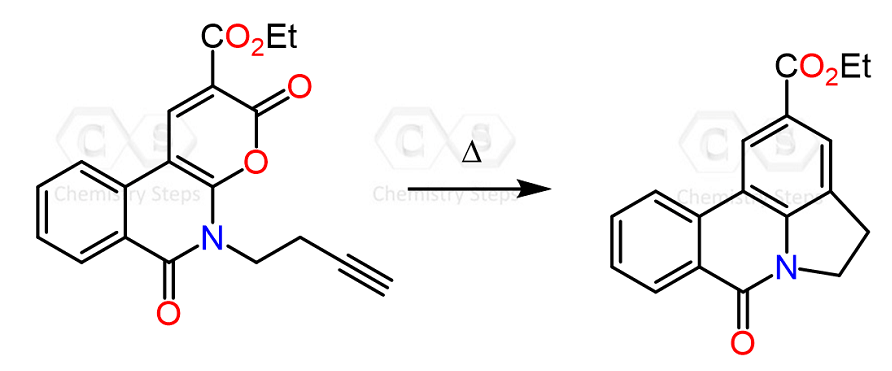
Let’s talk about the retro Diels-Alder reaction. In simpler words, this is the reverse of a given Diels-Alder reaction, which we are mainly going to need for identifying the starting materials that reacted in a particular Diels-Alder reaction.
If you remember or may have noticed, there is almost always a cyclohexene ring in the products of Diels-Alder reactions. This is because it is a 4+2 cycloaddition reaction, meaning we have four atoms from the diene and two from the dienophile that participate in the reaction:
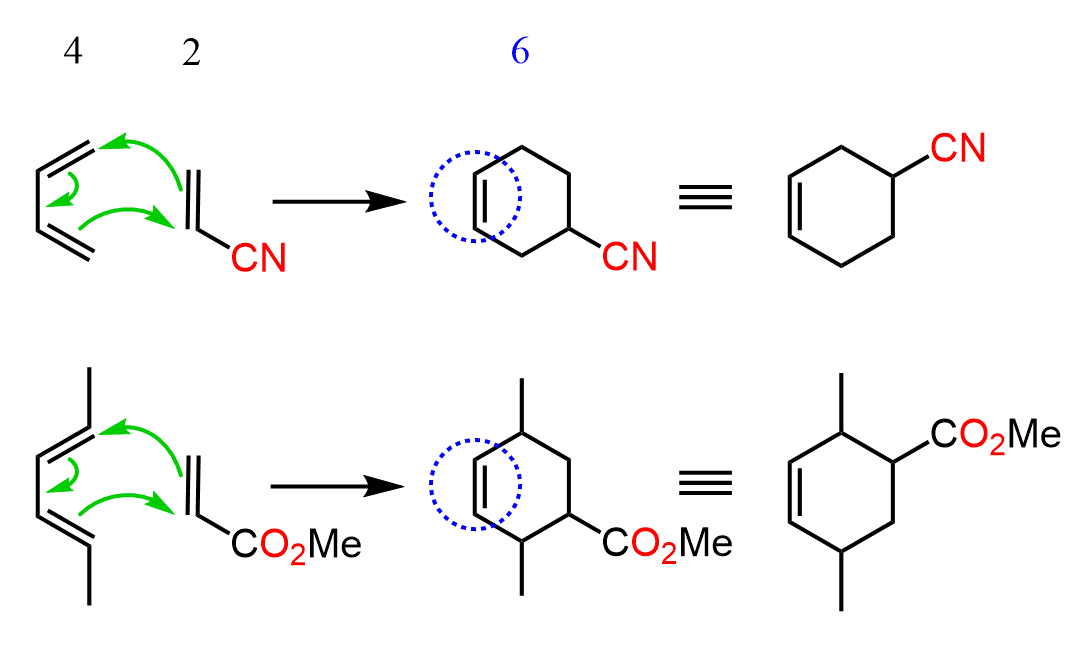
Now, going backwards, the easiest strategy for identifying the starting material is to add two imaginary double bonds right where the actual double bond of the cyclohexene is. When adding these double bonds, also erase the actual one, and this essentially is the structure of the diene that had been used. The only thing left to do is add a double bond to the dienophile, and we have the correct set of the starting material:
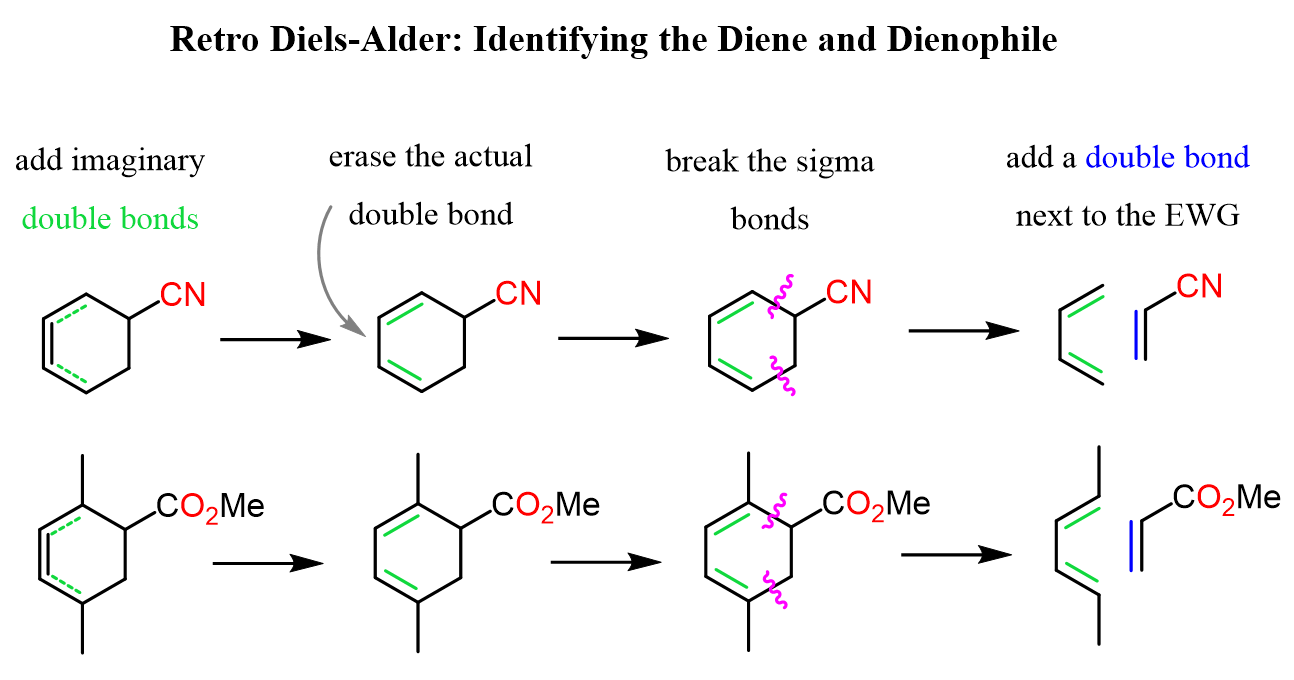
Although this does not change the actions and the overall idea, you can also start by adding an imaginary double bond next to the electron-withdrawing group of the dienophile, which is positioned right across the actual double bond in the product:
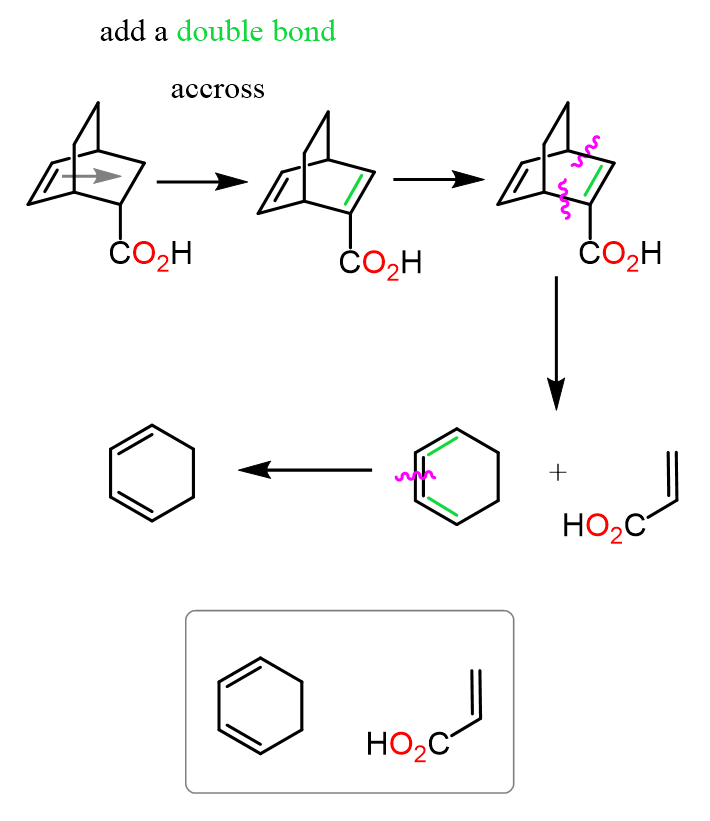
Remember, this is an important pattern/requirement in the Diels-Alder reaction: the presence of electron-withdrawing groups in the dienophile, which we can also use to identify the reactants.
The Mechanism of Retro Diels-Alder Reaction
When drawing the mechanism of a retro Diels-Alder reaction, the good news is that it is still an electrocyclic reaction and is not much different from the actual Diels-Alder reaction. All you need to do is start an arrow from the middle of the double bond and push it either up or down. This forces breaking the single bond in that direction by making a new double bond next to the electron-withdrawing group of the dienophile.
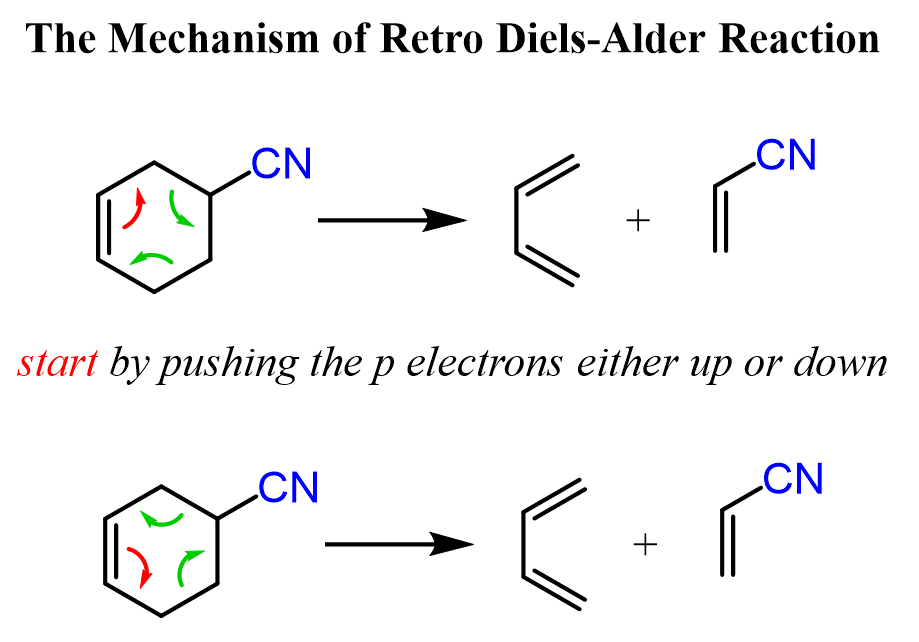
The last step (remember this all happens at the same time – it is a concerted mechanism) is to make a new double bond based on the electron flow – this generates the second double bond of the diene:
What Favors the Retro Diels-Alder Reaction
The retro Diels-Alder reaction is not just an imaginary tool we use to identify the reactants. It does actually happen at elevated temperatures (200–300 °C).
So, why does it happen at such high temperatures?
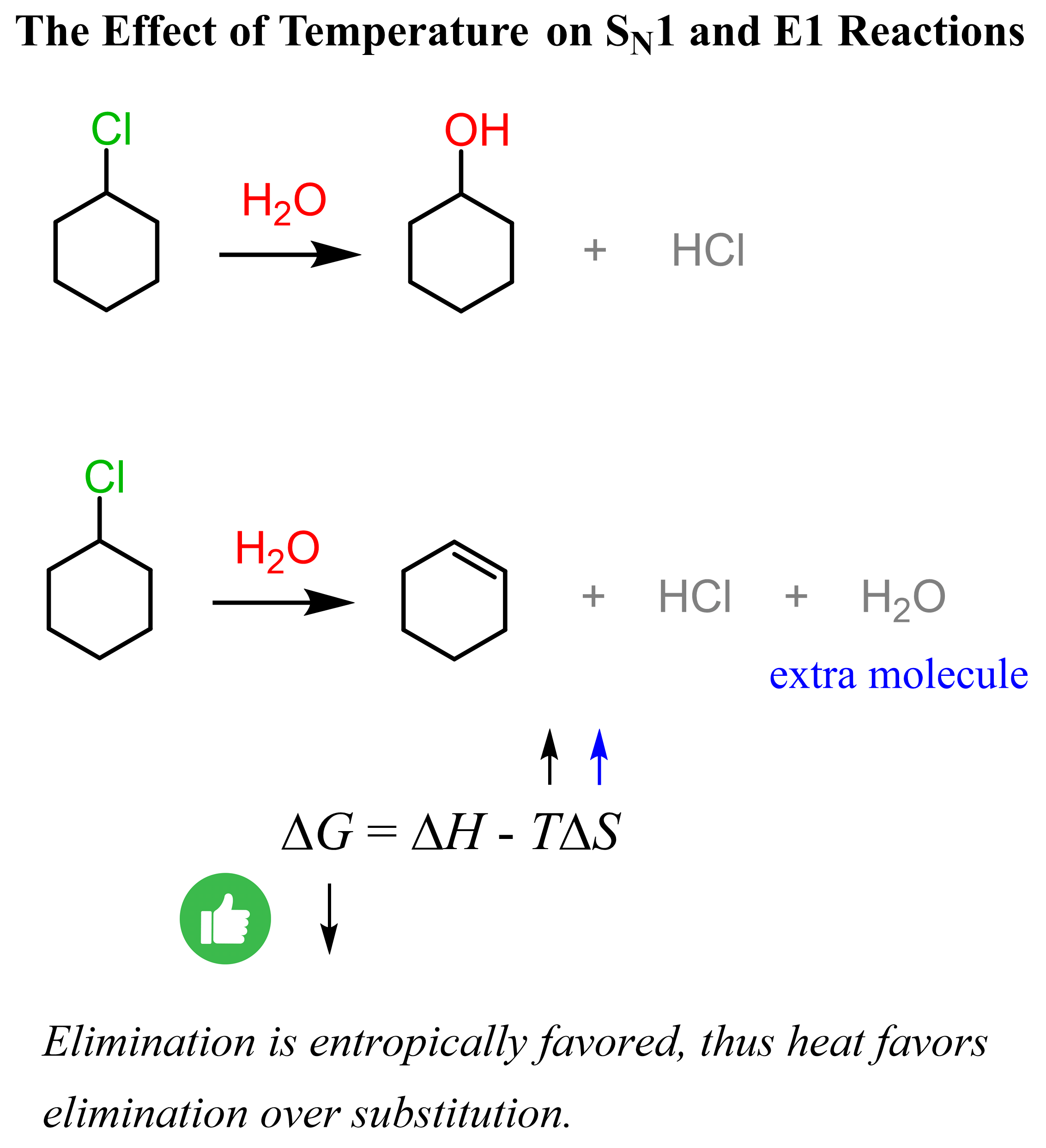
The key factor here is the entropy: there are more products than reactants in retro Diels-Alder reactions; more specifically, we have a 1:2 ratio. Remember from the Gibbs free energy that the TΔS factor comes with a negative sign, which means the higher the temperature, the smaller the ΔG, thus the spontaneity of the reaction.
This is similar to what we observed in the substation and elimination reactions, where always emphasized that heat favors elimination:
Another factor facilitating the retro Diels-Alder reaction is the formation of gases – most often CO2 and N2. These are especially favored in terms of entropy. For example, nitrogen gas is released in the following Diels-Alder reaction of a tetrazine:
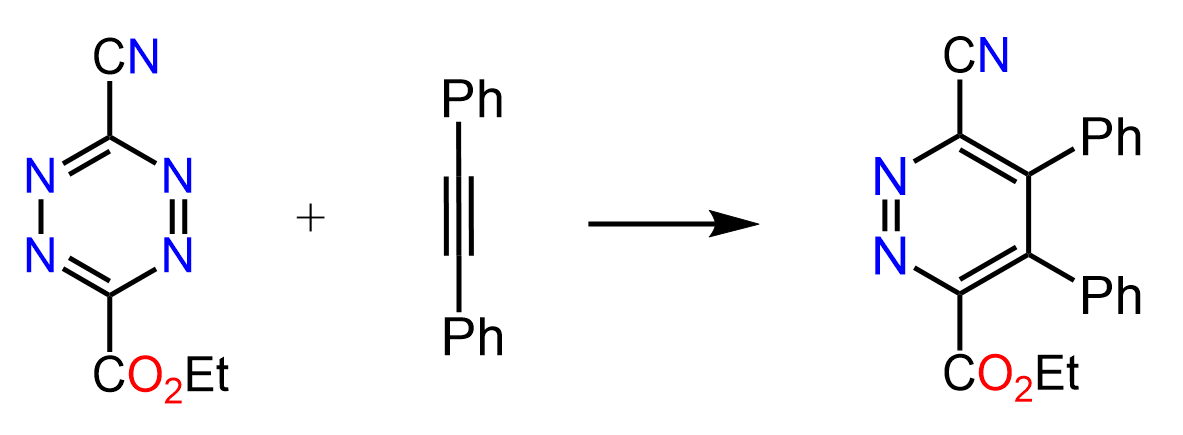
You may say, the product does not look right – it was supposed to be a bicyclic compound. And that is because of a retro Diels-Alder reaction followed by the cycloaddition reaction.
What happens is that first we have a Diels-Alder reaction, which forms a highly strained bicyclic compound, which then undergoes a retro Diels-Alder reaction to give the final product:
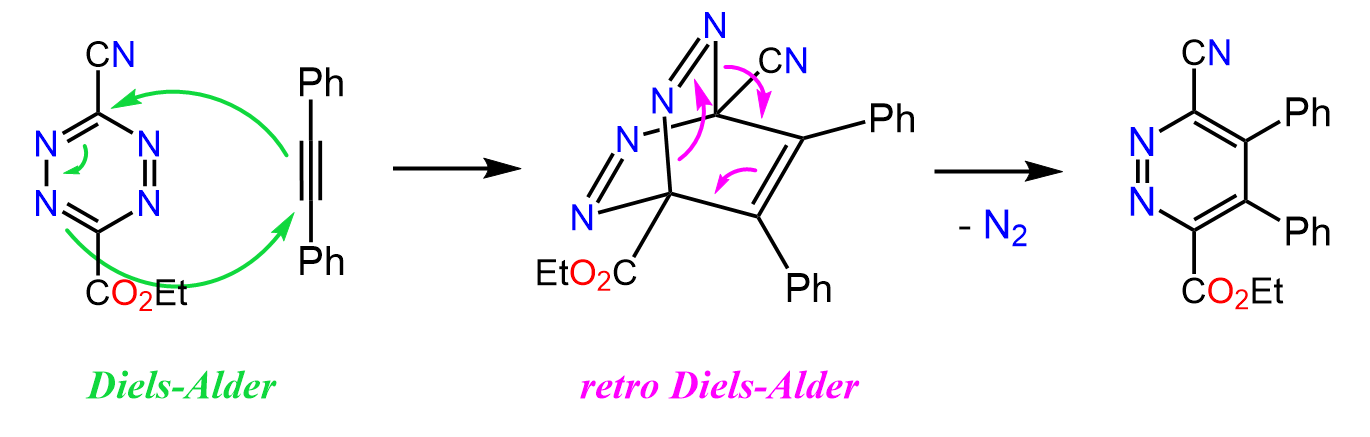
Notice that this time the retro Diels-Alder does not produce the starting dene and dienophile – it is driven forward irreversibly because of the loss of nitrogen.
The Diels-Alder reactions of tetrazines are examples of inverse electron-demand Diels–Alder reactions, where the roles are reversed compared to the normal Diels-Alder reaction: instead of an electron-rich diene reacting with an electron-poor dienophile, an electron-poor diene, tetrazine, reacts with an electron-rich dienophile. You can read more about this in the articles: Chem. Soc. Rev., 2017, 46, 4895-4950,
Chem. Asian J. 2017, 12, 2142 – 2159, RSC Adv., 2013, 3, 7642-7666
The Stereochemistry of Retro Diels-Alder Reaction
To identify the stereochemistry of the starting material in a Diels-Alder reaction, identify if the groups of the dienophile are cis or trans. For example, the groups in the following Diels-Alder adduct are trans, and therefore, they had also been trans in the reactant. If the groups are cis, then they have been cis in the starting martial too:
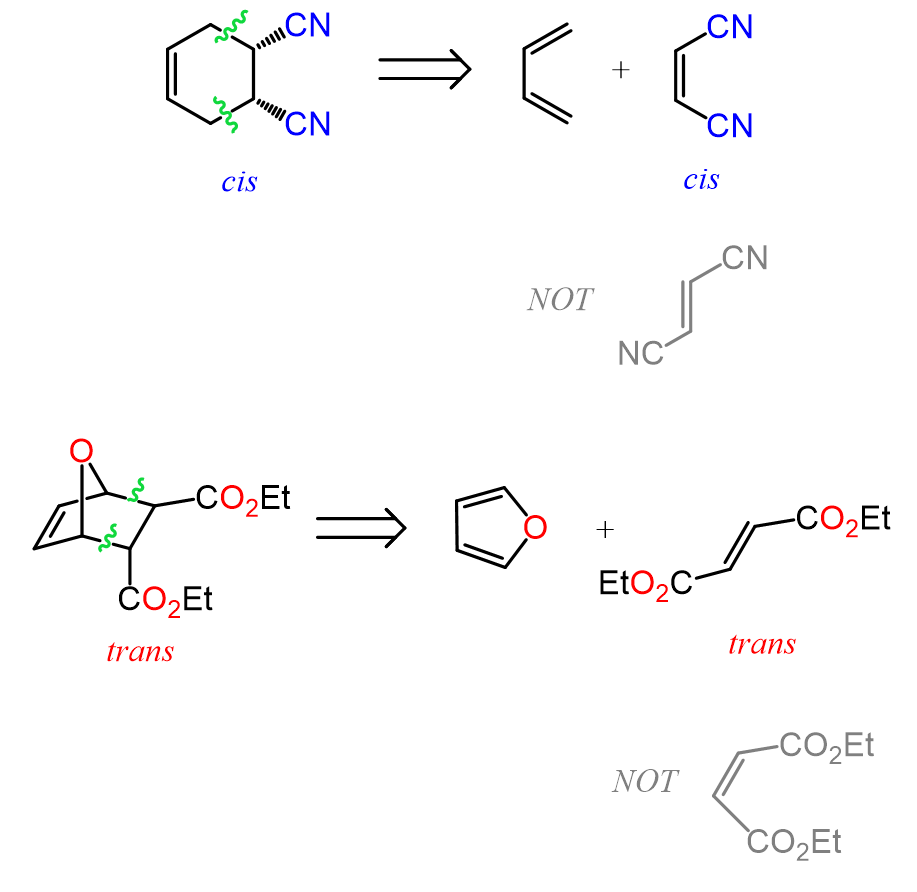
To summarize, the retro Diels-Alder reaction is a powerful tool for working backwards and identifying the starting materials of a Diels-Alder reaction by recognizing the characteristic cyclohexene pattern and strategically placing the missing double bonds. Mechanistically, it is a concerted process similar to the forward reaction, but favored at high temperatures due to entropy, especially when small, stable molecules like CO₂ or N₂ are released. In some cases, such as tetrazine reactions, the retro Diels-Alder step becomes essentially irreversible, driving the reaction forward. Finally, just like in the forward Diels-Alder reaction, stereochemistry is preserved, allowing you to determine whether substituents in the starting materials were cis or trans based on the product.
![]()