Markovnikov’s Rule
In the previous post, we talked about the addition reaction of hydrogen halides (HCl, HBr, and HI) to alkenes, and learned about Markovnikov’s rule, which states that in these reactions, the halogen adds to the more substituted carbon of the double bond. For example, these are the 3rd and 4th reactions we discussed, where you can see how the iodine and bromine end up on the secondary and tertiary positions:
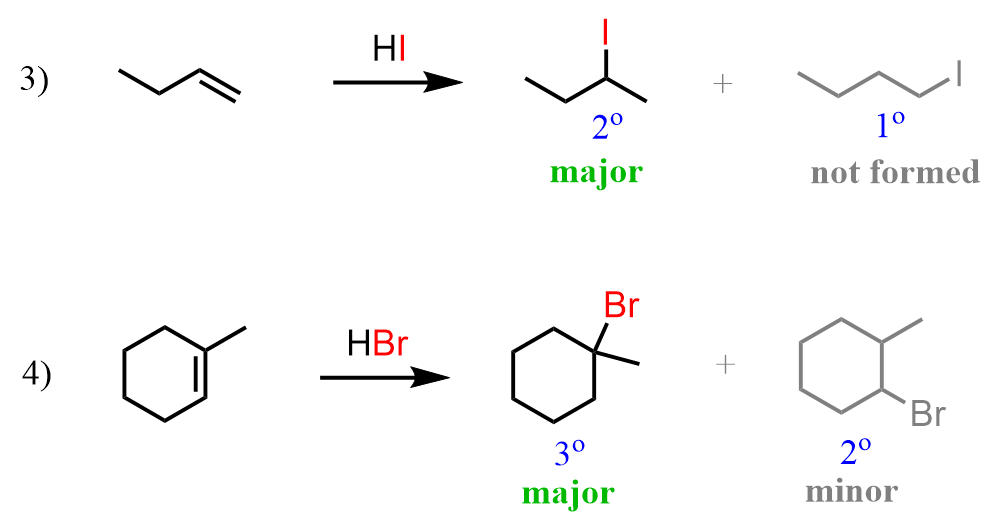
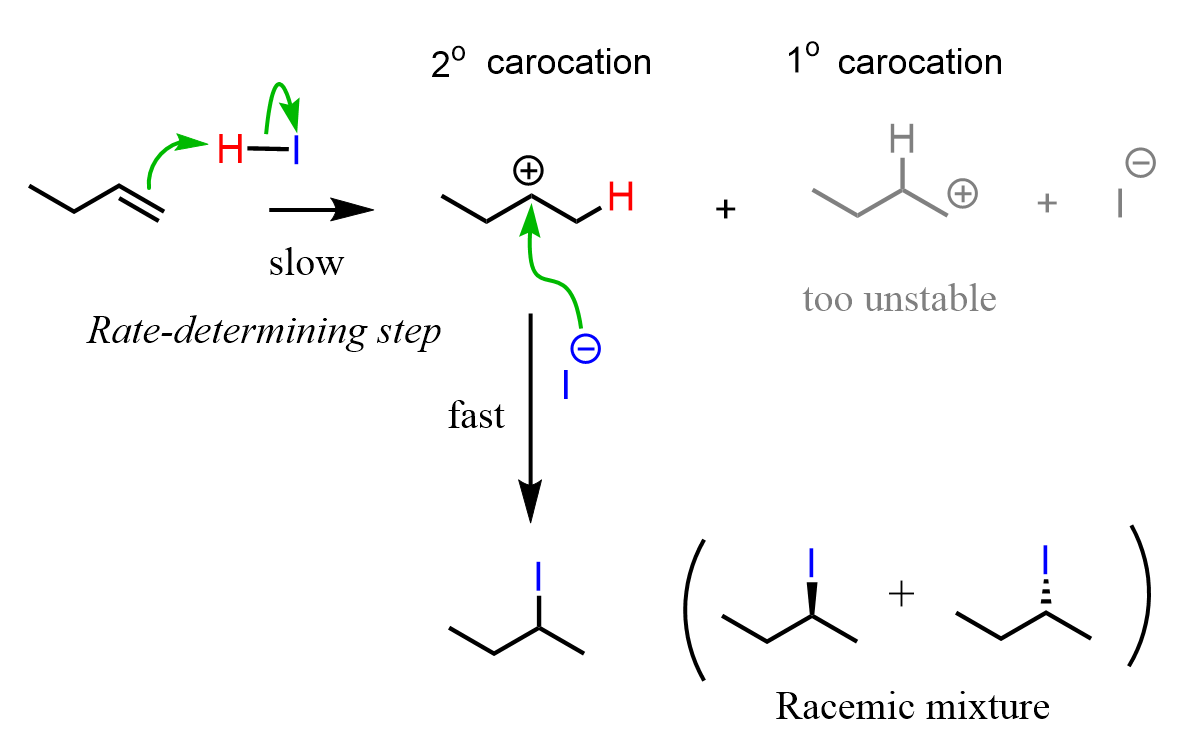
This is the regioselectivity of the hydrohalogenation reaction of alkenes, and it is explained by the fact that the reaction goes via a carbocation intermediate. The stability of carbocations increases with the number of alkyl groups, and therefore, the more substituted carbocation is formed, which leads to the more substituted alkyl halide:
Hydride Shifts in Alkene Addition Reactions
The formation of carbocation intermediates brings the possibility of rearrangements during hydrohalogenation of alkenes, just like we have seen in SN1 and E1 reactions.
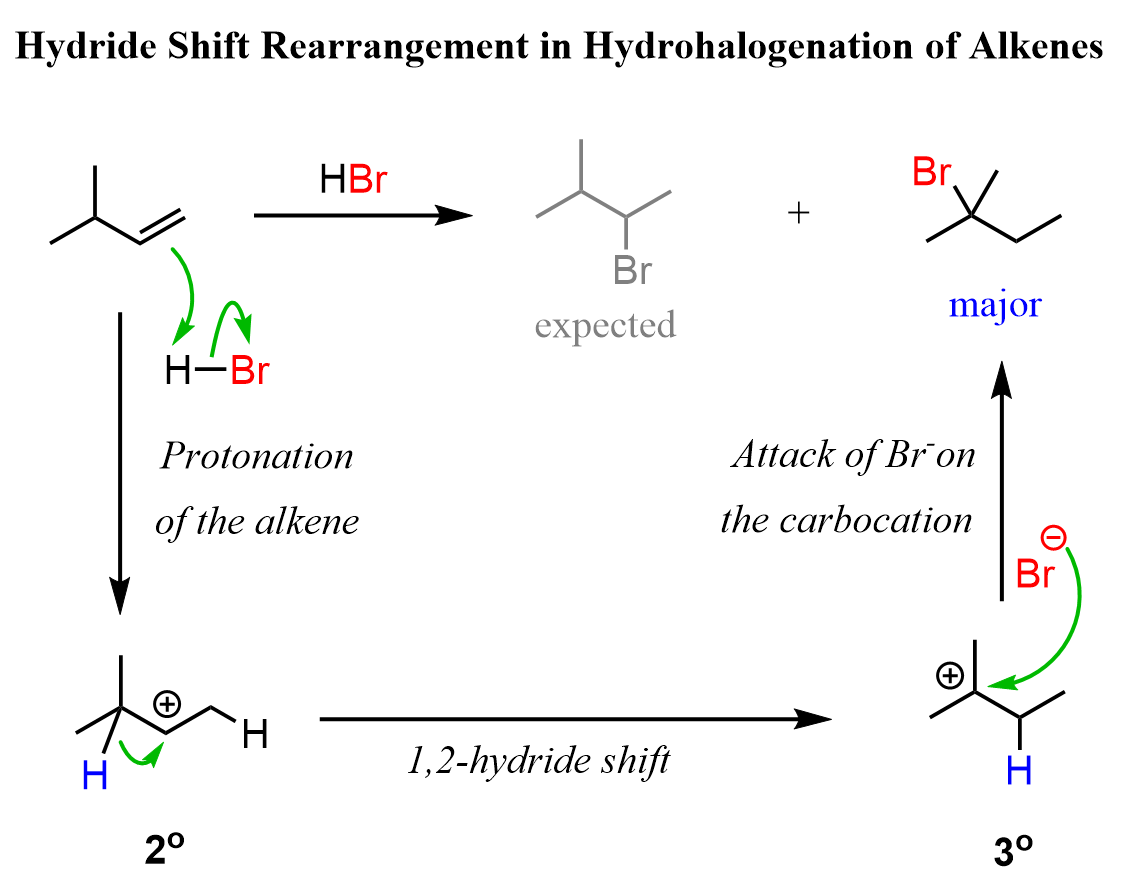
For example, when the following alkene is treated with HBr, based on Markovnikov’s rule, we may expect the Br to add to the middle carbon because of the greater stability of the secondary carbocation:
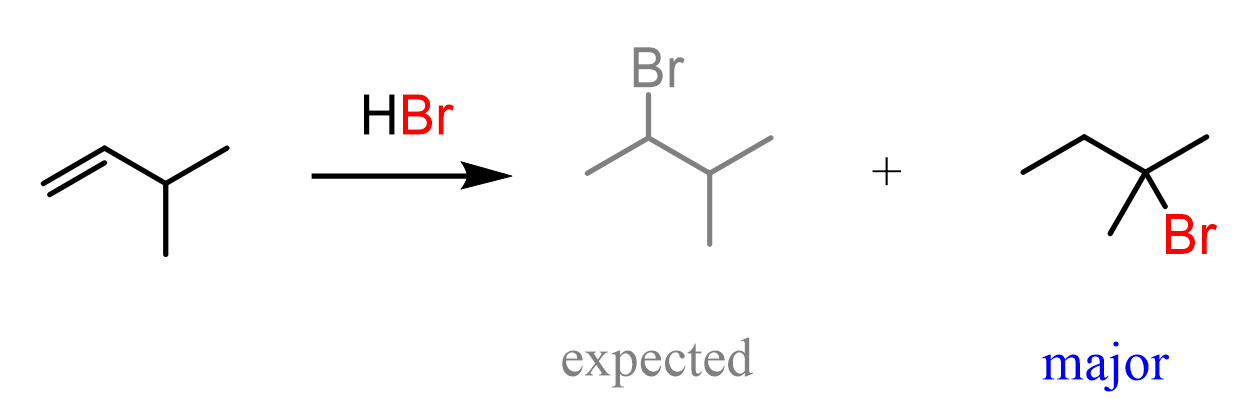
So, how does this happen? In the first step, we know that the hydrogen addition will form a secondary carbocation because it is more stable than the possible primary carbocation. This is true; upon protonation, the secondary carbocation is formed; however, before reacting with the bromide ion, it undergoes a 1,2-hydride shift rearrangement to form a more stable tertiary carbocation:
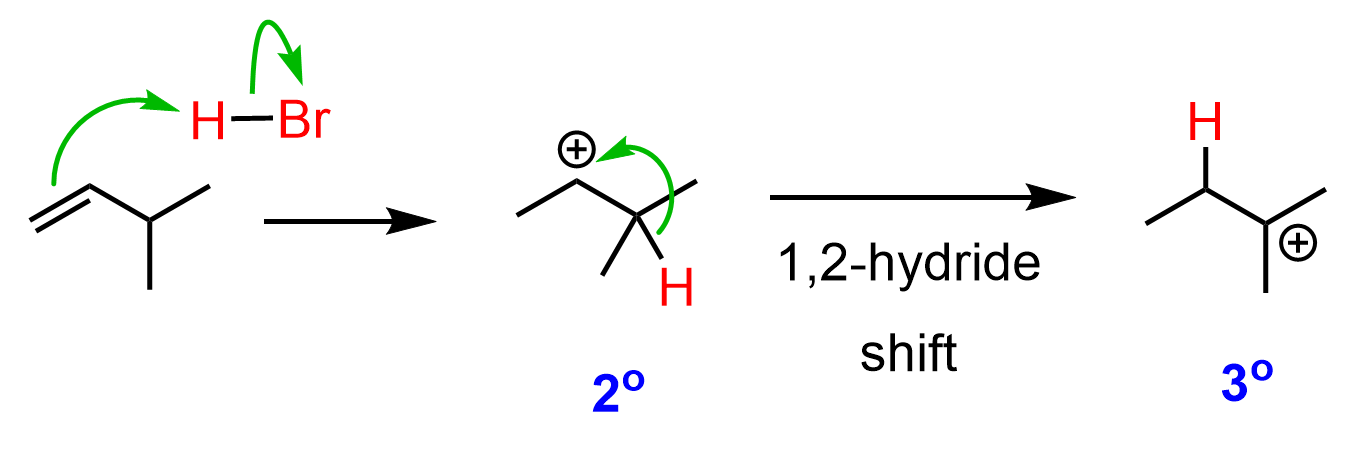
Once the tertiary carbocation is formed, it is attacked by the bromide, resulting in a tertiary alkyl halide as the major product of the reaction:
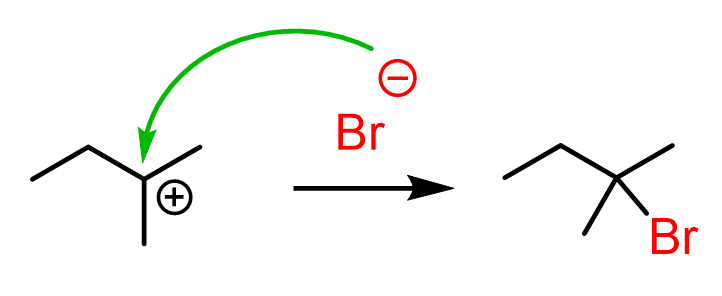
Let’s combine the steps in one scheme to represent the mechanism of hydride shift rearrangement in the hydrohalogenation of alkenes:
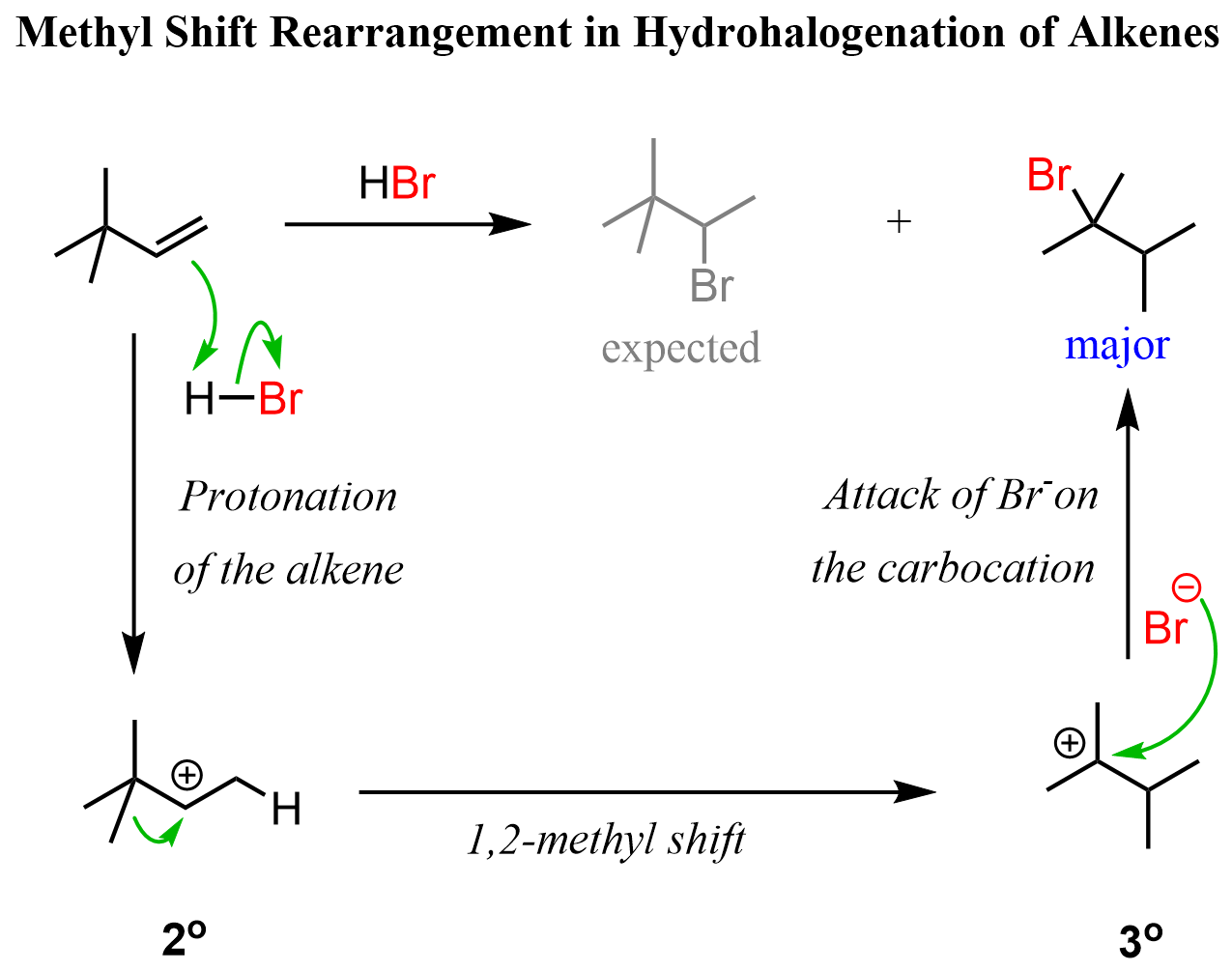
Methyl Shifts in Alkene Addition Reactions
Rearrangements during hydrohalogenation of alkenes, and other reactions going via carbocation intermediates, can also happen via 1,2-methyl (more accurately methide) shifts. For example, instead of the hydrogen, there was a methyl group on the alkene we discussed above, a 1,2-methyl shift would occur to transform the secondary carbocation to a more stable tertiary carbocation:
Ring Expansion Rearrangements in Alkene Additions
Alkyl shifts forming a more stable carbocation intermediate may also lead to ring expansion rearrangements. This is especially common for cycloalkanes with high ring strain, such as cyclobutane. For example, we can view the following alkene as bearing a cyclobutyl substituent:
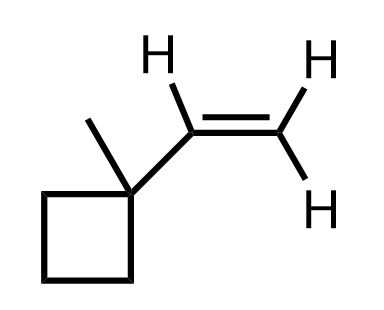
When the alkene is protonated, a secondary carbocation is formed, which can be converted into a more stable tertiary carbocation via a ring-expansion rearrangement:
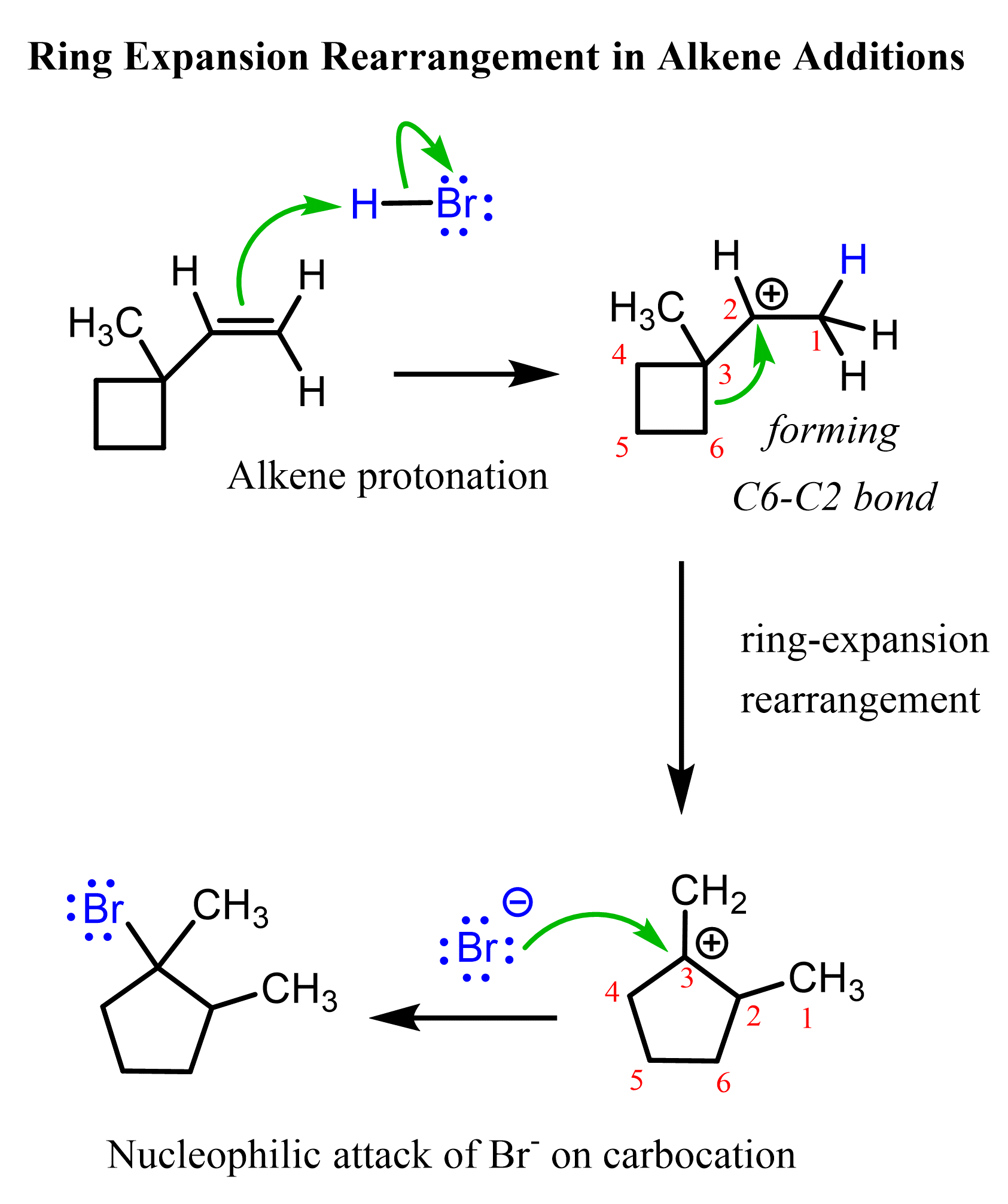
Check this article for more examples and practice problems on ring expansion rearrangements of alkyl halides, alcohols, and alkenes in substitution and elimination reactions.
Rearrangements in Alcohol Addition to Alkenes
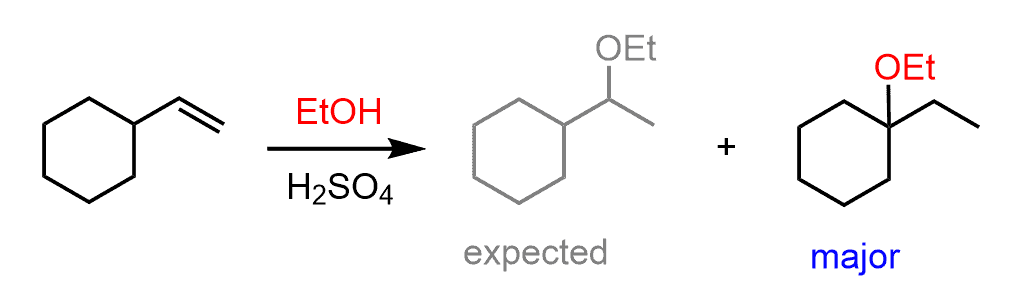
Although all the examples we discussed so far were related to the addition of HX acids to alkenes, you need to keep in mind the possibility of rearrangements in other addition reactions, such as the acid-catalyzed hydration or the addition of alcohols to alkenes.
For example, the major product of the following alcohol addition reaction is not the ether with the alkoxy group on the more substituted carbon of the double bond based on Markovnikov’s rule:
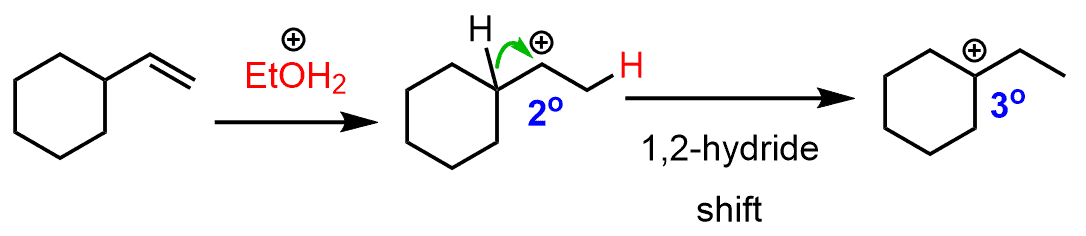
The secondary carbocation that is formed after the protonation of the double bond undergoes a hydride shift rearrangement to form the more stable tertiary carbocation:
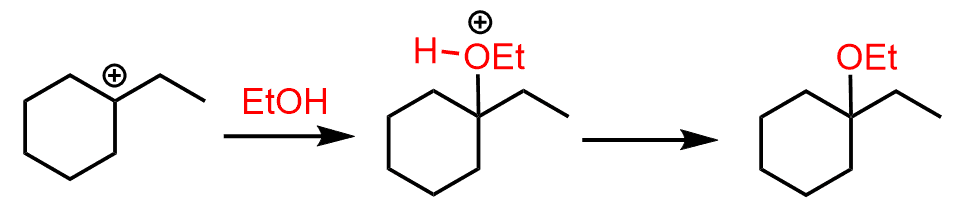
The nucleophilic attack of the ethanol results in an ether with a tertiary alkyl group:
Rearrangements in Acid-Catalyzed Hydration of Alkenes
Just like in hydrohalogenation reactions, the hydration of alkenes can also proceed through carbocation intermediates, which means rearrangements are possible. In acid-catalyzed hydration, the alkene first gets protonated to form a carbocation. If a more stable carbocation can form through a hydride or alkyl shift, the molecule will rearrange before the water attacks. This leads to unexpected products if you’re not watching for rearrangements.
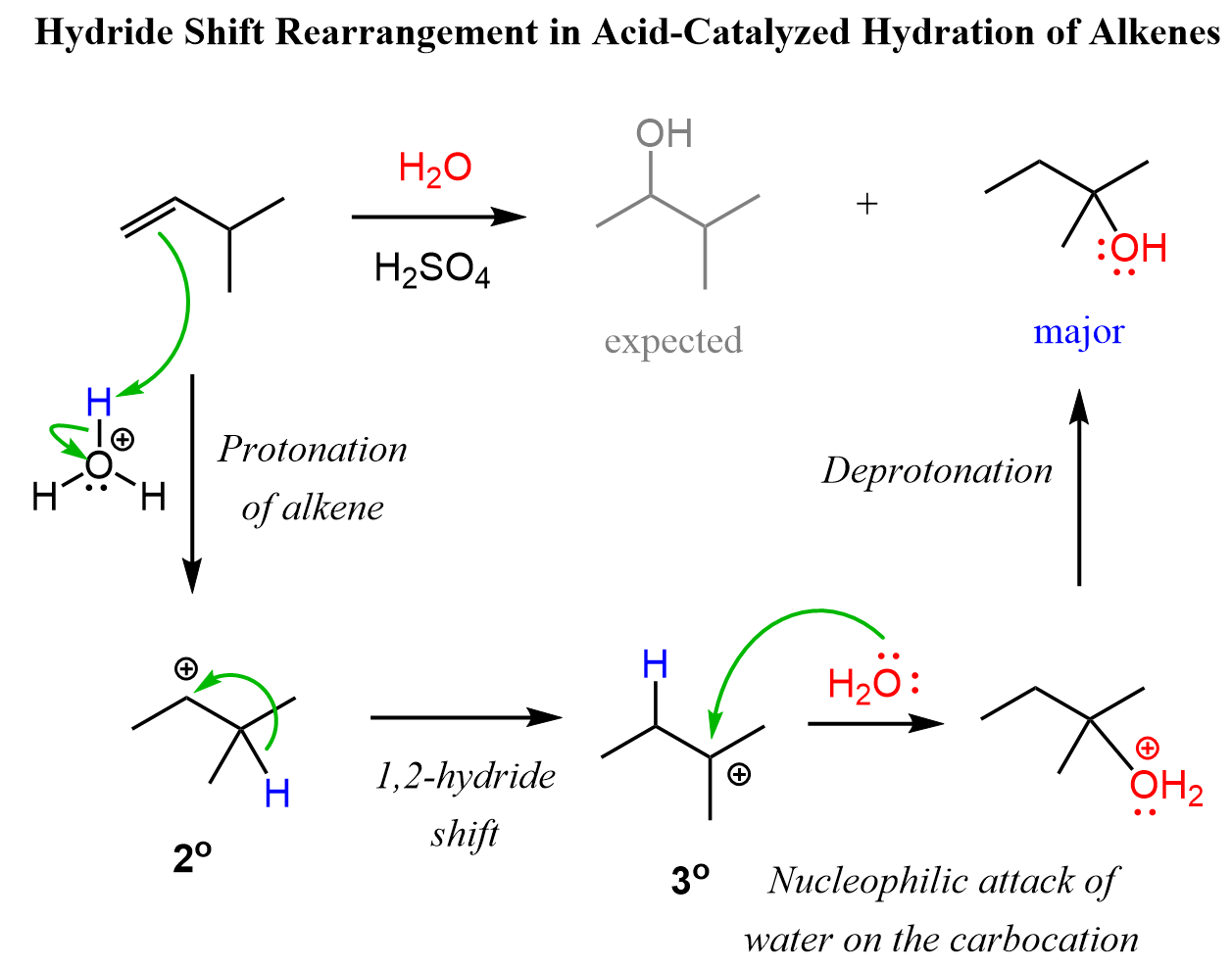
So, whenever hydration involves a carbocation—especially with unsymmetrical alkenes—don’t forget to check if a carbocation rearrangement could change the regiochemistry of the final alcohol!
Can We Avoid Rearrangements in Addition Reactions of Alkenes?
To avoid these rearrangements, chemists often use oxymercuration-demercuration instead. This method adds water across the double bond with Markovnikov regioselectivity, but crucially, it does not form a free carbocation. Instead, the reaction goes through a cyclic mercurinium ion intermediate, which blocks rearrangement and leads to a more predictable product. So when you’re hydrating an alkene where rearrangement is a concern, oxymercuration-demercuration is the preferred method for clean, controlled hydration.

The key difference, compared to acid-catalyzed hydration, is that the intermediate in the oxymercuration-demercuration reaction is the mercurinium ion, which does not undergo a rearrangement. What is also important is that the water attacks the more substituted carbon of the mercurinium ion, thus the corresponding alcohol is formed (Markovnikov addition). Check the post about oxymercuration-demercuration for more details.
As for the hydrohalogenation, which converts alkenes to alkyl halides, we can still use the oxymercuratio-demercuration reaction, and convert the resulting alcohol to the corresponding alkyl halide using mesylation followed by a reaction with sodium halide or the classical SOCl2 (thionyl chloride) or PBr3 reagents:
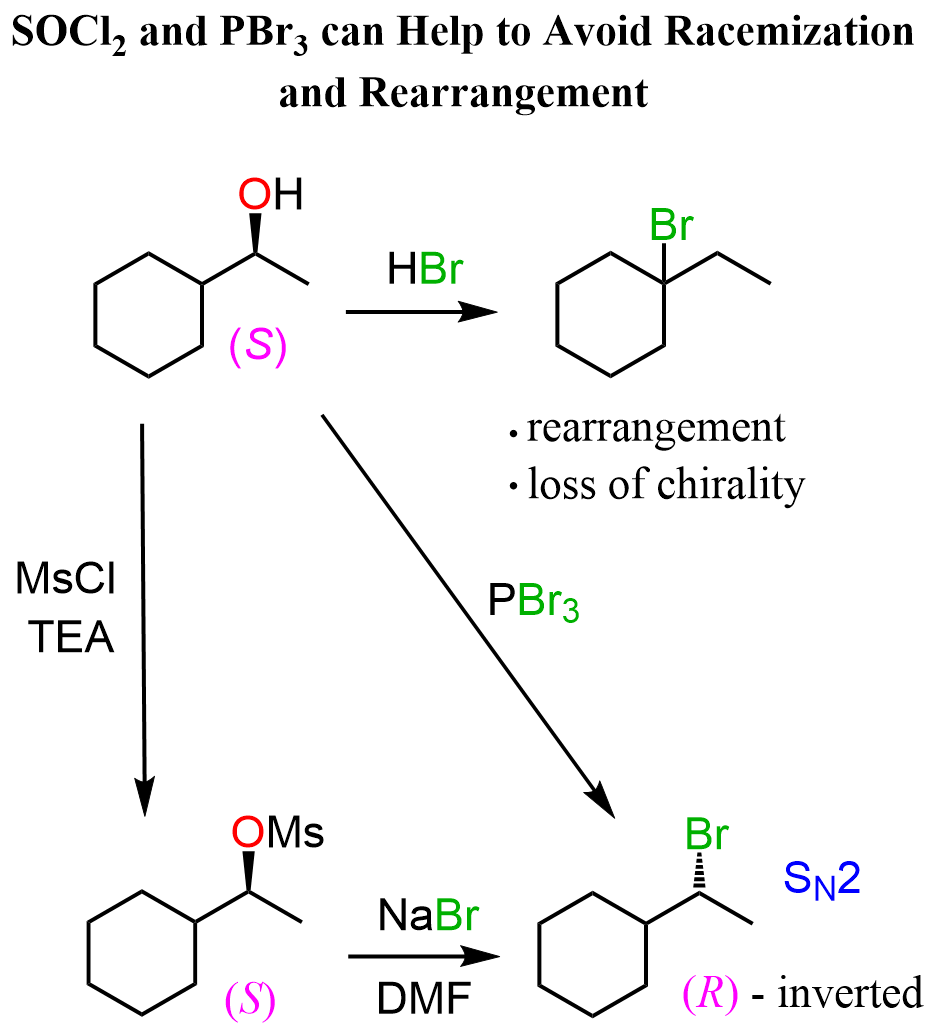
To summarize this section, remember that, when possible, rearrangements are going to happen. Therefore, you need to keep them in mind when looking at any reaction involving a carbocation intermediate. In a typical undergraduate organic chemistry course, these are going to be SN1, E1, and the electrophilic addition reactions to alkenes, such as those of hydrohalic acids (hydrogen halides), and acid-catalyzed hydration. One strategy for avoiding rearrangements in addition reactions to unsymmetrical alkenes is the use of the oxymercuration-demercuration reaction.
Check Also
- Electrophilic Addition Reactions to Alkenes
- Markovnikov’s Rule
- Markovnikov’s Rule with Practice Problems
- Addition of Water to Alkenes
- Acid-Catalyzed Hydration of Alkenes with Practice Problems
- Oxymercuration-Demercuration
- Addition of Alcohols to Alkenes
- Free-Radical Addition of HBr: Anti-Markovnikov Addition
- Hydroboration-Oxidation: The Mechanism
- Hydroboration-Oxidation of Alkenes: Regiochemistry and Stereochemistry with Practice Problems
- Halogenation of Alkenes and Halohydrin Formation
- The Regiochemistry of Alkene Addition Reactions
- The Stereochemistry of Alkene Addition Reactions
- Cis product in an anti-Addition Reaction of Alkenes
- Ozonolysis of Alkenes with Practice Problems
- Syn Dihydroxylation of Alkenes with KMnO4 and OsO4
- Anti-Dihydroxylation of Alkenes with MCPBA and Other Peroxides with Practice Problems
- Oxidative Cleavage of Alkenes with KMno4 and O3
- Alkene Reactions Practice Problems
- Changing the Position of a Double Bond
- Changing the Position of a Leaving Group
- Alkenes Multi-Step Synthesis Practice Problems
- Alkene Addition Reactions Practice Quiz
- Reactions Map of Alkenes
