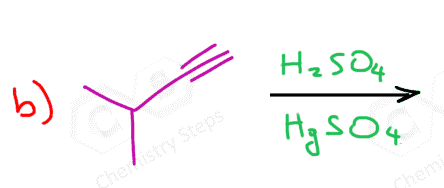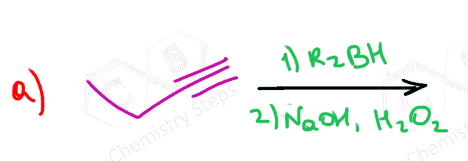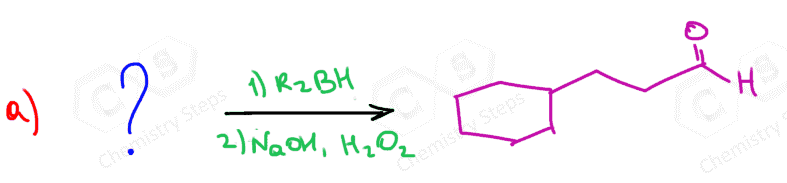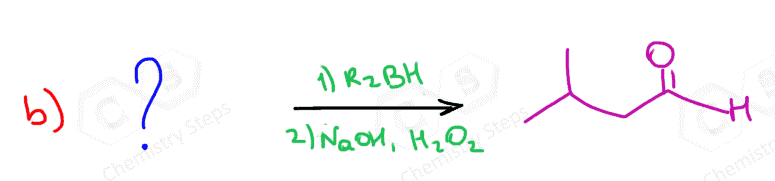Alkynes to Ketones
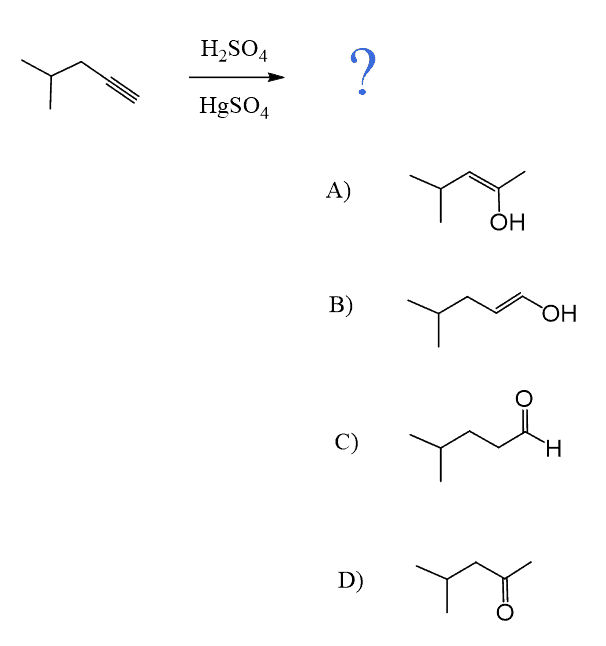
Alkynes can be converted to aldehydes and ketones via hydration reactions such as 1) Acid-Catalyzed Hydration, 2) Oxymercuration-Demercuration, and 3) Hydroboration-Oxidation.
The first two are used for Markovnikov conversion of terminal alkynes to ketones, whereas the hydroboration-oxidation is used for anti-Markovnikov carbonylation of alkynes where the final product is an aldehyde:

The acid-catalyzed hydration goes via formation of an enol which, due to instability, undergoes a keto-enol tautomerization forming a ketone.

Notice that the same conversion can be achieved using HgSO4 instead of acidic catalysis. We have also seen this in the oxymercuration-demercuration of alkenes. However, the reason for using a mercury catalyst here is the slower reaction of alkynes compared to alkenes. In the case of alkenes, HgSO4 is used to prevent possible rearrangements of the carbocation formed upon protonation of the double bond.
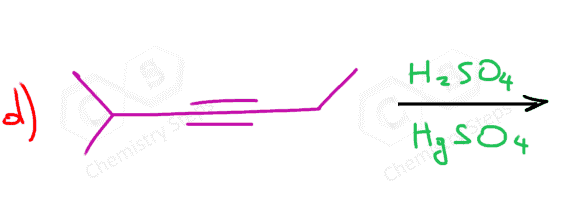
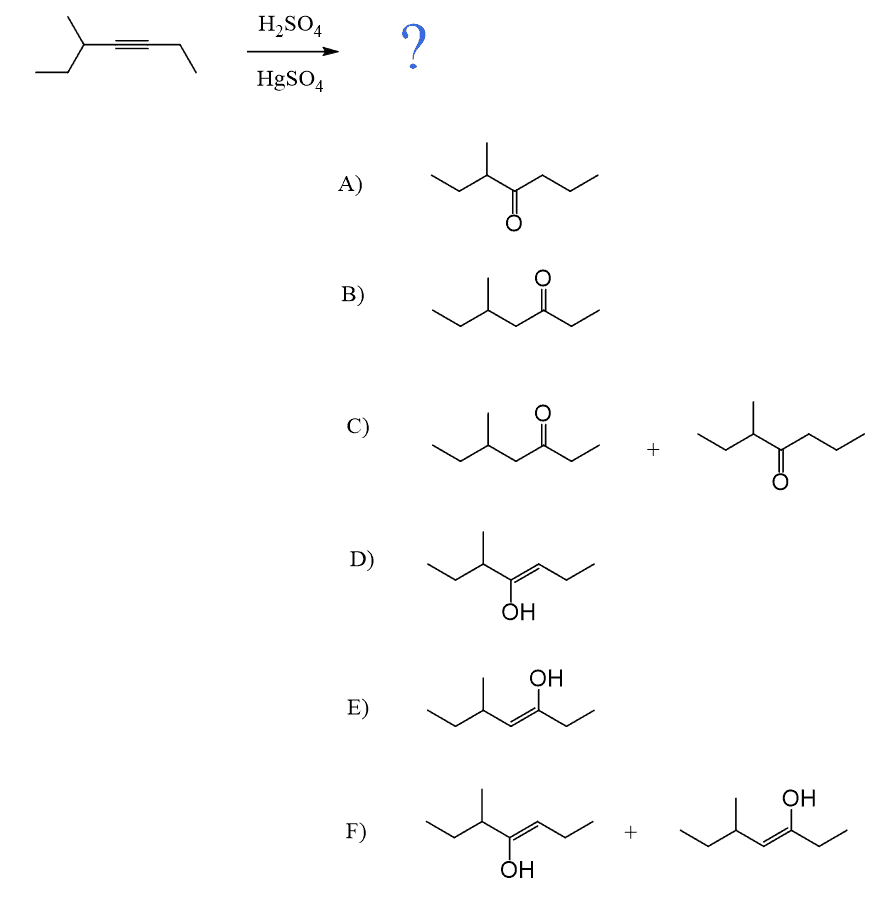
Unsymmetrical internal alkynes give a mixture of two ketones as there is no regioselectivity in the formation of the vinyl carbocation as both have equal stability:
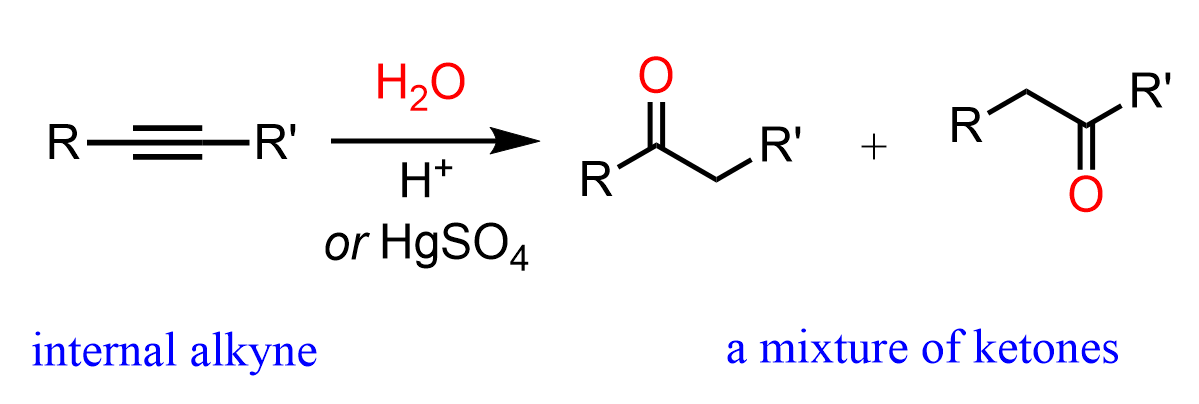
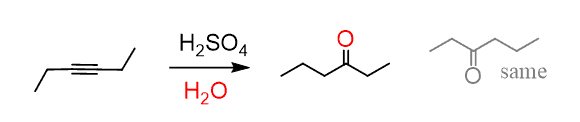
Symmetrical internal alkynes can only form one ketone regardless of the hydration method:
Alkynes to Aldehydes
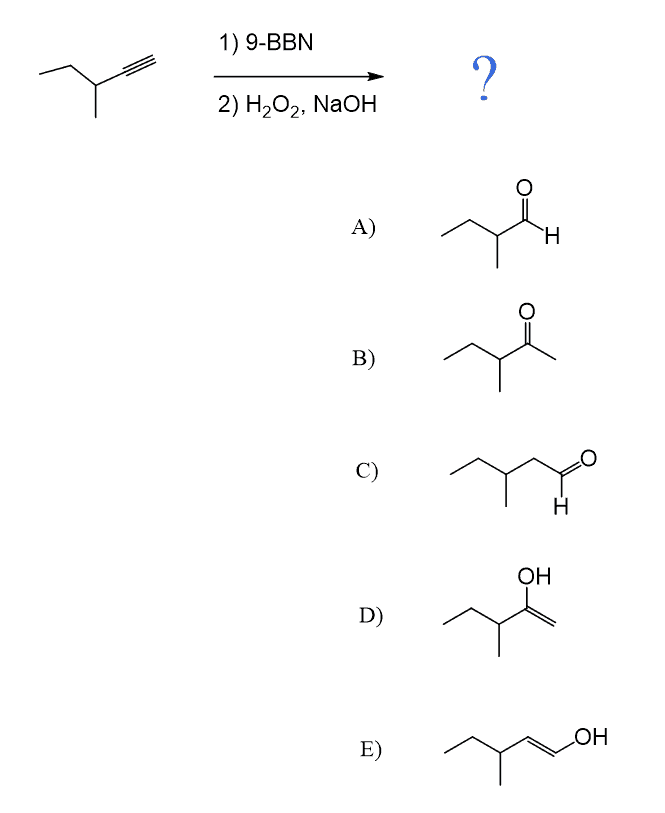
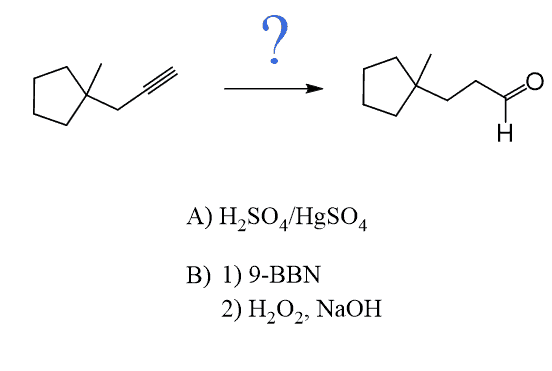
The only direct way of converting alkynes to aldehydes is their hydroboration-oxidation. This only works for terminal alkynes as the anti-Markovnikov addition of an OH group to one of the π bonds gives an enol that tautomerizes to the corresponding aldehyde:

Because alkynes have two π bonds, diallyl boranes (R2BH) are often used to prevent the second addition of the boron to the boron-substituted alkene. The most common alkyl borane is the 9-BBN:

You can find the mechanism of hydroboration-oxidation as well as acid-catalyzed-hydration and oxymercuration in this article on “Hydration Reactions of Alkynes”.
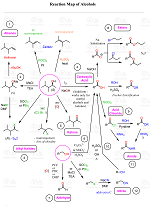
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!