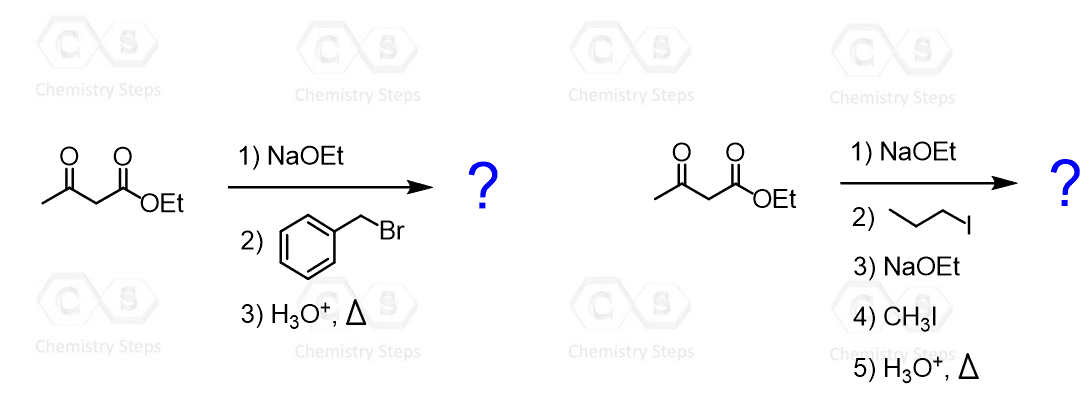
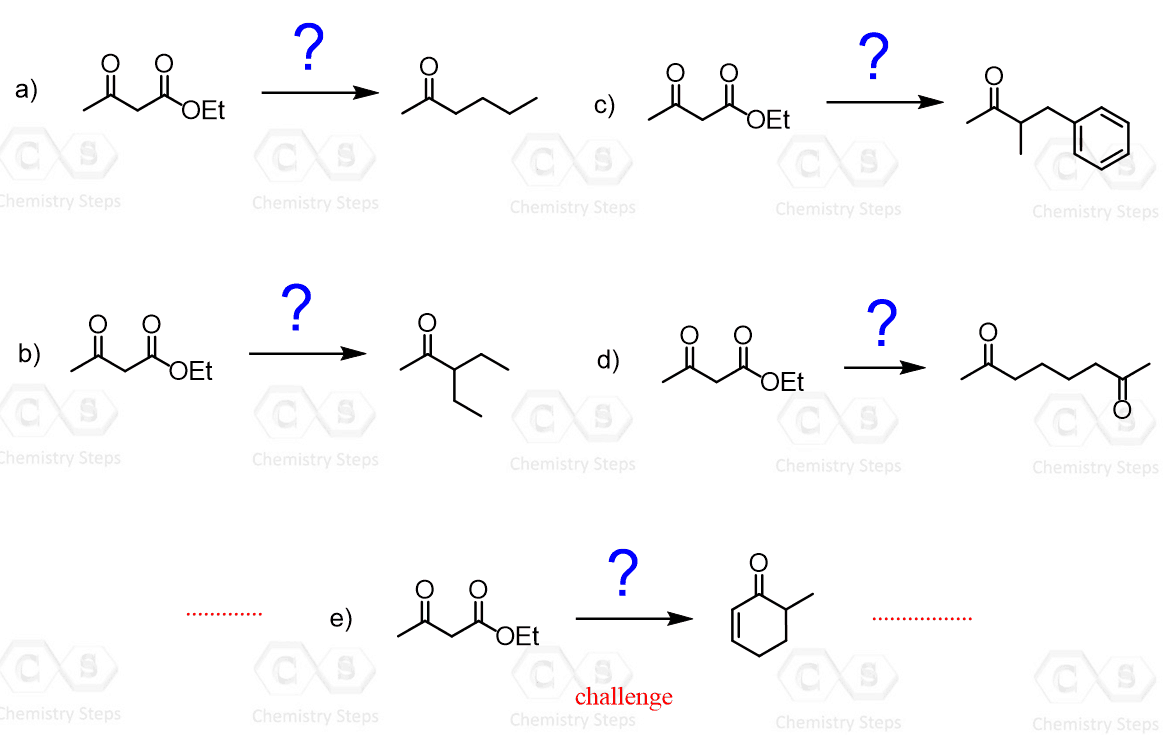
In the previous post, we discussed the acetoacetic ester synthesis, which is a method for preparing substituted methyl ketones via alkylation of the acetoacetic ester followed by hydrolysis and decarboxylation. Below are some practice problems on the acetoacetic ester synthesis to help reinforce the key steps, reaction sequence, and product prediction.

hello Professor: I would like your opinion as to an alternate synthesis I have for your challenge problem above. I started as you did by producing the enolate ion and then adding a methyl group. From there I added 3-bromo-1-propanol, oxidized the OH group to an aldehyde, and then ran an Aldol using ethoxide using the methyl group on the keto portion of the AAE (acetoacetic ester). I hydrolyzed the ester, and then heated removing the carboxylate group and dehydrating the beta-hydroxy ketone I formed yielding the product.
I would like to know if my concepts are clear even if there is a mistake somewhere in the synthetic route.
Thanks for your time. A great web-site!
Hello,
Sounds like a feasible plan where only the decarboxylation step is done later. You can go ahead and email me (gevorg at chemistrysteps.com) your synthesis scheme so we can double-check.