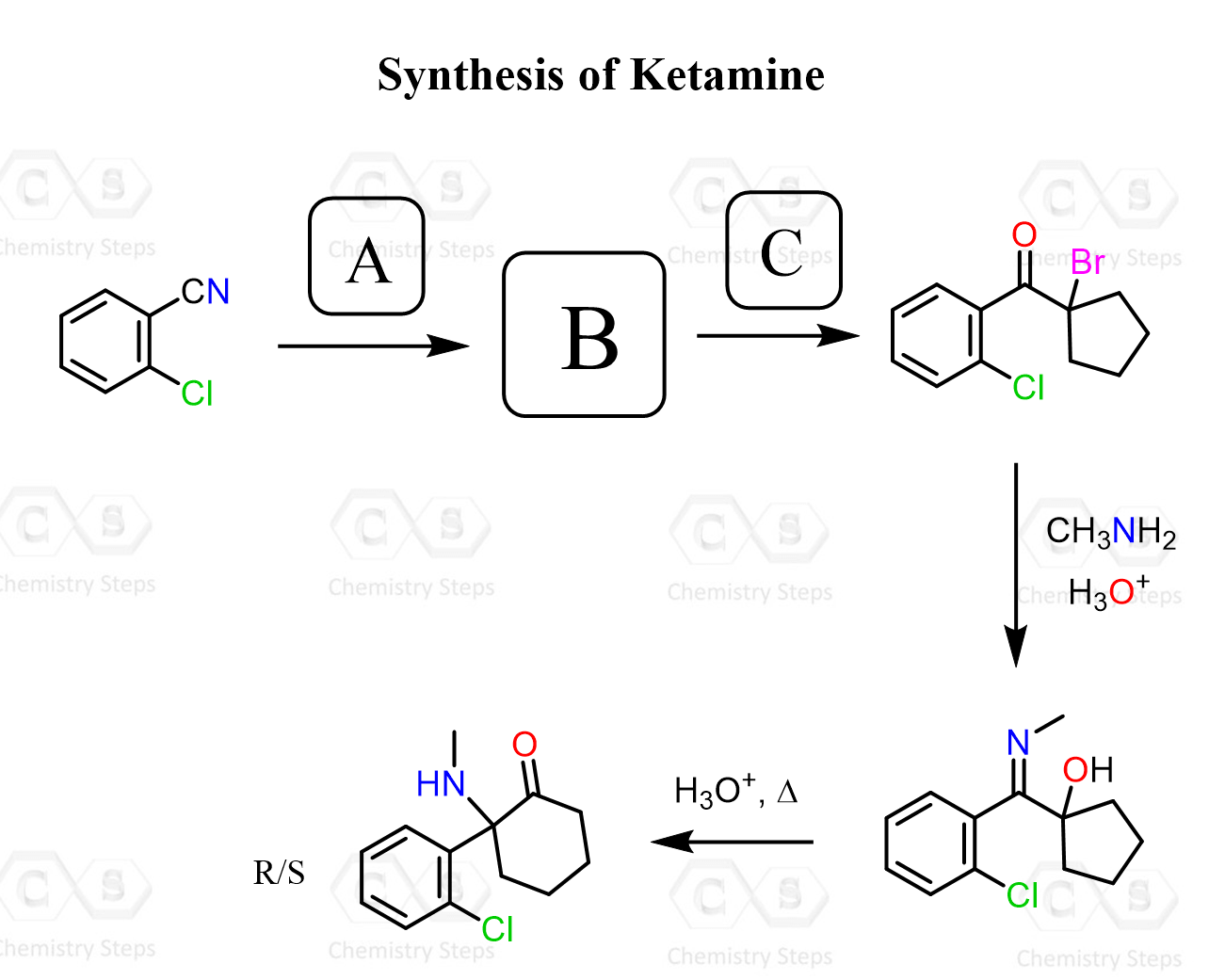
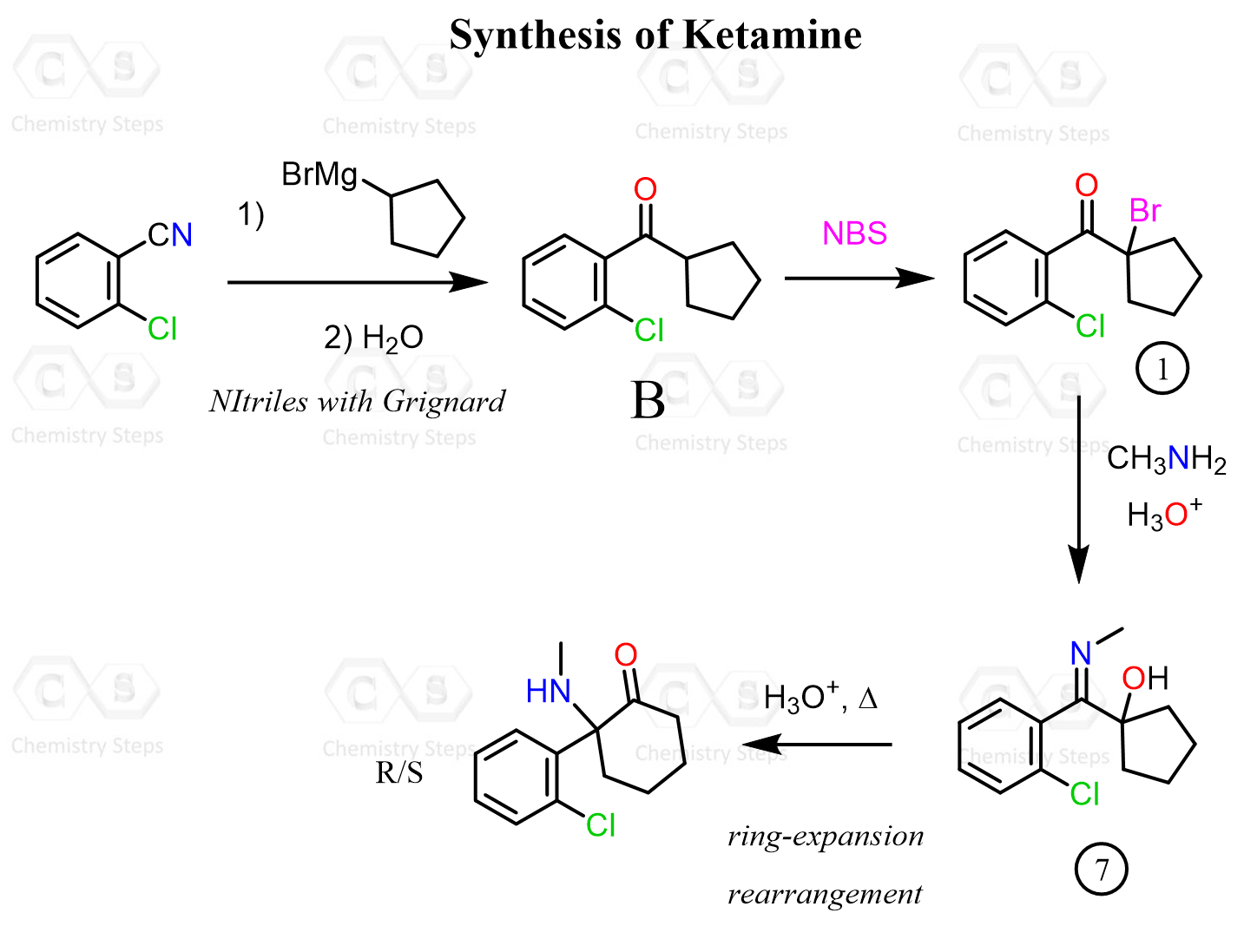
Aldehydes and ketones react with primary amines, in mildly acidic conditions, forming an imine (a Schiff base). The reaction with secondary amines produces enamines:

Just like the reaction with water and alcohols, this is also an addition reaction to the carbonyl group. The nucleophile is the nitrogen with its lone pairs and the electrophile is the carbonyl group.
Let’s discuss the mechanism of this addition reaction, starting with primary amines.
Imines from Primary Amines – The Mechanism
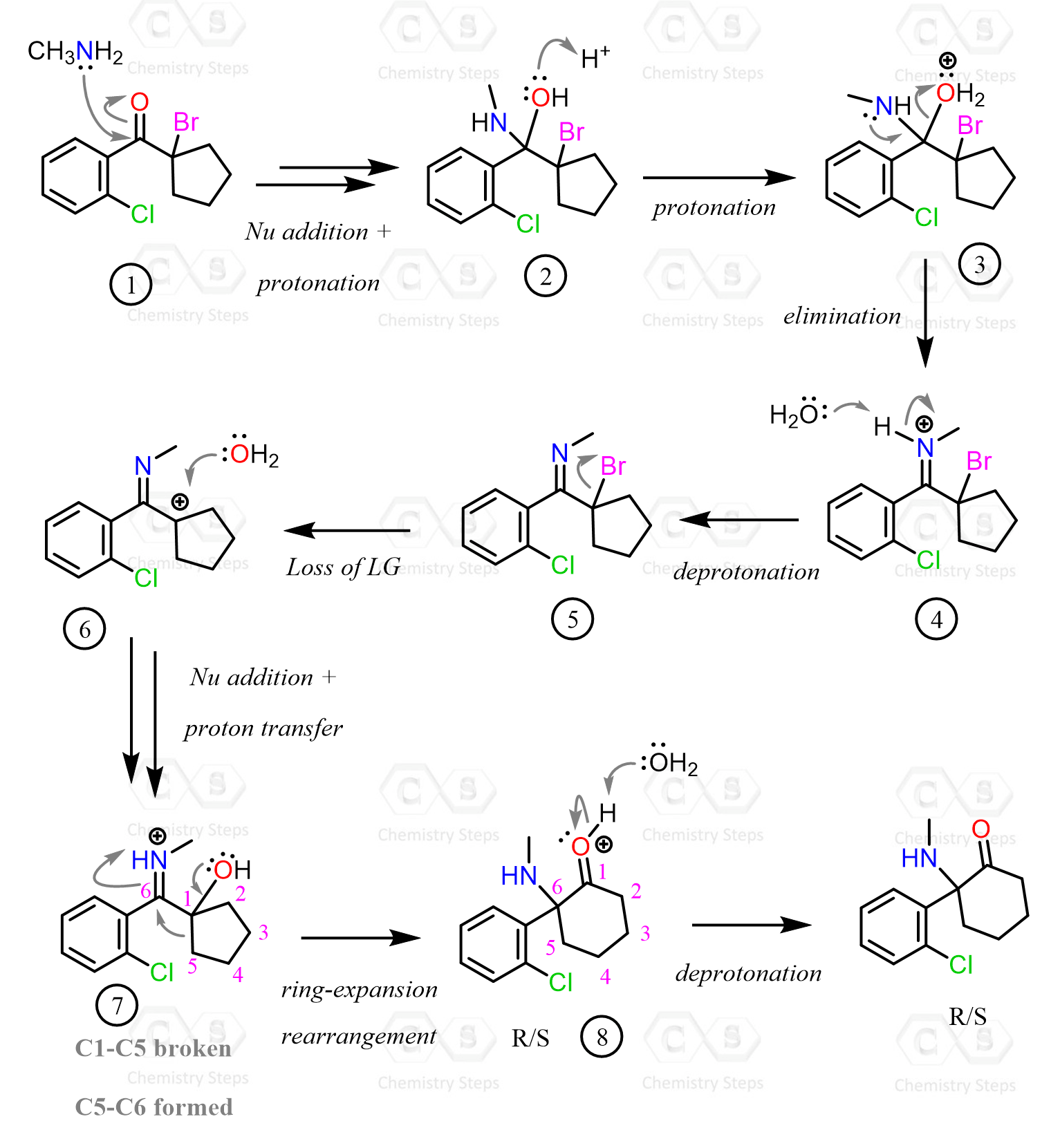
There are two main parts in the net transformation: 1) nucleophilic addition of the primary amine and 2) elimination of H2O.
All the steps are reversible, and the overall equilibrium of the reaction is shifted by removing H2O, as we have also seen in the reaction of aldehydes and ketones with alcohols.
A nucleophilic attack of the amine, followed by two proton transfer steps, produces an unstable intermediate called a carbinolamine:

The oxygen of the carbinolamine is then protonated and expelled by the lone pairs of the nitrogen:

The resulting intermediate (iminium ion) is deprotonated to generate an imine as a mixture of (E) and (Z) isomers when applicable:

And here is the summary of the reaction of primary amines with aldehydes and ketones:

Let’s also focus on a few additional details of this reaction.
First, we mentioned that the reaction is carried out in mildly acidic conditions (pH 4-5). Increasing the acidity as a catalyst does not help. In fact, it slows the reaction down, because the first thing that happens upon the addition of acid is the protonation of the amine itself, forming an ammonium ion:
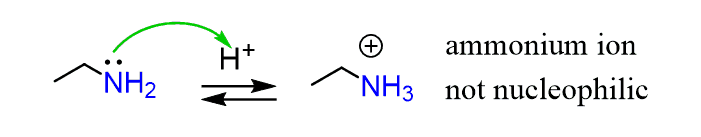
The ammonium ion is, of course, not nucleophilic as it does not have a lone pair.
So, increasing the acidity effectively decreases the concentration of the nucleophile.
The second observation is a change in the order of the steps compared to the reaction with water and alcohols. Those are weaker nucleophiles, and the acid plays the role of a catalyst in the first step of the reaction by coordinating to the carbonyl oxygen:
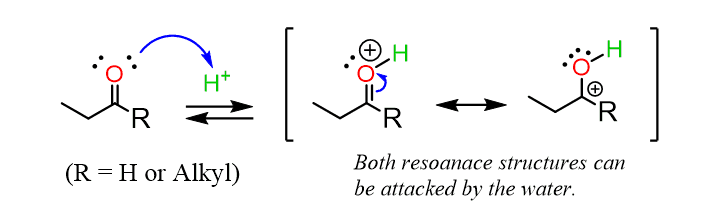
This decreases the electron density of the C=O carbon, thus making it more electrophilic and susceptible to nucleophilic attack.
Applications of Imines
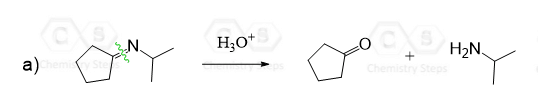
Imine formation is an important reaction both in biological processes as well in organic synthesis. For example, we have seen how it is used in the Wolff-Kishner reaction for the alkylation of aromatic compounds through acylation-reduction:

Imines are also important intermediates in the synthesis of amines by reductive amination using Sodium cyanoborohydride:

The reaction of aldehydes and ketones with secondary amines will be covered in the next post.
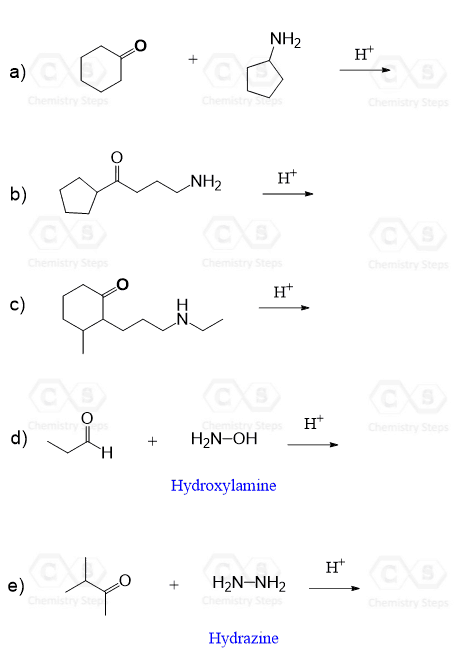
And here are some practice problems you can work on:
Reactions of Aldehydes and Ketones with Amines – Practice Problems