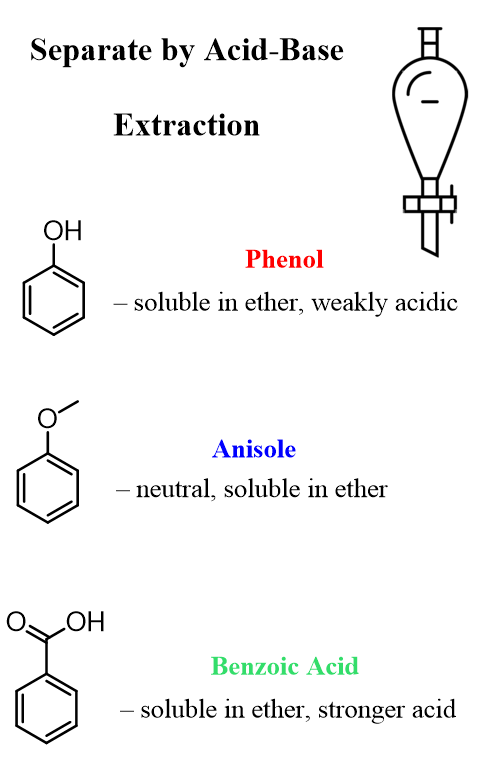
How to Separate a Mixture of Phenol, Anisole, and Benzoic Acid Using Acid-Base Extraction
When separating a mixture of organic compounds, we can take advantage of two key properties:
- Solubility: Whether a compound dissolves in a nonpolar organic solvent like ether or in water.
- Acid-base reactivity: How a compound reacts with an acid or a base (e.g., HCl, NaHCO₃, NaOH, etc.) to form water-soluble salts.
By combining these properties, we can separate the components of a mixture, provided they have respective properties.
In this post, we will discuss the separation of phenol, anisole, and benzoic acid using water and diethyl ether as solvents.
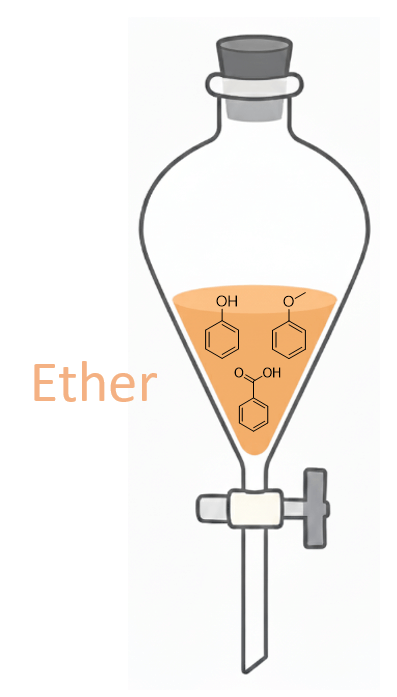
Step 1: Dissolve All the Compounds in Ether
The first step is to dissolve all the components in the organic solvent diethyl ether. Ether is immiscible with water, so when we add an aqueous solution, the mixture will separate into two distinct layers.
By carefully choosing the right strategies, we can selectively push each compound into either the aqueous or organic layer based on its acid-base properties. This process-called liquid-liquid extraction, will allow us to separate phenol, anisole, and benzoic acid step by step.
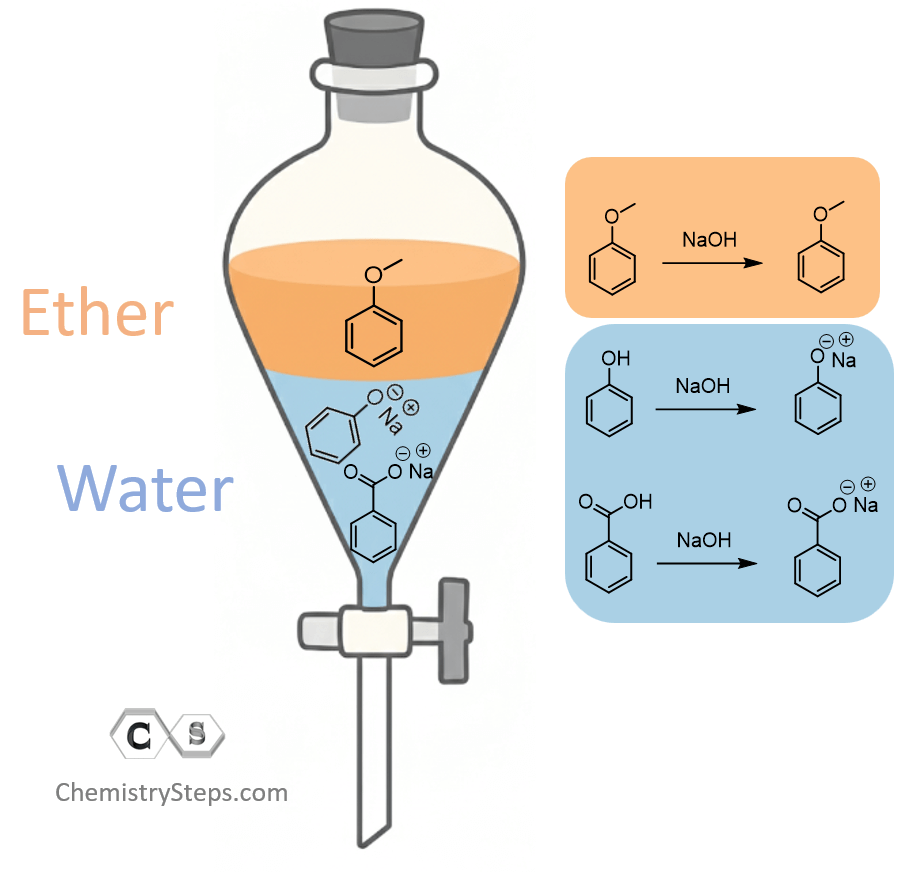
Step 2: Use an Acid or Base to Form a Water-Soluble Salt
Next, we need to consider the solubility of the compounds and their reactivity with either acids or bases. Anisole is essentially unreactive toward acids and bases, so we need to consider the reactivity of phenol and benzoic acid. Both have an acidic proton, although benzoic acid is a much stronger acid, as seen from their pKa values: phenol, pKa ≈ 10; benzoic acid, pKa ≈ 4.2.
What does this tell us? If we add a base like NaOH, it is going to react with phenol and benzoic acid, forming their water-soluble salts. So, in the separatory funnel (sep funnel), we will have the anisole dissolved in the ether (top layer) and the two salts in water (bottom layer):
Note: It is not a rule of thumb that the organic layer is always on the bottom—this depends on the densities of the solvents. In this case, diethyl ether is on top because its density is 0.713 g/mL, and water is on the bottom because it has a higher density of 1.00 g/mL.
Now, separating the salts becomes a challenge at this point, so we need to modify our strategy a little bit.
Step 3: Separate the Acid
Yes, it’s still step 3, and we are going to make it work this time.
What we can do is, instead of NaOH, add a weaker base such as NaHCO₃, which will allow deprotonating the benzoic acid while keeping the phenol as it is in the organic layer:
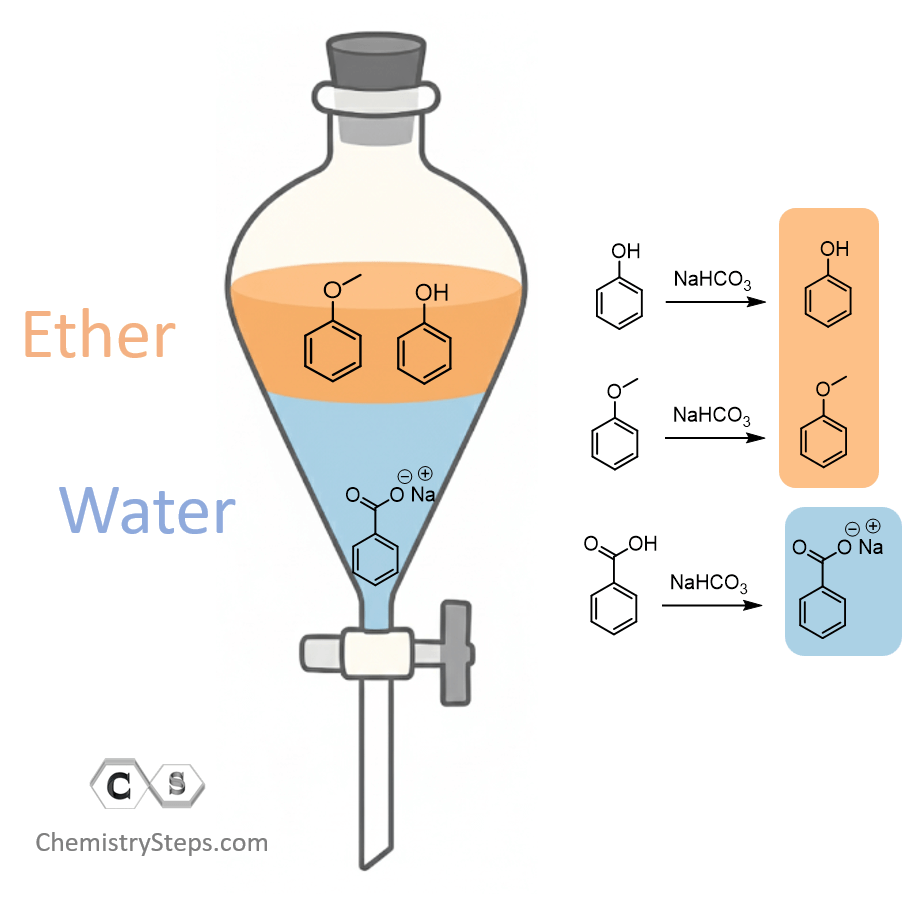
So, at this stage, we have phenol and anisole in the organic layer (top), and the sodium benzoate salt in the bottom water layer.
Step 4: Recover the Acid
We can now separate the solvent layers and add acid to the water layer to protonate the benzoate salt, converting it to its original form of acid, which will go to the organic solvent.
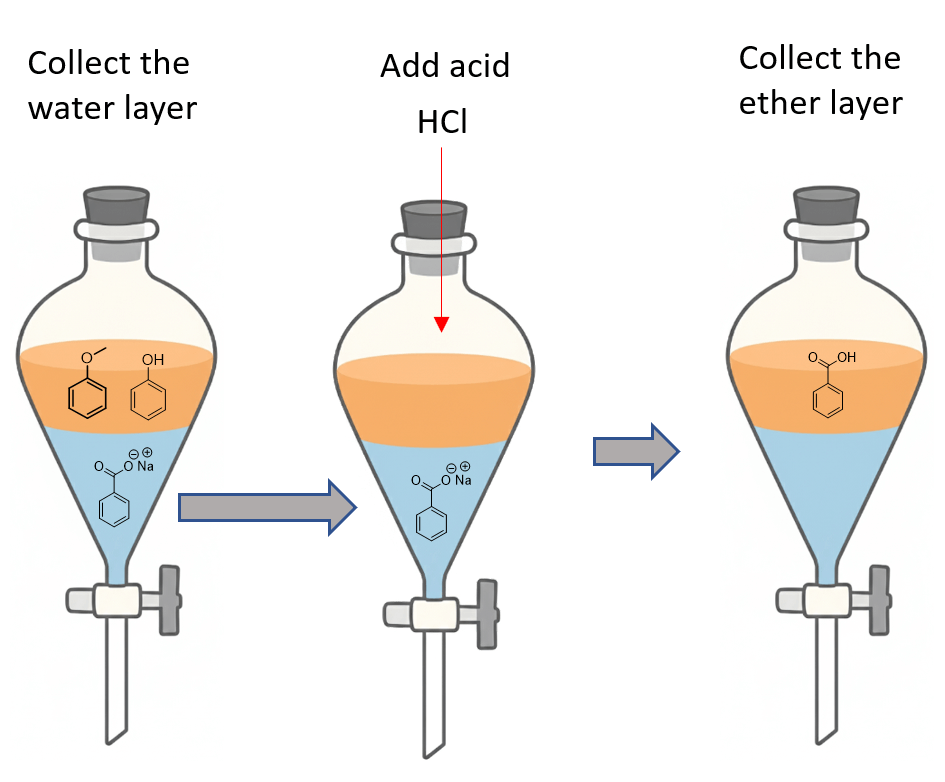
So, we first collected the water layer in a beaker, transferred it to another funnel with ether, and added HCl to convert the salt back to benzoic acid. We then collected the ether layer, and the acid was isolated by evaporating the ether.
Step 5: Separate Phenol and Anisole
At this point, we are left with a mixture of phenol and anisole, and the next step is to separate these two compounds.
Phenol can be deprotonated with a relatively strong base such as sodium hydroxide, forming sodium phenoxide. This salt is water-soluble and therefore will go to the water layer. We can first add some water before adding the base, or if it is a dilute solution, we can add it once to the ether solution.
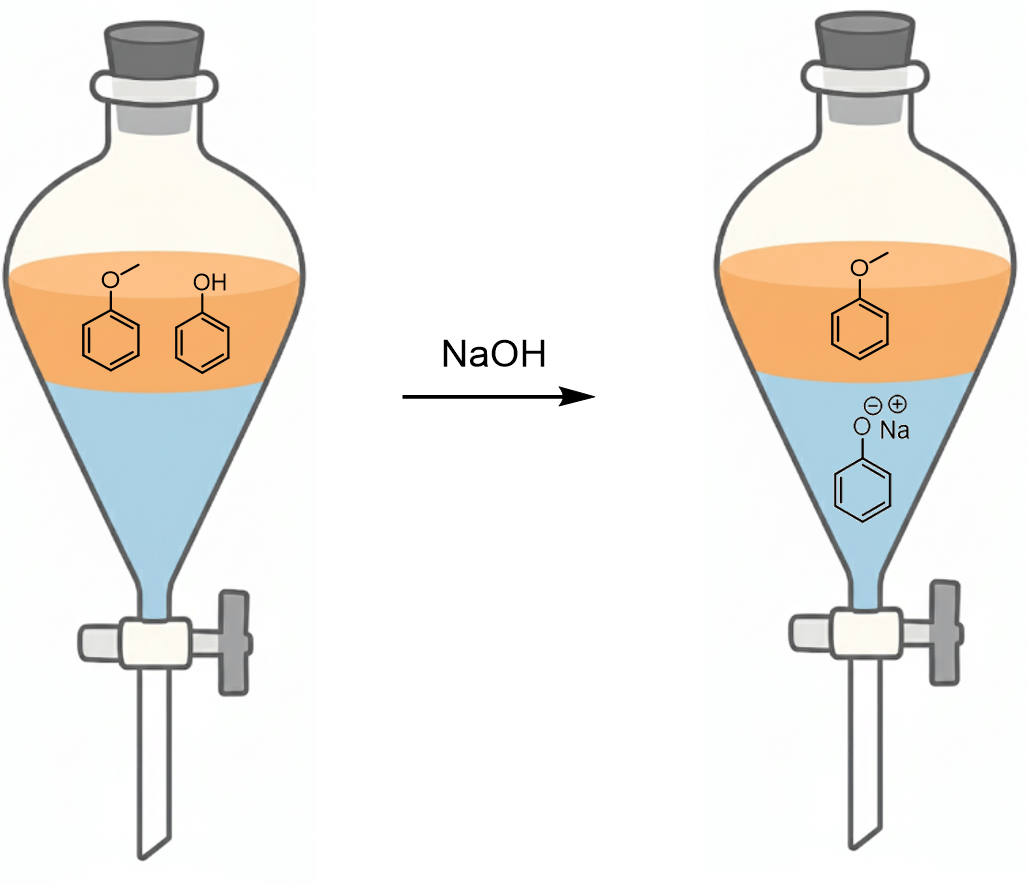
So, now, in the top layer, we have anisole in ether, and in the bottom layer, we have an aqueous solution of the phenoxide and some excess NaOH. We first separate these layers and evaporate the ether to collect the anisole.
For the phenoxide, like we did with benzoic acid, we add some HCl solution to this aqueous solution, neutralizing the base and converting the salt back to phenol, which is then extracted with an ether, and after removing the solvent, phenol is collected.
Summary chart separating Phenol, Anisole, and Benzoic Acid by Acid-Base Extraction
When separating a mixture of phenol, anisole, and benzoic acid, we take advantage of two properties: solubility and acid-base reactivity.
- Dissolve in ether – All three compounds are first dissolved in diethyl ether, which forms a separate layer from water.
- Extract benzoic acid – Add NaHCO₃. Only benzoic acid reacts (pKa ≈ 4.2) to form sodium benzoate, which moves to the aqueous layer. Acidify with HCl to recover benzoic acid.
- Extract phenol – Add NaOH to the ether layer. Phenol (pKa ≈ 10) is deprotonated to sodium phenoxide, which moves to the aqueous layer. Acidify with HCl to recover phenol.
- Isolate anisole – Anisole is neutral and does not react, so it remains in the ether layer and can be isolated by evaporating the solvent.
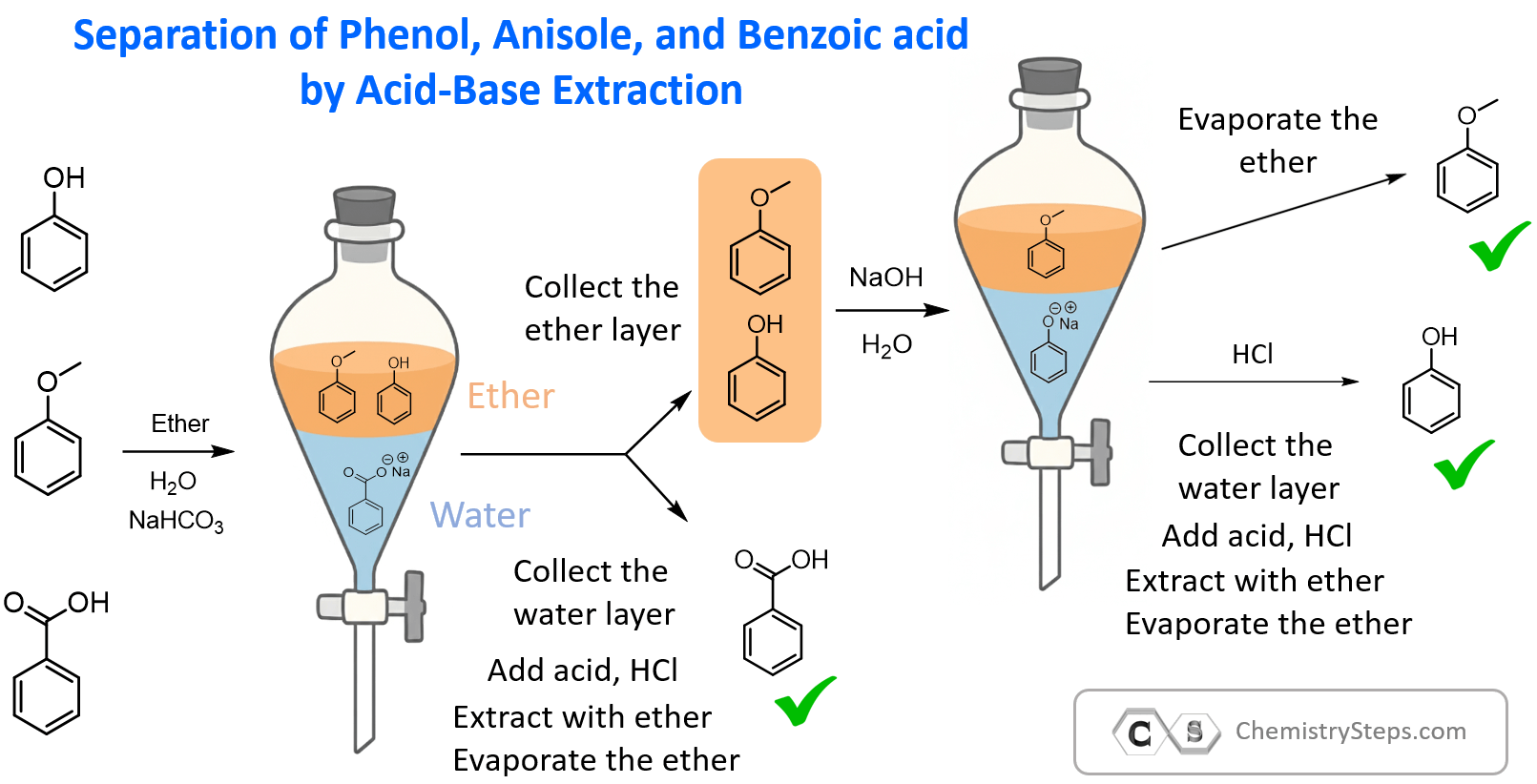
So, this is how we utilize the solubility and acid–base properties of organic compounds to separate them by acid–base extractions.
👨🔬 How would you separate the components of a mixture consisting of hexanoic acid, ethyl 4-aminobenzoate, and naphthalene?
Check Also
- Organic Acids and Bases
- Organic acid-base mechanisms
- Acid Strength and pKa
- How to Determine the Position of Equilibrium for an Acid-Base Reaction
- Inductive and Resonance (Mesomeric) Effects
- Factors That Determine the pKa and Acid Strength
- How Resonance Affects Acidity and Basicity
- How to Choose an Acid or a Base to Protonate or Deprotonate a Given Compound
- Lewis Acids and Bases
- Basicity of Amines
- Organic Acids and Bases Practice Problems
- Organic Acids and Bases Quiz
