Introduction and Nomenclature of Diols
In an earlier article, we discussed the nomenclature of alcohols and mentioned that the presence of a hydroxyl group is identified by changing the parent suffix from “e” to “ol”:
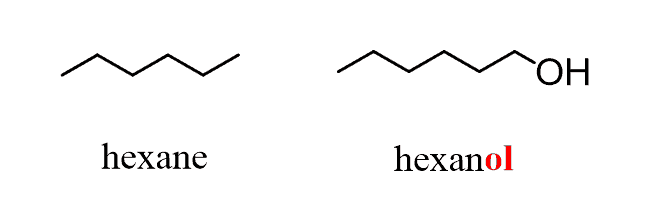
Now, compounds with two hydroxy groups are called diols in the IUPAC nomenclature. So, to name a diol, the suffix -diol is added to the name of the parent chain alkane, and the positions of these hydroxyl groups are indicated with numbers just like for any functional group we have seen before.
For example,

Notice that, unlike regular alcohols, an “e” appears in between the parent and the suffix “diol”.

Simple diols are also commonly named as glycols, which is also accepted by the IUPAC system.

Having two hydroxyl groups brings some changes to the physical properties of diols due to the additional hydrogen bonding. For example, diols are used as antifreeze to cool down the internal combustion engines of automobiles. And in order to qualify for this application, they need to have a significantly lower freezing point and higher boiling point than water. The combination of ethylene glycol (f.p. = -12.9 °C) and propylene glycol (f.p. = -59 °C) is most commonly used in antifreeze solutions.
Preparation of Diols
Diols can be prepared from diketones by reducing the two carbonyl groups using a variety of reducing agents such as NaBH4, LiAlH4, H2/Pd and etc., that we discussed before.

Another common approach for preparing diols is the dihydroxylation of alkenes. Remember, this can be either a syn or anti-dihydroxylation depending on the reagent:
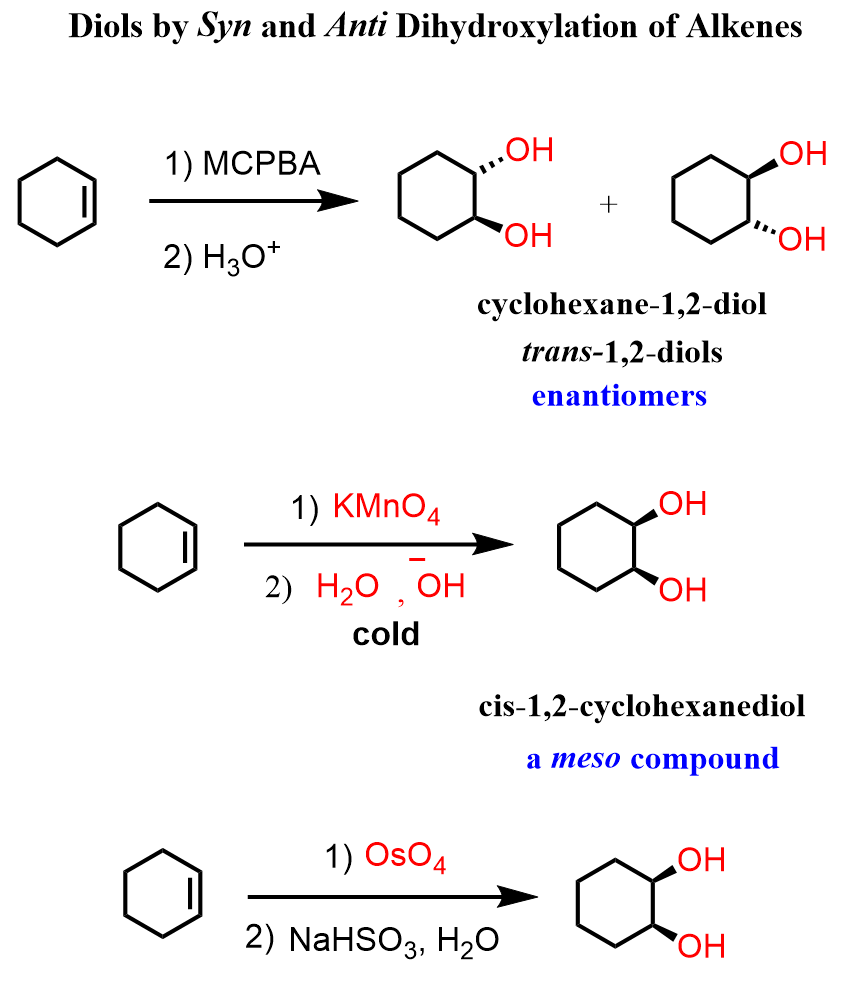
The syn dihydroxylation is done using osmium tetroxide, and 1,2-diols (vic-diols – vicinal diols) are obtained. This can also be achieved with potassium permanganate, although this approach has the risk of cleaving the double bond and over-oxidizing it to a carboxylic acid.
The anti-dihydroxylation of alkenes is carried out with MCPBA and other peroxides.
You can find more about these reactions, including the mechanisms and the stereochemistry, in the corresponding articles linked above.
Reactions of Diols
You can expect to observe all the reactions of alcohols when working with diols. The OH groups can be converted into halides, can react as nucleophiles, and oxidized to different carbonyl-containing functional groups depending on the structure of the diol and the oxidizing agent.
For example, ethylene glycol is used for the synthesis of polyesters, which are widely used in clothing, plastics, furniture, tires, and many other products.
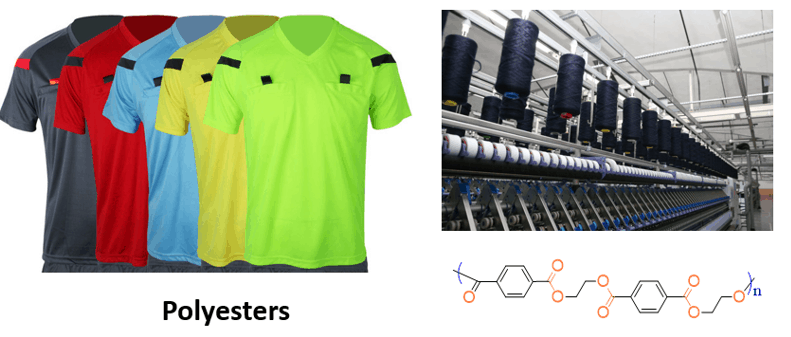
The polyester structure shown in the image is polyethylene terephthalate (PET), which is prepared by a condensation reaction between 1,4-benzenedicarboxylic acid (terephthalic acid) and 1,2-ethanediol:

The ethylene glycol in this reaction acts as a nucleophile, adding to the carbonyl group and converting the acid to an ester. Check this article about the Fischer esterification for more details.
Vicinal diols can be oxidized to aldehydes and ketones depending on the structure of the alcohol with sodium periodate (NaIO4):

So, there are two things happening here: 1) the OH group is oxidized to a carbonyl, and 2) the C-C bond with the oxygens is cleaved. And the pattern is that a primary OH group gives formaldehyde, secondary OH groups produce a ketone, and a tertiary alcohol results in a ketone. The mechanism of this reaction is covered in a separate article, which you can find here.
An important application of 1,2- and 1,3-diols is their use as a protective group for aldehydes and ketones. When aldehydes and ketones are reacted with an excess alcohol, an acetal is formed where two alkoxy (OR) groups are connected to the same carbon atom:
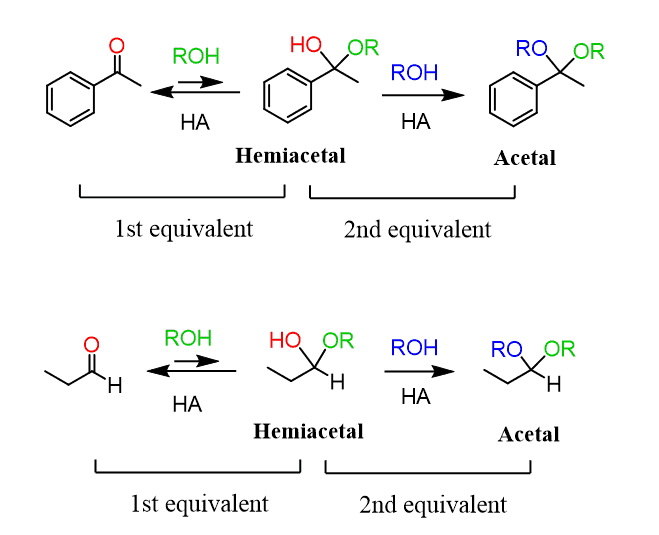
These two equivalents of the OH group can also be incorporated by a diol, producing a cyclic acetal, which is especially favored when five or six-membered rings are formed:

Cyclic acetals are very stable under basic conditions but can be removed when treated with acids. Therefore, cyclic acetals are used as protecting groups for aldehydes and ketones:
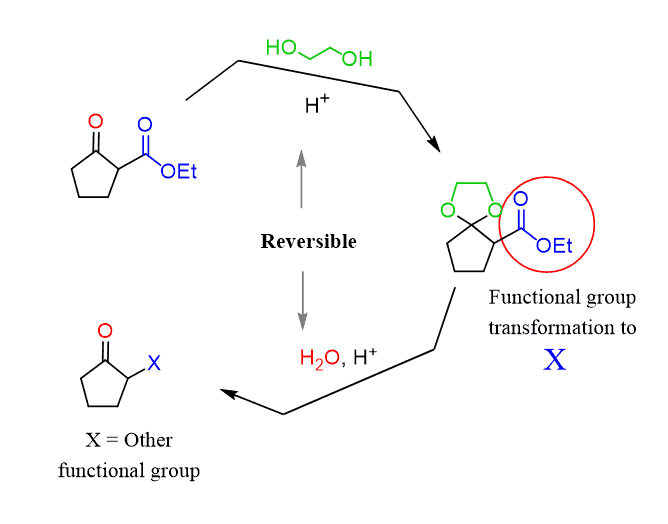
In the reaction above, ethylene glycol was used to convert the ketone into an acetal and protect it while reducing the ester group.
An important reaction of some 1,2-diols (vicinal diols) is what is called the pinacol rearrangement. Here, the diol is converted into a ketone when treated with an acid.

The mechanism and some other details of the pinacol rearrangement are covered in a separate post, which you can find here.
Geminal Diols
In all the examples above, we had 1,2-diols, which can also be referred to as vicinal diols. When the two OH groups are connected to the same carbon atom, we now have geminal diols (gem-diols). These are formed when an aldehyde is dissolved in water. The system establishes an equilibrium between the aldehyde and the geminal diol, called a hydrate of the aldehyde.

The addition of water to most aldehydes and ketones is unfavorable, and the equilibrium is shifted mainly toward the carbonyl compound.
A few exceptions are the aldehydes with nearby electron-withdrawing groups and the simplest aldehyde, formaldehyde, which exists primarily as the gem-diol in aqueous solution:

Because of this, it is not possible to isolate most gem-diols from the aqueous solutions in which they are formed. If we try to evaporate the water, for example, the equilibrium simply shifts to simply shifts toward the carbonyl compound as expected by Le Chatelier’s principle:

Check this 66-question, Multiple-Choice Quiz with a 2-hour Video Solution on the naming, preparation, and reactions of Alcohols.
Alcohols Quiz – Naming, Preparation, and Reactions
Check Also
- Nomenclature of Alcohols: Naming Alcohols based on IUPAC Rules with Practice Problems
- Preparation of Alcohols via Substitution or Addition Reactions
- Reaction of Alcohols with HCl, HBr, and HI Acids
- Mesylates and Tosylates as Good Leaving Groups
- SOCl2 and PBr3 for Conversion of Alcohols to Alkyl Halides
- Alcohols in Substitution Reactions Practice Problems
- POCl3 for Dehydration of Alcohols
- Dehydration of Alcohols by E1 and E2 Elimination
- The Oxidation States of Organic Compounds
- LiAlH4 and NaBH4 Carbonyl Reduction Mechanism
- Alcohols from Carbonyl Reductions – Practice Problems
- Grignard Reaction in Preparing Alcohols with Practice Problems
- Grignard Reaction in Organic Synthesis with Practice Problems
- Protecting Groups For Alcohols in Organic Synthesis
- Oxidation of Alcohols: PCC, PDC, CrO3, DMP, Swern, and All of That
- NaIO4 Oxidative Cleavage of Diols
- The Pinacol Rearrangement
- The Williamson Ether Synthesis
- Alcohol Reactions Practice Problems
- Naming Thiols and Sulfides
- Reactions of Thiols
- Alcohols Quiz – Naming, Preparation, and Reactions
- Reactions Map of Alcohols


This was something of value,,,the notes are organised and truly meant a lot and saved me from failure,,, thanks
Good to hear this, Sylvia!