Fischer esterification is the acid-catalyzed reaction of carboxylic acids and alcohols:

Before we get into the details of this reaction, remember that there are other ways of preparing esters. For example, the carboxylic acid can be converted into a carboxylate salt and further react with an alkyl halide via the SN2 mechanism:

Another common way of making esters is the reaction of acyl chlorides with alcohols or alkoxides:

Each of these can be classified as an esterification reaction; however, the Fischer esterification specifically refers to the acid-catalyzed reaction of carboxylic acids and alcohols.
Fischer Esterification Mechanism
The mechanism of Fischer Esterification is an example of nucleophilic addition-elimination, and the overall result of it is the replacement of the OH group by the OR. In general, it is a slow reaction carried out in reflux using a strong acid such as sulfuric or phosphoric acid. When dissolved in alcohol, these strong acids produce the conjugate acid of the alcohol, which then serves as the actual catalyzing acid.
All the steps in Fischer esterification are reversible, and it is often carried out in large excess of alcohol to shift the equilibrium toward the ester product according to the Le Châtelier’s principle. Another strategy to push the equilibrium forward, depending on the physical properties of the components, is the removal of water from the reaction mixture.
In the first step, the electrophilic carbonyl group is activated by protonation of the C=O oxygen, thus making it more reactive towards the nucleophilic attack of the alcohol:

If you have noticed, in the first example reactions and in the mechanism, the color-coded atoms indicate that the oxygen of the RO in the ester comes from the alcohol. And it is confirmed experimentally, using the isotope label method, that the ester does inherit the oxygen from the alcohol.
For the isotope label method, you simply use an 18O-containing alcohol, and if the molecular mass of the ester corresponds to this change, it proves the suggested mechanism of Fischer esterification:

More Details and Limitations of Fischer Esterification
A few interesting factors to consider in the Fischer esterification.
First, we know that alcohols are not the greatest nucleophiles, and this would be especially relevant in such strong acidic conditions where a significant portion of the OH groups is in the conjugate acid form.
This explains why Fischer esterification is not the optimal way of making phenol esters. Phenol is a quite strong acid (pKa 10 vs pKa 16 of regular alcohols), which also indicates its conjugate base is a weaker base and a weaker nucleophile.

This has to do with the delocalized character of the lone pairs of the oxygen, which are in resonance with the aromatic ring. Again, this makes it even a weaker nucleophile than other alcohols, which is especially true in acidic conditions.
Another limitation of Fischer esterification is the fact that tertiary alcohols undergo a “fast” dehydration in the presence of strong acids:

In these situations, alternative strategies such as the higher reactivity of acid chlorides and the carboxylate salts, discussed above, can be used to prepare the desired ester.

There is another common question about the first step of Fischer esterification;
Can the OH oxygen be protonated instead of the carbonyl oxygen?

What you need to remember is that the carbonyl oxygen, in general, is a stronger Lewis base than the neighboring OH.
The lone pairs on the hydroxyl can serve as a Lewis base, but the intermediate formed in this case is not resonance-stabilized, while the positively charged carbonyl oxygen is resonance-stabilized:
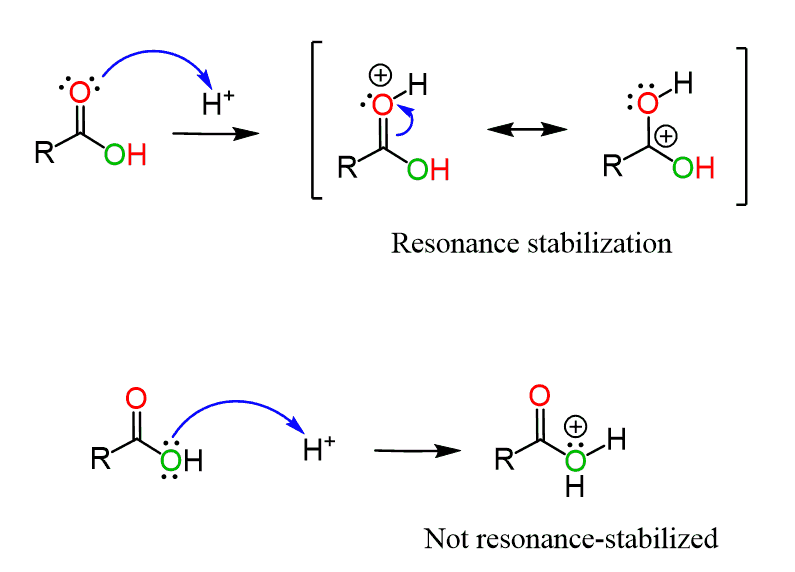
We have also talked about this selectivity when discussing the mechanism of thionyl chloride (SOCl2) with carboxylic acids.
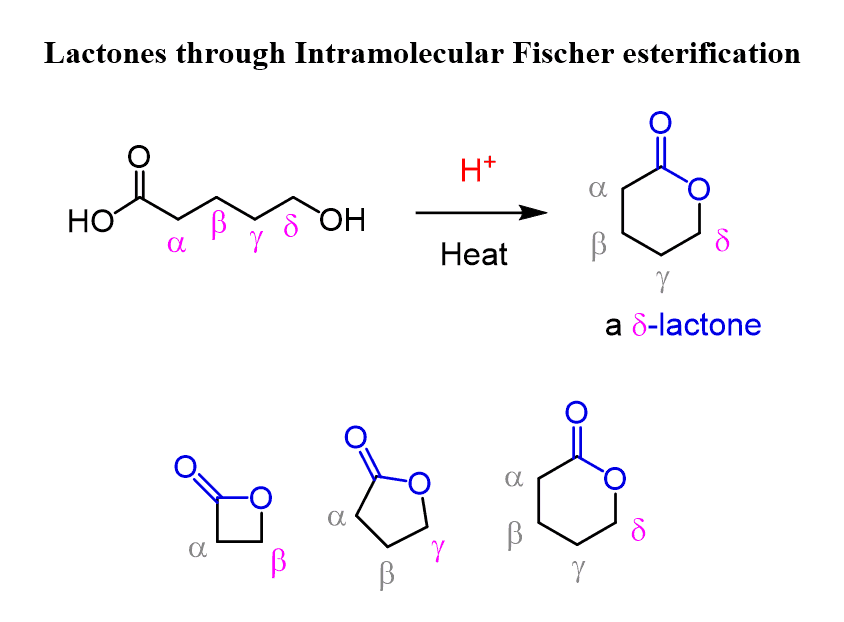
Lactones through Intramolecular Fischer Esterification
Intramolecular Fischer esterification is also possible, especially when a five- or six-membered ring is formed. These cyclic esters are called lactones, and depending on the position of the hydroxyl group, four, five, and six-membered rings can be prepared. These are classified as β-lactones, γ-lactones, and δ-lactones, respectively:
Need some good practice on the reactions of carboxylic acids and their derivatives?
Check this 45-question, Multiple-Choice Quiz with a 50-min Video Solution covering the reactions of acids, esters, lactones, amides, acid chlorides and etc.
Carboxylic Acids and Their Derivatives Quiz
Check Also
- Preparation of Carboxylic Acids
- Naming Carboxylic Acids
- Naming Nitriles
- Naming Esters
- Naming Carboxylic Acid Derivatives – Practice Problems
- Ester Hydrolysis by Acid and Base-Catalyzed Hydrolysis
- What is Transesterification?
- Esters Reaction with Amines – The Aminolysis Mechanism
- Ester Reactions Summary and Practice Problems
- Preparation of Acyl (Acid) Chlorides (ROCl)
- Reactions of Acid Chlorides (ROCl) with Nucleophiles
- R2CuLi Organocuprates – Gilman Reagent
- Reaction of Acyl Chlorides with Grignard and Gilman (Organocuprate) Reagents
- Reduction of Acyl Chlorides by LiAlH4, NaBH4, and LiAl(OtBu)3H
- Preparation and Reaction Mechanism of Carboxylic Anhydrides
- Amides – Structure and Reactivity
- Naming Amides
- Amides Hydrolysis: Acid and Base-Catalyzed Mechanism
- Amide Dehydration Mechanism by SOCl2, POCl3, and P2O5
- Amide Reduction Mechanism by LiAlH4
- Reduction of Amides to Amines and Aldehydes
- Amides Preparation and Reactions Summary
- Amides from Carboxylic Acids-DCC and EDC Coupling
- The Mechanism of Nitrile Hydrolysis To Carboxylic Acid
- Nitrile Reduction Mechanism with LiAlH4 and DIBAL to Amine or Aldehyde
- The Mechanism of Grignard and Organolithium Reactions with Nitriles
- The Reactions of Nitriles
- Converting Nitriles to Amides
- Carboxylic Acids to Ketones
- Esters to Ketones
- Carboxylic Acids and Their Derivatives Practice Problems
- Carboxylic Acids and Their Derivatives Quiz
- Reactions Map of Carboxylic Acid Derivatives


I would just like to say that I am beyond thankful for this page!!! At my ordinary exam I failed (F) because I simply didn’t understand anything during the semester. My teacher and my instructor were both not-good at their jobs, which also affected my interest. Within 10 day, I had my re-exam and I passed with a C (super proud!!). Without even getting comments from my teacher after the exam, I could spot my mistakes and why my original answers not always were right. Thank you, thank you, thank you!!
This truly makes my day. Congratulations on passing the class and good luck in tackling the future challenges to achieve your goals!
Thanks!
Everything is super clear, it helped me a lot! 👍👍👍
Glad to hear that, Sara.