Acid-Catalyzed hydration is the addition of water to an alkene which forms an alcohol:

The reaction goes through a stepwise mechanism which starts with the protonation of the double bond:

The presence of an acid is necessary as the water by itself is a weak acid and cannot protonate the double bond. However, the hydronium ion, formed in acid solutions, is strong enough and the electron-rich double bond attacks it to form a carbocation. Once the carbocation is formed, it can now be attacked by the water:

After the deprotonation of the hydronium ion, the corresponding alcohol is formed as the final product:
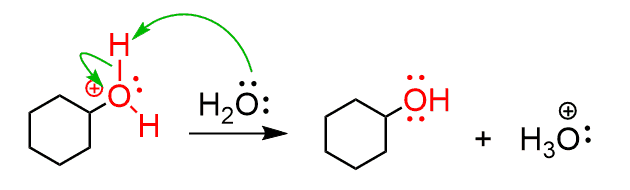
All the steps of acid-catalyzed hydration of alkenes are reversible (and most reaction are; it is a matter of how large the equilibrium constant is) and we only show them with one arrow as we are interested in the forward reaction and trying to shift the equilibrium to that side.

The protonation of the double is the rate-determining step as the π bond of the alkene and the σ bond in the hydronium ion are broken but only one bond is formed.
Notice also, that a dilute acidic solution is used to catalyze the reaction since the presence of a strong acid would cause E1 elimination of the alcohol:

Regiochemistry of Acid-catalyzed hydration
We have seen that the acid-catalyzed hydration of the cyclohexene produces one alcohol as the major product. This is because cyclohexene is a symmetrical alkene and there is no preference/difference as to where we add the OH group. However, if an unsymmetrical alkene is used, there are two possibilities as to where you can put the OH group:

Which of the alcohols is the major product?
As you may have noticed, this mechanism is similar to the addition of HX acids to alkenes according to the Markovnikov’s rule. Remember, for an unsymmetrical alkene, the more stable carbocation determines the major product of the reaction:

In this case, the first carbocation is the more stable intermediate and dictates the formation of the corresponding alcohol:

So, in summary, the regioselectivity of acid-catalyzed hydration indicates that the OH group goes to the more substituted carbon.

Rearrangements in alkene hydrations
Formation of carbocation intermediates in acid-catalyzed hydration of alkenes brings the possibility of rearrangements just like we have seen in SN1 and E1 reactions.
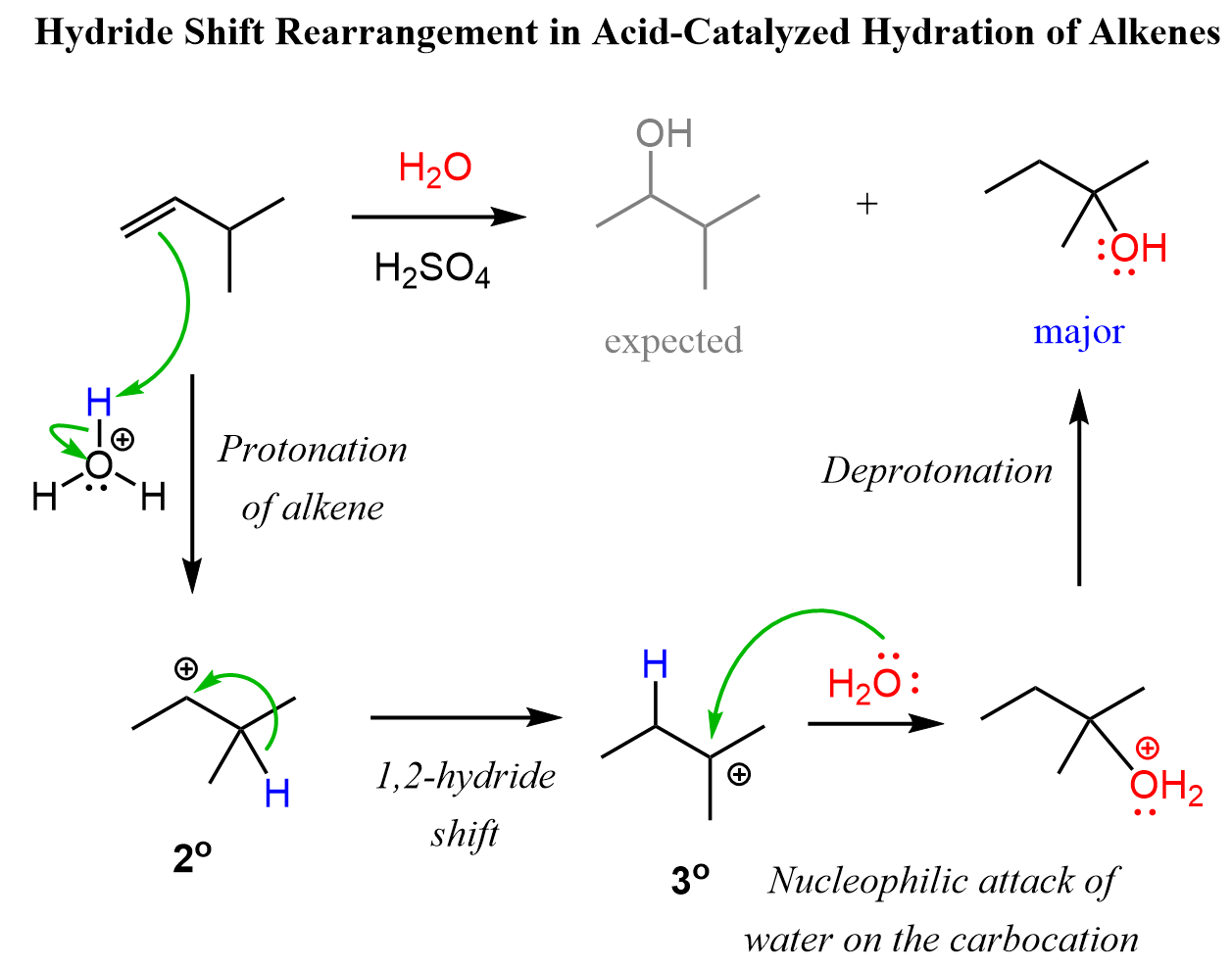
For example, the major product of the following hydration reaction is not the following alcohol as you may expect based on the Markovnikov’s rule:

The secondary alkyl halide is expected because of the secondary carbocation’s greater stability than the primary. However, it is rearranged to a tertiary carbocation via a hydride shift rearrangement to form the more stable tertiary carbocation:

The nucleophilic attack of the water followed by deprotonation results in a tertiary alcohol as the major product:

Let’s also summarize all the steps of an acid-zatalyzed hydration of an alkene involving a hydride shift rearrangement in one scheme:
Rearrangements are generally not desired as they produce unpredictable products. The Oxymercuration-Demercuration reaction is used to avoid them during alkenes’ hydration.
Stereochemistry of Acid-catalyzed hydration
If acid-catalyzed hydration produces an alcohol with one chirality center, then the products will be a mixture of two enantiomers:

The reason for this is the same as you learned in SN1 reactions: carbocations are sp2-hybridized, flat centers (we are talking about the positively charged carbon) and the nucleophilic attack occurs from both faces:
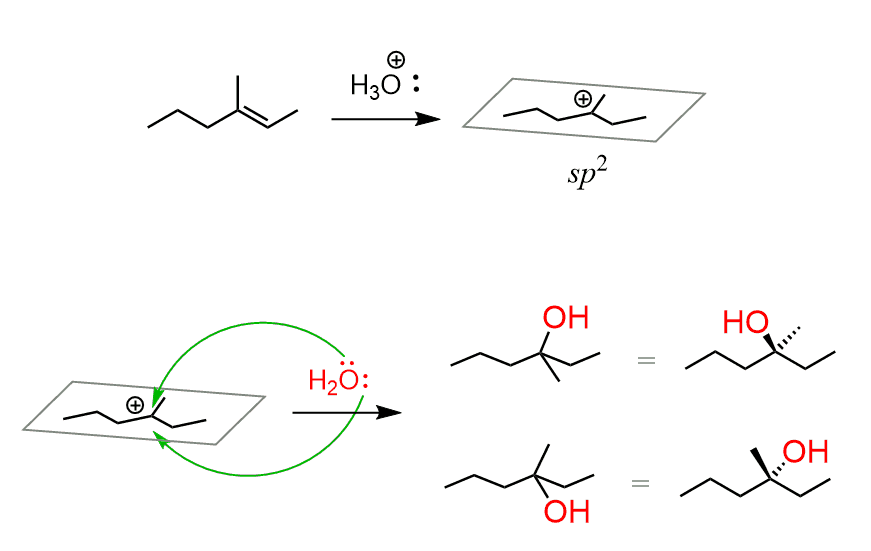
If, on the other hand, the reaction produces more than one stereogenic center, then all the possible stereoisomers with two stereogenic centers are formed. This becomes complicated, and you will most likely not need it in your class.
However, if you do, check the following post for the details:

Why is it that you write sulfuric acid as a reagent instead of writing hydronium when you use hydronium in the mechanism?
The hydronium ion (H3O+) is formed in acidic solutions as a result of protonation of water and it represents the presence of both water and an acid. On the reaction scheme, you can show either H2SO4/H2O or H3O+. The H3O+ is better for showing the mechanism but one problem with showing it on its own is that it does not specify the acid and not every acid is suitable for this reaction.
Could you remind me why there’s no stereochemistry for answers B and C?
The stereochemistry has been added.