To convert an alkene to an alkyne, we need to first halogenate it to prepare a dihalide, which can then be treated with a strong base such as sodium amide (NaNH2) to achieve a double elimination:

A triple bond can also be formed from a geminal dihalide:

To distinguish these, you can go with geminal as being “jammed” on one carbon:

More scientifically, it comes from the Latin geminus – “twin.”
The Mechanism of Dihalide Elimination
The reaction goes by the E2 mechanism and is similar to how alkenes are prepared. The difference here is that two halogen atoms are needed because the triple bond is formed by two consecutive elimination reactions:

Although the elimination can be performed with regular bases such as hydroxides and alkoxides, a stronger base is needed to achieve an elimination of the intermediate vinyl halide. Most often, Sodium amide (NaNH2), dissolved in liquid ammonia (NH3), is used.
The aqueous workup is needed to protonate the alkynide ion that is formed because of the relatively acidic proton of the terminal alkyne (pKa of 25).
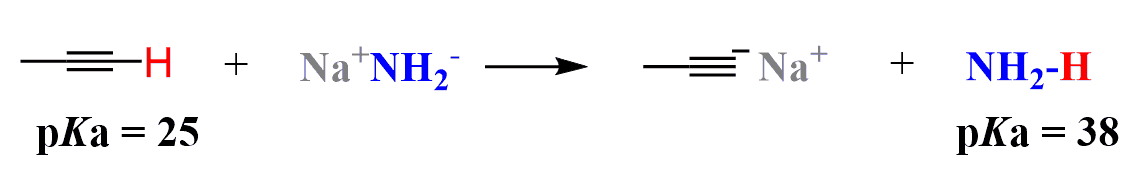
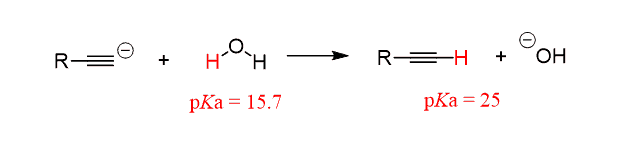
Even when an internal alkyne is prepared, an excess of sodium amide is often used to make sure the reaction goes to completion. Therefore, the water work-up is used in any case the protonate the alkynide ion and also quench the possible excess of NaNH2.
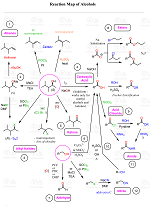
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!