Markovnikov’s rule is a general trend predicting that in the reactions of HX (HCl, HBr, HI) species with unsymmetrical alkenes, the X adds to the more substituted carbon of the double bond. The reason for this is the greater stability of the more substituted carbocation intermediate formed after the addition of the hydrogen:

Looking from the perspective of hydrogen, Markovnikov’s rule can also be phrased as: In the addition of HX to an unsymmetrical alkene, the H atom is added to the less substituted carbon to form the more stable, more substituted carbocation.
Although Markovnikov’s rule was first observed and formulated for the hydrohalogenation of alkenes (addition of HCl, HBr, and HI), it is now a broad pattern to classify many addition reactions to alkenes and alkynes as a “Markovnikov” or “anti-Markovnikov” addition. It is now essentially a term to describe the regiochemistry of addition reactions to alkenes and alkynes.

We have also included the hydration reactions of alkenes and alkynes, and even though these are separate topics to discuss, you can see the general pattern of Markovnikov and anti-Markovnikov additions to alkenes and alkynes. Notice that for alkynes, the regioselectivity is only relevant for terminal triple bonds where one of the carbons is connected to hydrogen. In internal alkynes, both carbons of the triple bond are connected to one carbon; we don’t have a more substituted carbon atom
Exceptions to Markovnikov’s Rule
In general, deviations from Markovnikov’s rule are observed only when 1) HBr is used in the presence of a peroxide which changes the reaction to a radical mechanism and 2) the intermediate carbocation undergoes a rearrangement.
While the radical hydrobromination is an anti-Markovnikov addition as the Br ends up on the less substituted carbon, rearrangements are still a type of Markovnikov addition because the X component adds to an even more substituted carbon atom next to the double bond:

The reason for this is that in the radical mechanism, Br radical adds to the double bond first, forming the more substituted radical intermediate, whereas in the ionic mechanism, the protonation occurs first, favoring the stability of the more substituted carbocation.
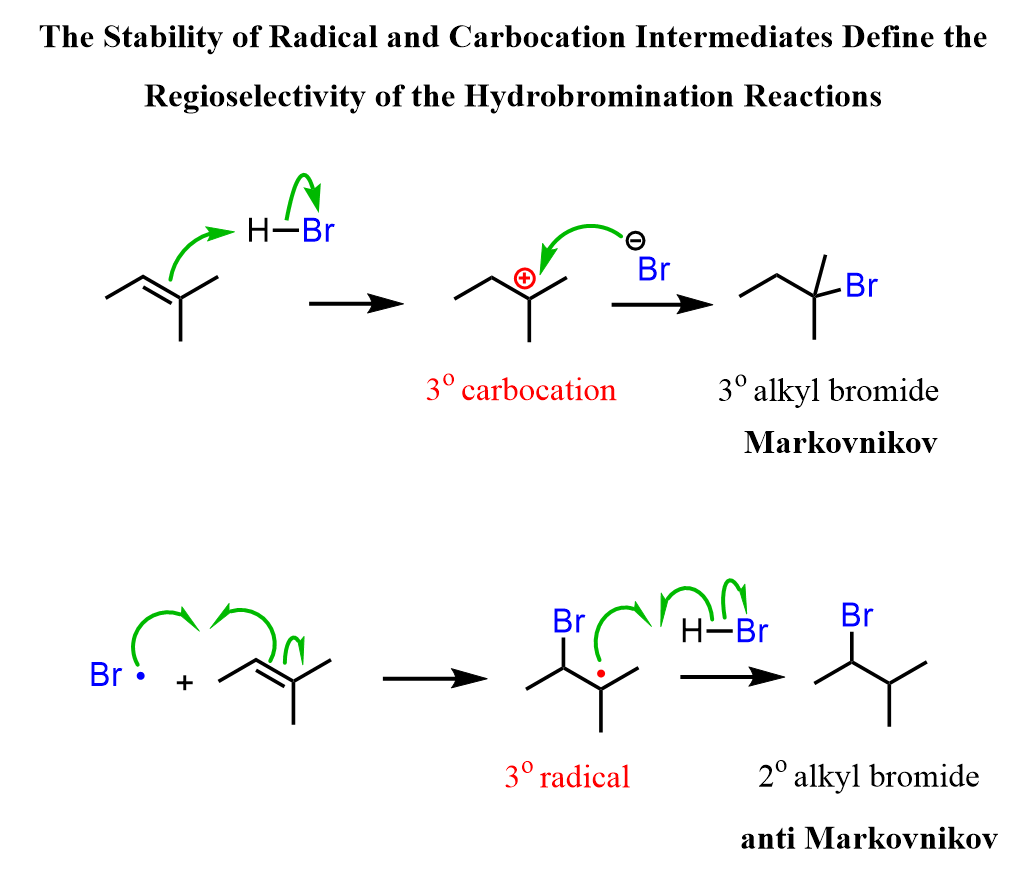
Recall that carbocations and radicals have the same pattern of stability – the more substituted, the more stable.

Keep in mind that this anti-Markovnikov hydrohalogenation is only possible with HBr and not with HCl or HI.
We won’t go too much into the details of the mechanisms in this post as the focus here is to summarize the patterns of regioselectivity in electrophilic additions to alkenes. For detailed discussions, you can click on the corresponding articles for each reaction.
Rearrangements in Hydrohalogenation of Alkenes
Because of the carbocation intermediate, rearrangements can occur during hydrohalogenation reactions. Always check if the carbocation has a way of transforming into a more stable one via hydride or methyl shifts or ring expansion rearrangement. For example, the major product of the following hydrohalogenation is the tertiary alkyl halide instead of the expected secondary product:
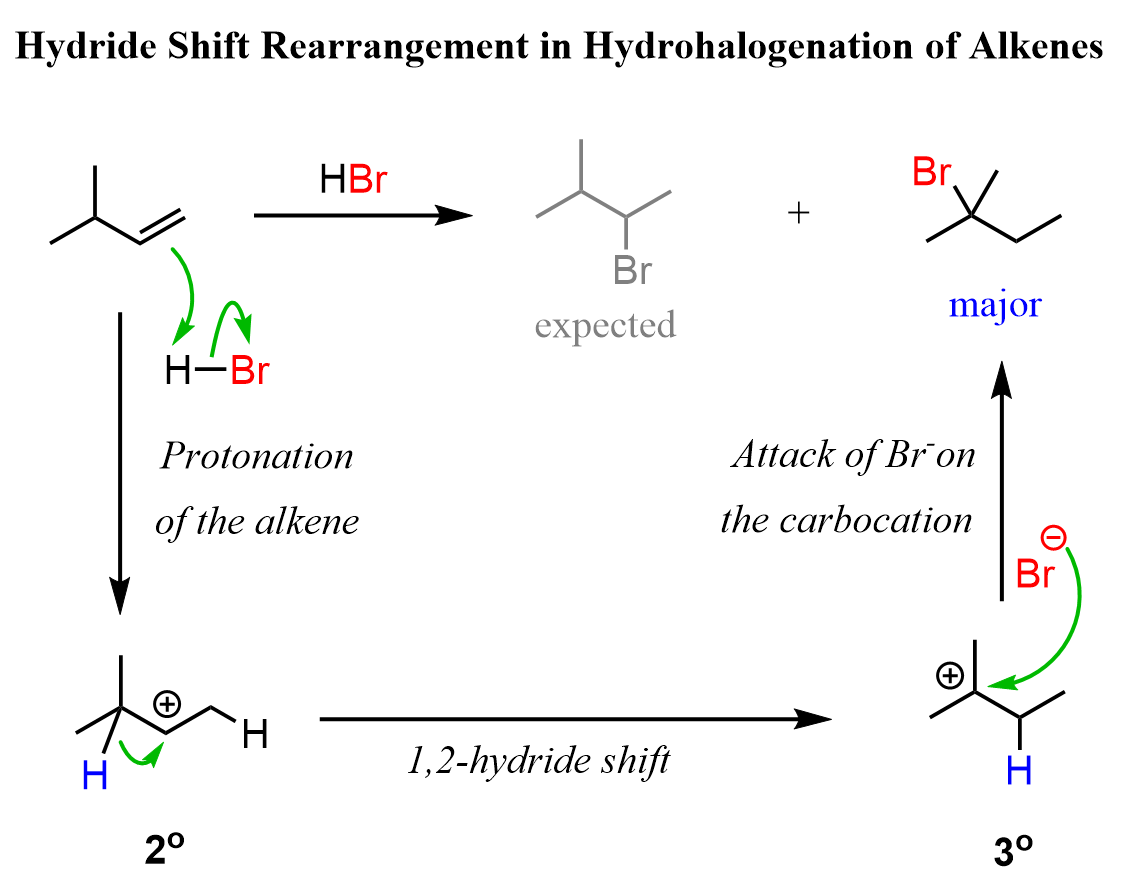
To summarize, the hydrohalogenation of alkenes, we can say that:
1) Regioselectivity is irrelevant for symmetrical alkenes
2) For carbocations, not prone to rearrangements, hydrohalogenation follows Markovnikov’s rule
3) Radical addition of HBr to alkenes gives the anti-Markovnikov product (no rearrangement)
4) When possible, rearrangements do occur
Addition of Water to Alkenes
From the regiochemical perspective, the addition of water to alkenes is seen as adding an OH group to the double bond. There are three main ways of doing it:
1) Acid-Catalyzed Hydration, 2) Oxymercuration-Demercuration, and 3) Hydroboration-oxidation
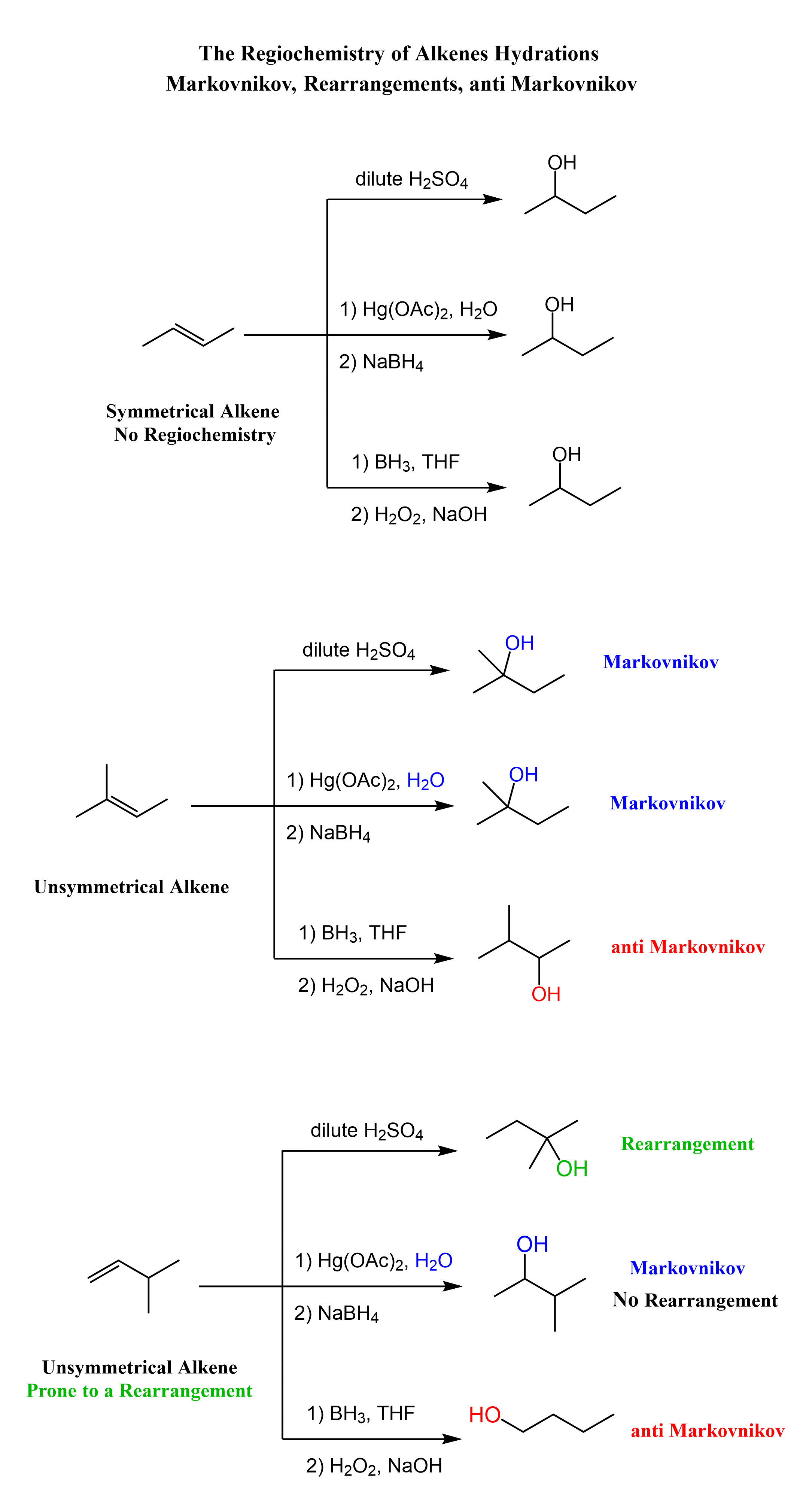
The first two are used for a Markovnikov hydration of the alkene, meaning they add the OH on the more substituted carbon of the double bond, whereas hydroboration oxidation is used for the anti-Markovnikov hydration of the alkene. The oxymercuration-demercuration is used as an alternative to prevent rearrangements in acid-catalyzed hydration:
You can find all the details, including the mechanism and stereochemistry of these reactions, in the corresponding articles linked in this discussion.