Addition of Organolithiums to Carboxylic Acids
One of the most common ways of converting carboxylic acids to ketones is their reaction with organolithiums.

The first step is the deprotonation of the acid since organolithiums are also very strong bases. What is interesting about this reaction is that we cannot perform a similar acyl substitution with virtually any other nucleophile because of the restricted reactivity of the carboxylate ion generated upon the initial acid-base reaction:
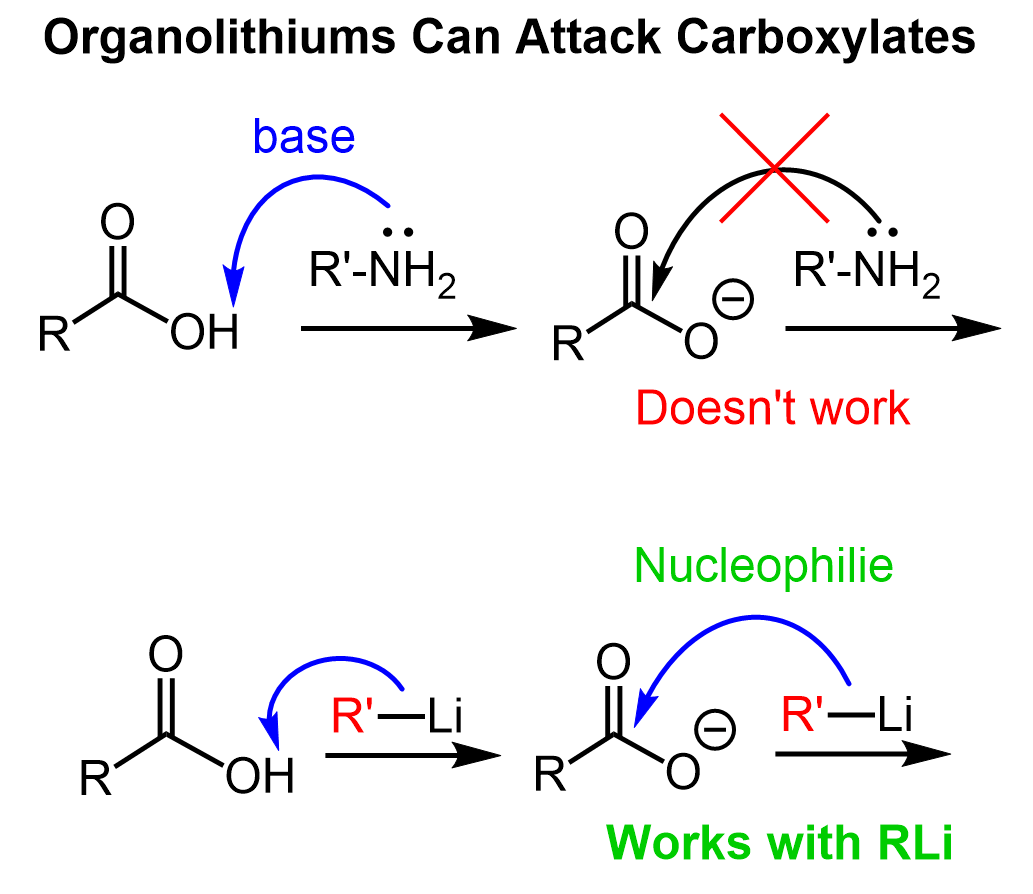
Organolithiums, however, are extremely powerful bases and nucleophiles capable of adding to the carboxylate ion. This addition forms a tetrahedral dianion intermediate which lacks a relatively good leaving group and stays in the solution until water is added to hydrolyze it the corresponding ketone:

An interesting variation of this method of converting a carboxylic acid to a ketone was reported in a recent publication where the authors use DCC (N,N’-dicyclohexane carbodiimide) and an organolithium:

We have seen that DCC is a coupling reagent used mainly for the conversion of carboxylic acids to amides. The role of DCC is to convert the OH into a good leaving group, and, in the case of amide synthesis, to prevent the acid-base reaction of the acid with the amine. In simple graphics, we can visualize the role of DCC as follows:

The mechanism of DCC and EDC coupling in amide synthesis is covered in this article.
So, for the conversion of carboxylic acids to ketone, the nucleophile is changed from an amine to an organolithium which is even a stronger base than any amine, DCC prevents the acid-base reaction.
In the first step, the imine (DCC) deprotonates the carboxylic acid and becomes highly electrophilic which promotes a nucleophilic attack by the carboxylate group forming the DCC-activated intermediate which, after the nucleophilic addition-elimination adds to the carbonyl and thus, forming a ketone:

The reaction is quite applicable as it is reported to work with aliphatic, aromatic, and heteroaromatic acids.
Using Organocuprates
The OH group of carboxylic acids can also be turned into a good leaving group via acid chlorides. These are the most reactive among carboxylic acid derivatives and can easily be converted into alcohols and, more importantly for today’s post, into ketones.
For example, Grignard reagents will react with acyl chlorides to produce alcohols, while organocuprates (Gilman’s) also known as lithium dialkyl cuprates can be used for making ketones from acyl chlorides:
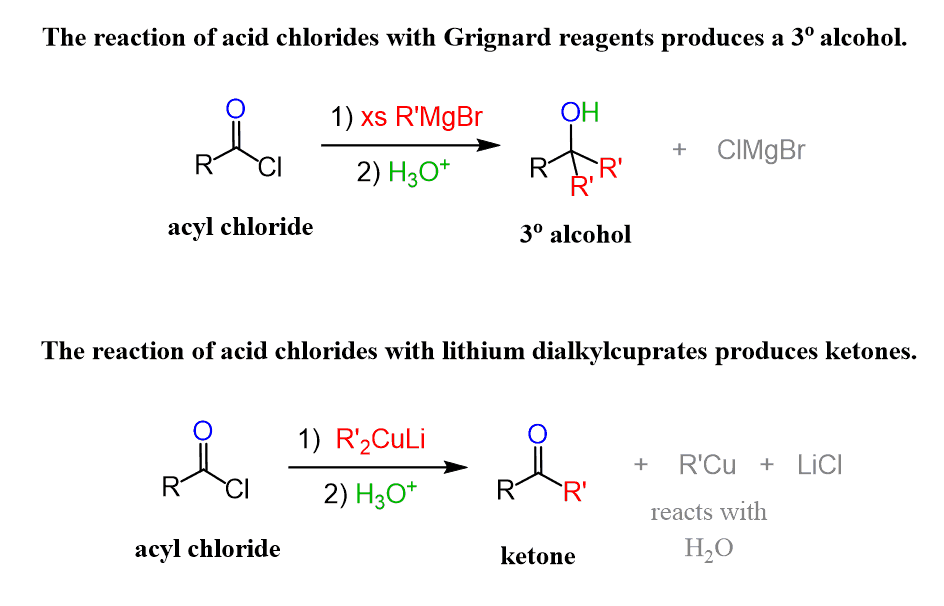
The question is – how come the Grignard and Gilman reagents react differently with acid chloride?
The short answer is that the Gilman reagent is less reactive than Grignard because the alkyl groups are connected to copper rather than magnesium. This makes their carbanionic character less pronounced since the C−Cu bond is less polarized than a C−Mg bond.
Acyl chlorides can be prepared by reacting carboxylic acids with thionyl chloride (SOCl2), phosphorous trichloride (PCl3), or phosphorous pentachloride (PCl5):

The overall strategy then for converting a carboxylic acid into a ketone can be shown as follows:

You can find more about the reaction of acid chlorides with organometallics here.
Weinreb Ketone Synthesis
Another method for converting carboxylic acids to ketones is the use of Weinreb amide (N-methoxy-N-methyl amides) which are most often prepared by a nucleophilic acyl substitution of acid chloride.
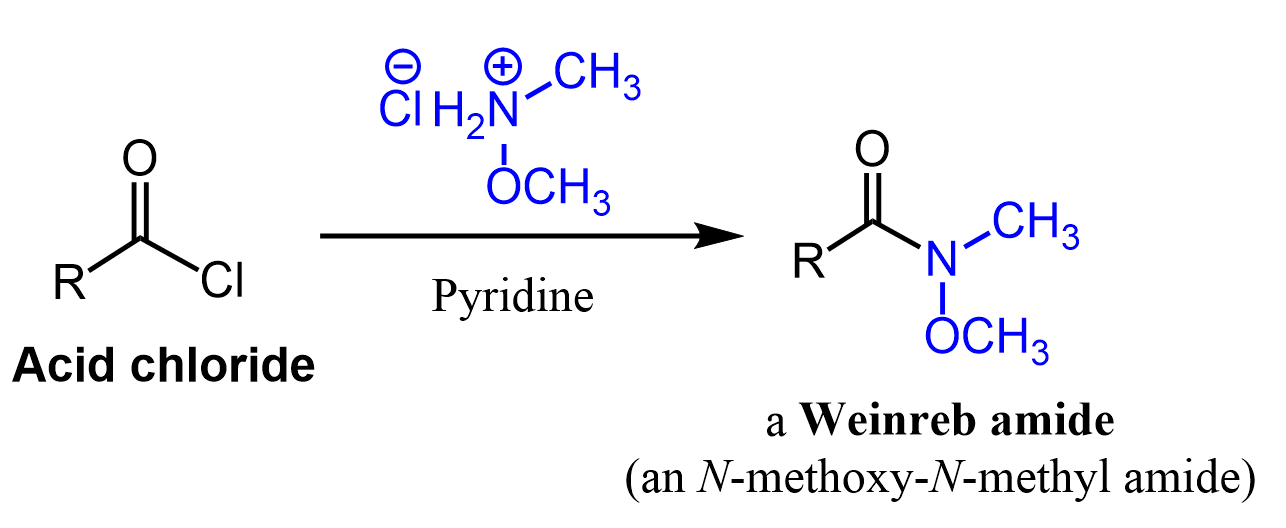
The first step is the nucleophilic addition of the organolithium or organomagnesium to the amide. The tetrahedral intermediate, formed after the addition of organolithium or organ magnesium (Grignard) reagents to the Weinreb amide, is stabilized by chelation of the magnesium atom by the two oxygen atoms.

Like the dianion intermediate in the direct reaction of carboxylic acids with organolithiums, this intermediate only expels the best leaving group upon acidic hydrolysis giving the desired ketone.
Weinreb amide is quite stable and can be purified and isolated which makes it a great precursor for different reactions.
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Preparation of Carboxylic Acids
- Naming Carboxylic Acids
- Naming Nitriles
- Naming Esters
- Naming Carboxylic Acid Derivatives – Practice Problems
- Fischer Esterification
- Ester Hydrolysis by Acid and Base-Catalyzed Hydrolysis
- What is Transesterification?
- Esters Reaction with Amines – The Aminolysis Mechanism
- Ester Reactions Summary and Practice Problems
- Preparation of Acyl (Acid) Chlorides (ROCl)
- Reactions of Acid Chlorides (ROCl) with Nucleophiles
- The reaction of Acyl Chlorides with Grignard and Gilman (Organocuprate) Reagents
- Reduction of Acyl Chlorides by LiAlH4, NaBH4, and LiAl(OtBu)3H
- Preparation and Reaction Mechanism of Carboxylic Anhydrides
- Amides – Structure and Reactivity
- Naming Amides
- Amides Hydrolysis: Acid and Base-Catalyzed Mechanism
- Amide Dehydration Mechanism by SOCl2, POCl3, and P2O5
- Amide Reduction Mechanism by LiAlH4
- Amides Preparation and Reactions Summary
- Amides from Carboxylic Acids-DCC and EDC Coupling
- The Mechanism of Nitrile Hydrolysis To Carboxylic Acid
- Nitrile Reduction Mechanism with LiAlH4 and DIBAL to Amine or Aldehyde
- The Mechanism of Grignard and Organolithium Reactions with Nitriles
- Carboxylic Acids and Their Derivatives Practice Problems
- Carboxylic Acids and Their Derivatives Quiz