Aldehydes and ketones have similar structures and chemical properties. The only difference is instead of the hydrogen we have another alkyl group on the carbonyl. So, how can we replace this hydrogen with an alkyl group?

Aldehydes to Ketones Using Organometallics
The most common way you will see is the reaction of the aldehyde with a Grignard or organolithium reagent followed by an oxidation to the ketone:

Any oxidizing agent can be used to oxidize secondary alcohols to ketones since ketones cannot be overoxidized with them.

Aldehydes to Ketones via Cyclic Thioacetals
Another way of alkylating aldehydes is their conversion to cyclic thioacetals followed by a substitution reaction with an alkyl halide:
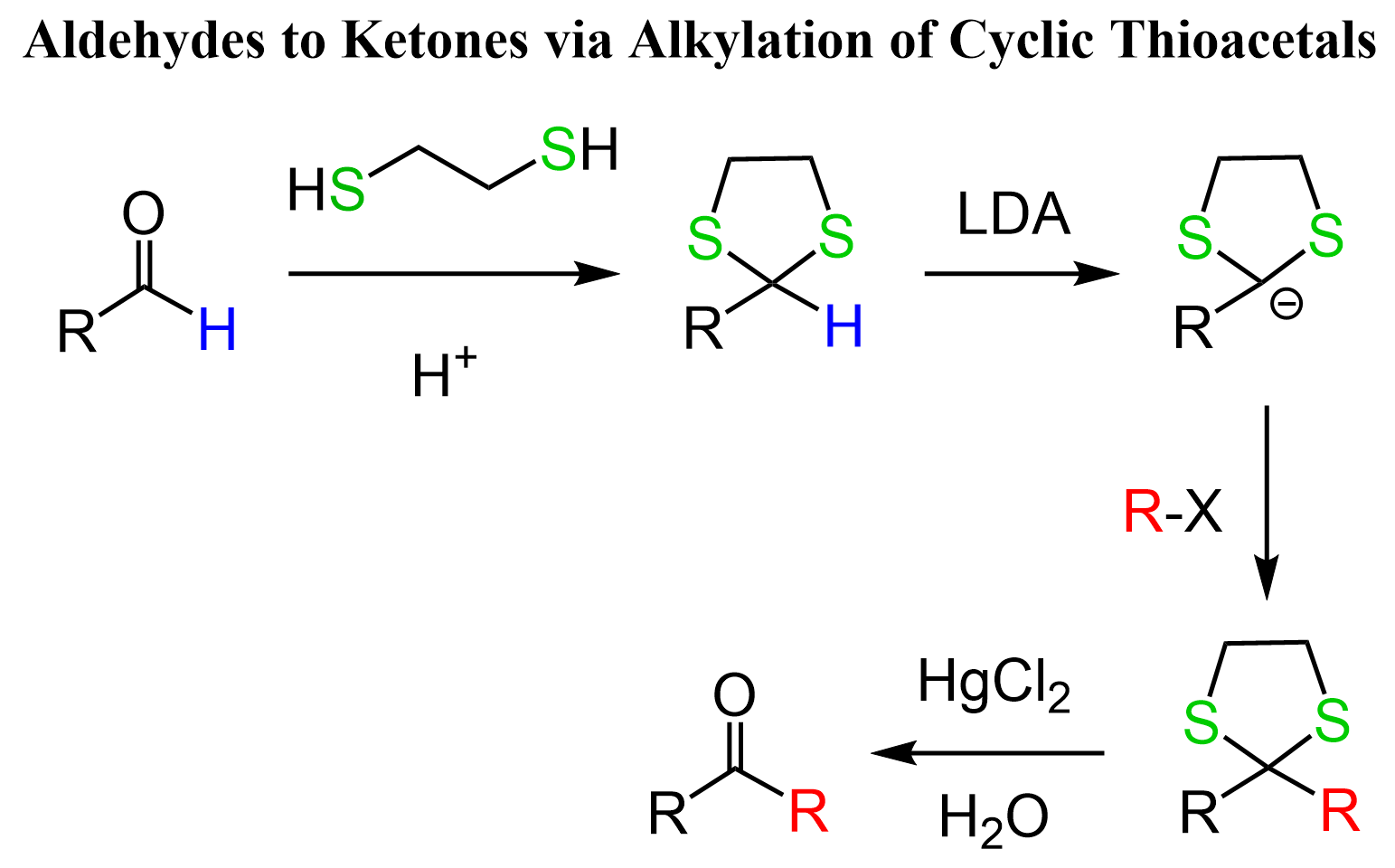
The first step is similar to what we learned about the protection of aldehydes and ketones by converting them to cyclic acetals. Now, cyclic thioacetals are a useful way of alkylating aldehydes since they decrease the pKa of the carbonyl protons from ~45 to ~32 and we can deprotonate it with strong bases such as LDA or organolithiums. The thioacetal is then hydrolyzed to a ketone using mercury salt catalysis.
Aldehydes to Ketones Without Adding an Alkyl Group
In both strategies we discussed above the aldehyde was converted to a ketone by alkylation which means the carbon chain of the molecule was lengthened. So, let’s say we need to convert the aldehyde to a ketone without adding extra carbons to the molecule:

We can reduce the aldehyde to alcohol, dehydrate the alcohol to an alkene, followed by a Markovnikov hydration, and oxidize the resulting secondary alcohol to the corresponding ketone:

This is what comes to mind for converting aldehydes to ketones. Let me know in the comments if there are other methods that be suitable for undergraduate organic chemistry coursework.
In the next article, we’ll discuss the reverse process, that is the conversion of ketones to aldehydes.
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Nomenclature of Aldehydes and Ketones
- How to Name a Compound with Multiple Functional Groups
- Preparation of Aldehydes and Ketones
- Nucleophilic Addition to Carbonyl Groups
- The Addition-Elimination Mechanism
- Reduction of Carbonyl Compounds by Hydride Ion
- Reactions of Aldehydes and Ketones with Water
- Reactions of Aldehydes and Ketones with Alcohols: Acetals and Hemiacetals
- Acetals as Protecting Groups for Aldehydes and Ketones
- Imines from Aldehydes and Ketones with Primary Amines
- Enamines from Aldehydes and Ketones with Secondary Amines
- Reactions of Aldehydes and Ketones with Amines-Practice Problems
- Acetal Hydrolysis Mechanism
- Imine and Enamine Hydrolysis Mechanism
- Reaction of Aldehydes and Ketones with CN Cyanohydrin Formation
- Hydrolysis of Acetals, Imines and Enamines-Practice Problems
- The Wittig Reaction: Examples and Mechanism
- The Wittig Reaction-Practice Problems
- Aldehydes and Ketones Reactions Practice Quiz