Baeyer-Villiger Oxidation
The Baeyer-Villiger oxidation is a rearrangement reaction that converts ketones to esters using peroxycarboxylic acids, such as meta-chloroperoxybenzoic acid (mCPBA).
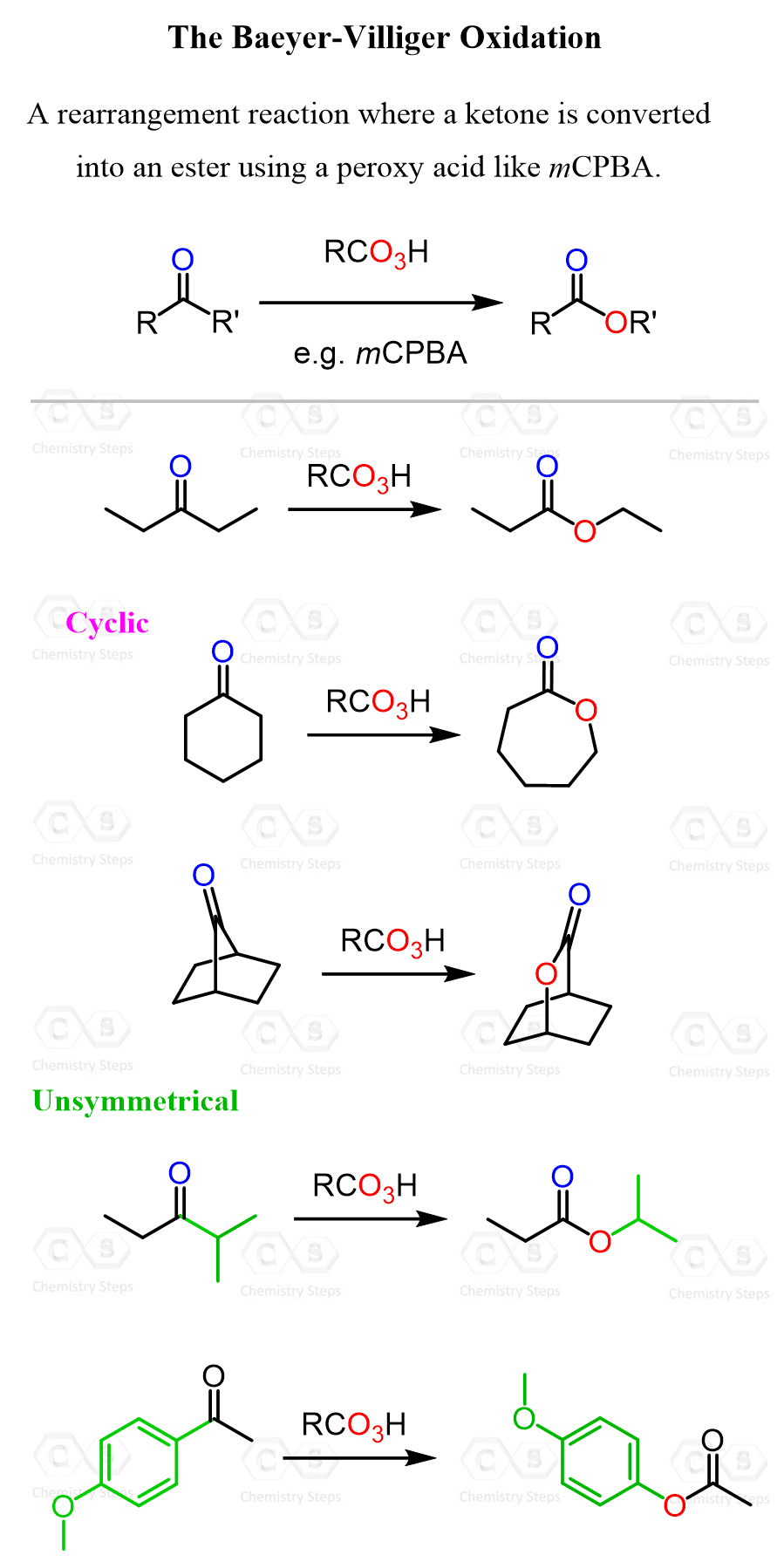
The Baeyer–Villiger oxidation can also convert aldehydes into carboxylic acids, but let’s be honest, including that as a key use feels like low-key cheating. After all, even though the mechanism differs, we all know there are plenty of simpler ways to oxidize an aldehyde to a carboxylic acid, right?
The Mechanism of Baeyer-Villiger Oxidations
Going from a ketone to an ester is an oxidation process, but we mentioned that the mechanism of Baeyer-Villiger is different from what we saw in conventional reactions using PCC, KMnO4, etc. It is a rearrangement reaction where a tetrahedral peroxy intermediate undergoes a familiar 1,2-shift, thus forming the R-O alkoxy component of the ester.
The first step of the reaction is the activation of the carbonyl group via protonation by the acid, followed by a nucleophilic attack by the peroxycarboxylate ion, which leads to the aforementioned intermediate capable of a rearrangement via 1,2-alkyl shift (migration):

After the migration step, a protonated ester is formed, and it simply loses the proton to give the final product.
The Regiochemistry of Baeyer-Villiger Oxidation
In the mechanism above, we have labeled the carbon chains as R and R’, so how do we know which of these is going to migrate and make the new C-O bond? This is defined by the migratory aptitude of the alkyl groups connected to the carbonyl. It has been found that tertiary alkyl groups migrate the fastest, followed by secondary, and then aryl groups. In case we are doing a Baeyer-Villiger on an aldehyde, keep in mind that hydrogen would go before the tertiary alkyl group: H > tertiary > secondary > aryl > primary > methyl.
For example, we can use the Baeyer-Villiger Oxidations to convert ethyl isopropyl ketone (2-methyl-pentan-3-one) to isopropyl propionate:
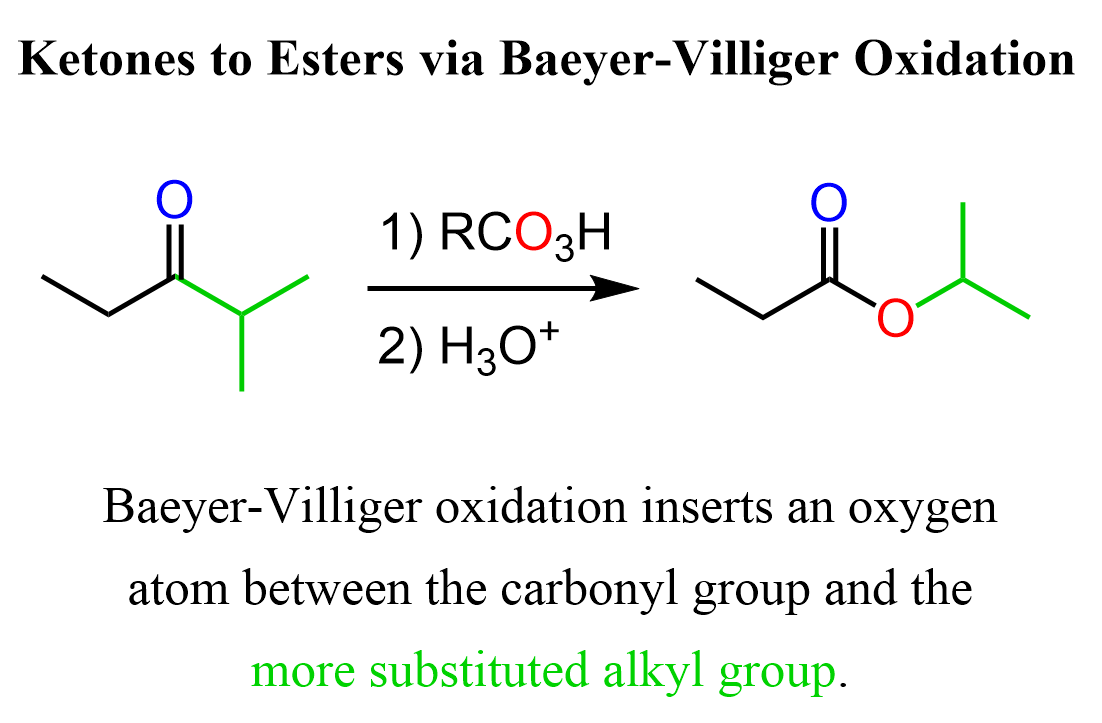
So, for the general pattern of the Baeyer-Villiger Oxidation, remember that it inserts an oxygen atom between the carbonyl group and the more substituted alkyl group.
This is related to both electron density and steric considerations, and it is a subject of ongoing discussions that are beyond the scope of this article. For aryl groups, the migratory aptitude increases with the electron density of the ring, so the presence of electron-donating groups makes the rearrangement step faster, which, being the rate-determining step, increases the overall rate of the reaction.
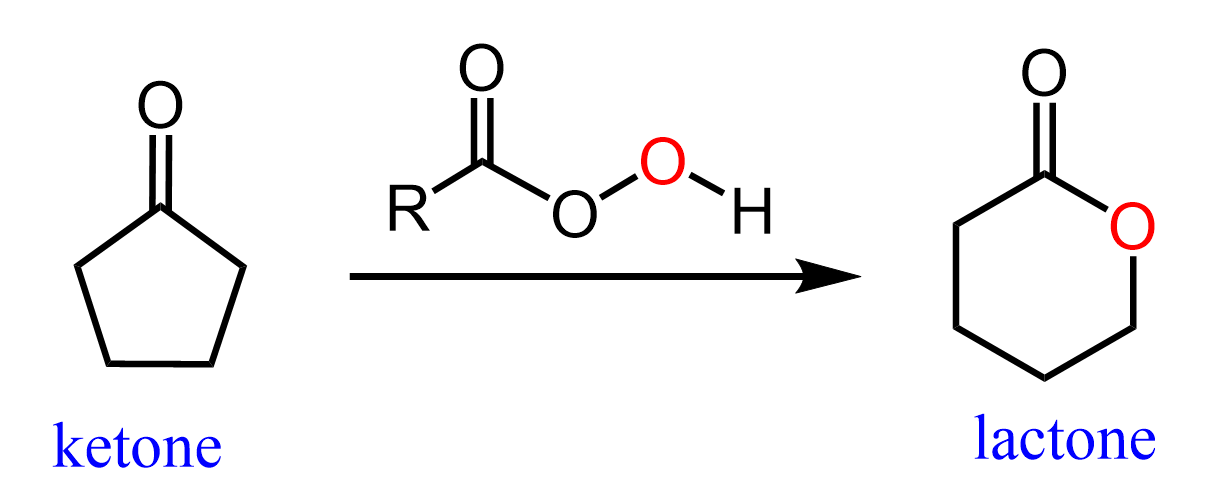
Cyclic Ketones to Lactones
The Baeyer-Villiger Oxidation can also be used for converting cyclic ketones to lactones (cyclic esters), as we have also shown in the first set of relations above:
This is what I wanted to discuss about the Baeyer-Villiger Oxidation, so let me know in the comments if there is anything else that needs to be added or addressed.
Check Also
- Nomenclature of Aldehydes and Ketones
- How to Name a Compound with Multiple Functional Groups
- Preparation of Aldehydes and Ketones
- Nucleophilic Addition to Carbonyl Groups
- Reduction of Aldehydes and Ketones
- Reactions of Aldehydes and Ketones with Water
- Reactions of Aldehydes and Ketones with Alcohols: Acetals and Hemiacetals
- Acetals as Protecting Groups for Aldehydes and Ketones
- Formation and Reactions of Imines and Enamines
- Reductive Amination
- Acetal Hydrolysis Mechanism
- Imine and Enamine Hydrolysis Mechanism
- Hydrolysis of Acetals, Imines, and Enamines-Practice Problems
- Reaction of Aldehydes and Ketones with CN, Cyanohydrin Formation
- The Wittig Reaction: Examples and Mechanism
- The Wittig Reaction: Practice Problems
- Aldehydes and Ketones to Carboxylic Acids
- Reactions of Aldehydes and Ketones – Practice Problems
- Aldehydes and Ketones Reactions Practice Quiz
- Reactions Map of Aldehydes
- Reactions Map of Ketones
