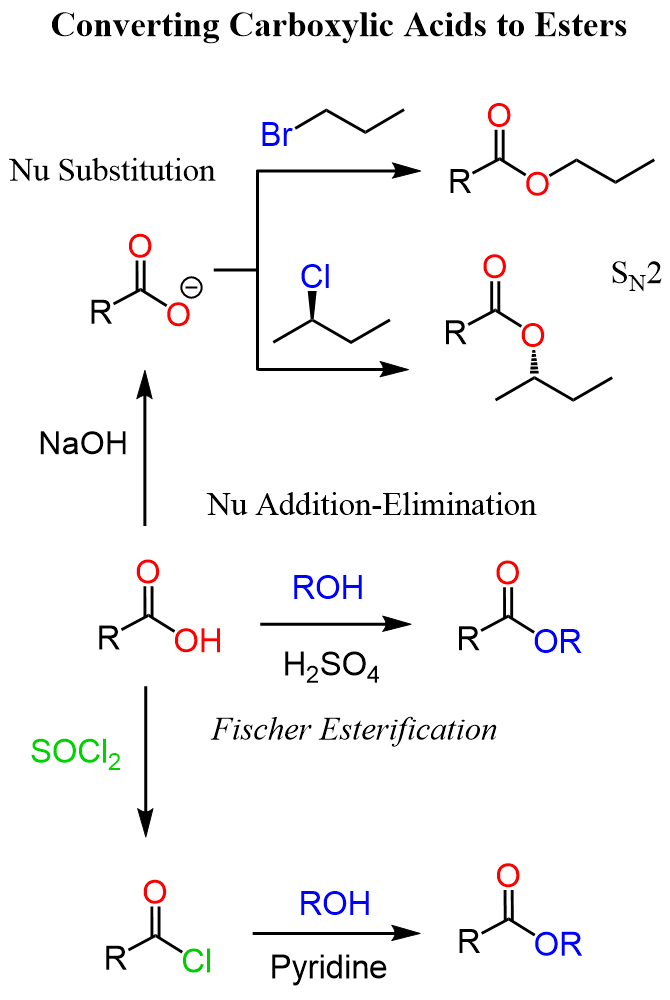
There are a few ways of converting carboxylic acids to esters, and the first one is the direct reaction between the acid and an alcohol. This is known as the Fischer esterification which is a type of addition-elimination reaction of carbonyl:

The Fischer esterification has some limitations due to the reaction conditions. It is a reaction carried out in acidic media with elevated temperatures, and not every molecule can survive these conditions.
Besides surviving, there are wanted reactions that can take place in the presence of an acid catalyst. For example, we know that alcohols undergo dehydration reactions in the presence of concentrated sulfuric acids. Now, while we do not use concentrated sulfuric acid here, the use of tertiary alcohols can certainly be problematic because they will still undergo some dehydration.

In addition, tertiary alcohols are even weaker nucleophiles than primary and secondary alcohols due to the steric hindrance of the oxygen. Aryl alcohol, phenols are also weaker nucleophiles not because of the steric hindrance but because of the resonance delocalization of the lone pair of the oxygen. Therefore, a good alternative to Fischer esterification is the use of acid chlorides which can be prepared from carboxylic acids by chlorinating agents such as SOCl2, PCl3, and PCl5.

The reaction is carried out under basic conditions and this may be more suitable for certain functional groups. This method is applicable to aromatic aliphatic alcohols:

Esters via Nucleophilic Substitution
Another strategy to convert the carboxylic acid to an ester is the use of the nucleophilicity of the carboxylate ion. Carboxylic acids are very poor nucleophiles, however, if we deprotonate them, the resulting carboxylate ions are quite nucleophilic and we can use them for SN2 substitution with alkyl halides:

In the end, let’s summarize the strategies for converting alcohols to esters in one synthetic scheme:
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Preparation of Carboxylic Acids
- Naming Carboxylic Acids
- Naming Nitriles
- Naming Esters
- Naming Carboxylic Acid Derivatives – Practice Problems
- Fischer Esterification
- Ester Hydrolysis by Acid and Base-Catalyzed Hydrolysis
- What is Transesterification?
- Esters Reaction with Amines – The Aminolysis Mechanism
- Ester Reactions Summary and Practice Problems
- Preparation of Acyl (Acid) Chlorides (ROCl)
- Reactions of Acid Chlorides (ROCl) with Nucleophiles
- Reaction of Acyl Chlorides with Grignard and Gilman (Organocuprate) Reagents
- Reduction of Acyl Chlorides by LiAlH4, NaBH4, and LiAl(OtBu)3H
- Preparation and Reaction Mechanism of Carboxylic Anhydrides
- Amides – Structure and Reactivity
- Naming Amides
- Amides Hydrolysis: Acid and Base-Catalyzed Mechanism
- Amide Dehydration Mechanism by SOCl2, POCl3, and P2O5
- Amide Reduction Mechanism by LiAlH4
- Amides Preparation and Reactions Summary
- Amides from Carboxylic Acids-DCC and EDC Coupling
- The Mechanism of Nitrile Hydrolysis To Carboxylic Acid
- Nitrile Reduction Mechanism with LiAlH4 and DIBAL to Amine or Aldehyde
- The Mechanism of Grignard and Organolithium Reactions with Nitriles
- Carboxylic Acids to Ketones
- Esters to Ketones
- Carboxylic Acids and Their Derivatives Practice Problems
- Carboxylic Acids and Their Derivatives Quiz