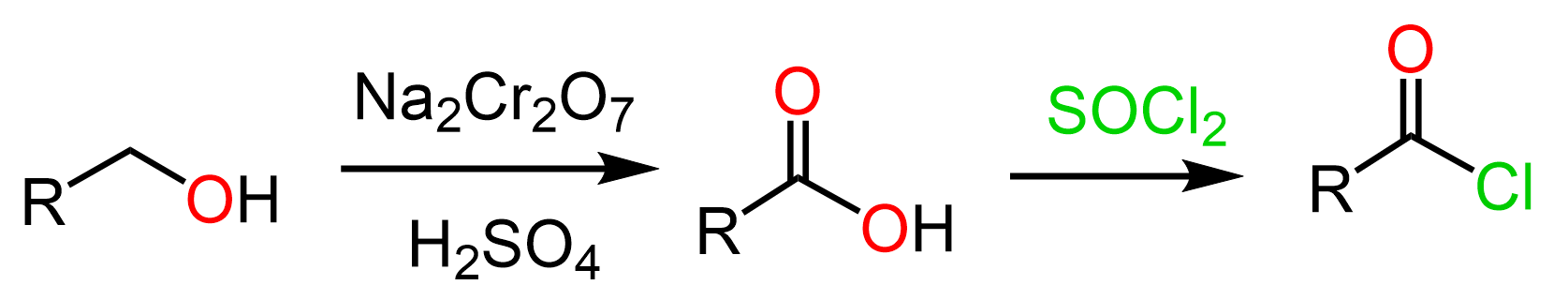
Primary Alcohols to Acid Chlorides
Acid or acyl chlorides are carboxylic acid derivatives, therefore, to convert a primary alcohol to an acid chloride, we need to first oxidize to a carboxylic acid. The carboxylic acid then can be converted to the corresponding acid chloride using thionyl chloride (SOCl2), PCl3, or PCl5.
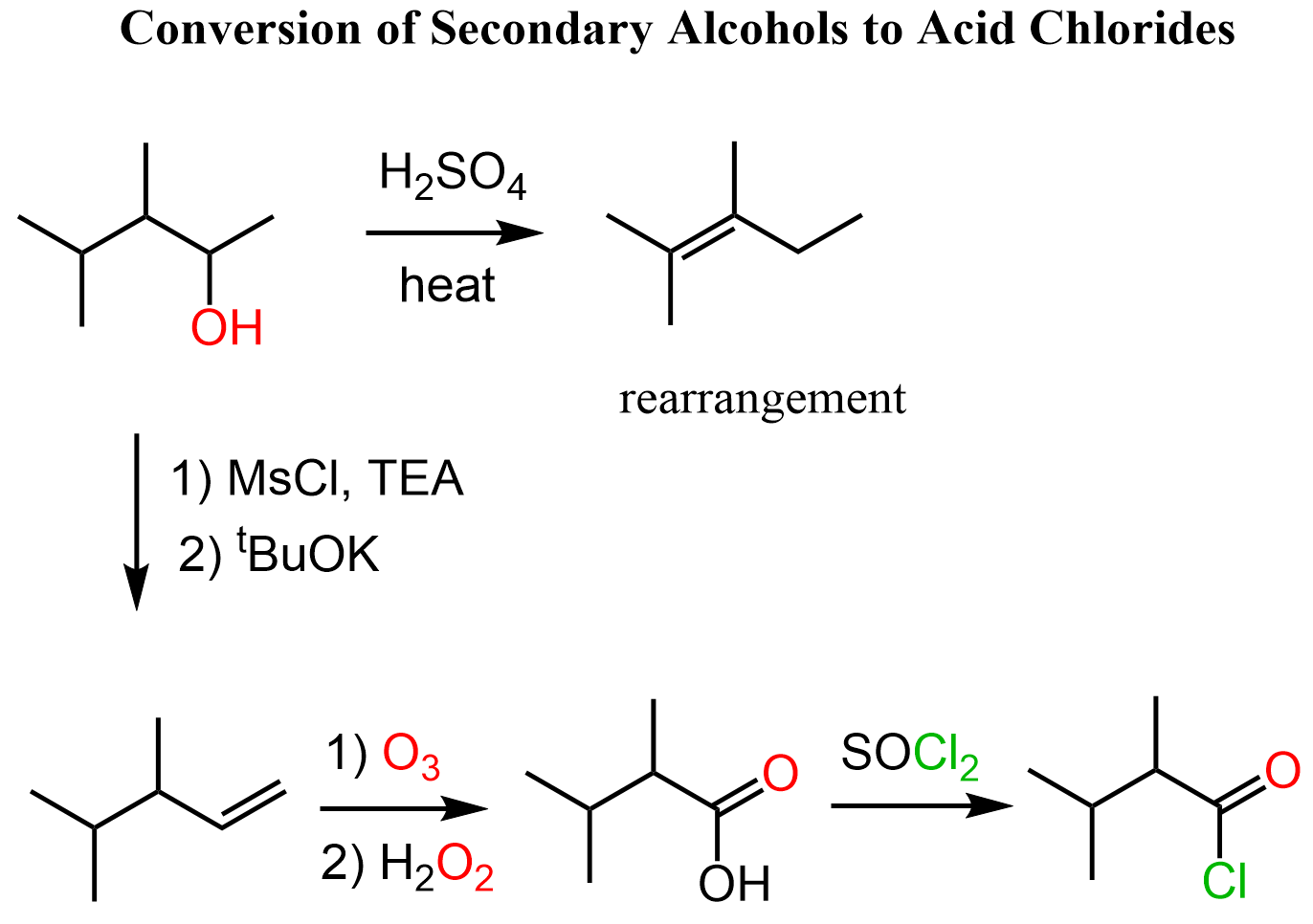
Secondary Alcohols to Acid Chlorides
Secondary alcohols cannot be oxidized to carboxylic acids as the reaction stops at ketones. Therefore, we can either convert the ketone to a carboxylic acid via a haloform (if it is a methyl ketone) reaction or to ester via the Baeyer-Villiger Oxidation and hydrolyze the ester to the corresponding acid. The acid is then converted to acyl chloride as we saw above:
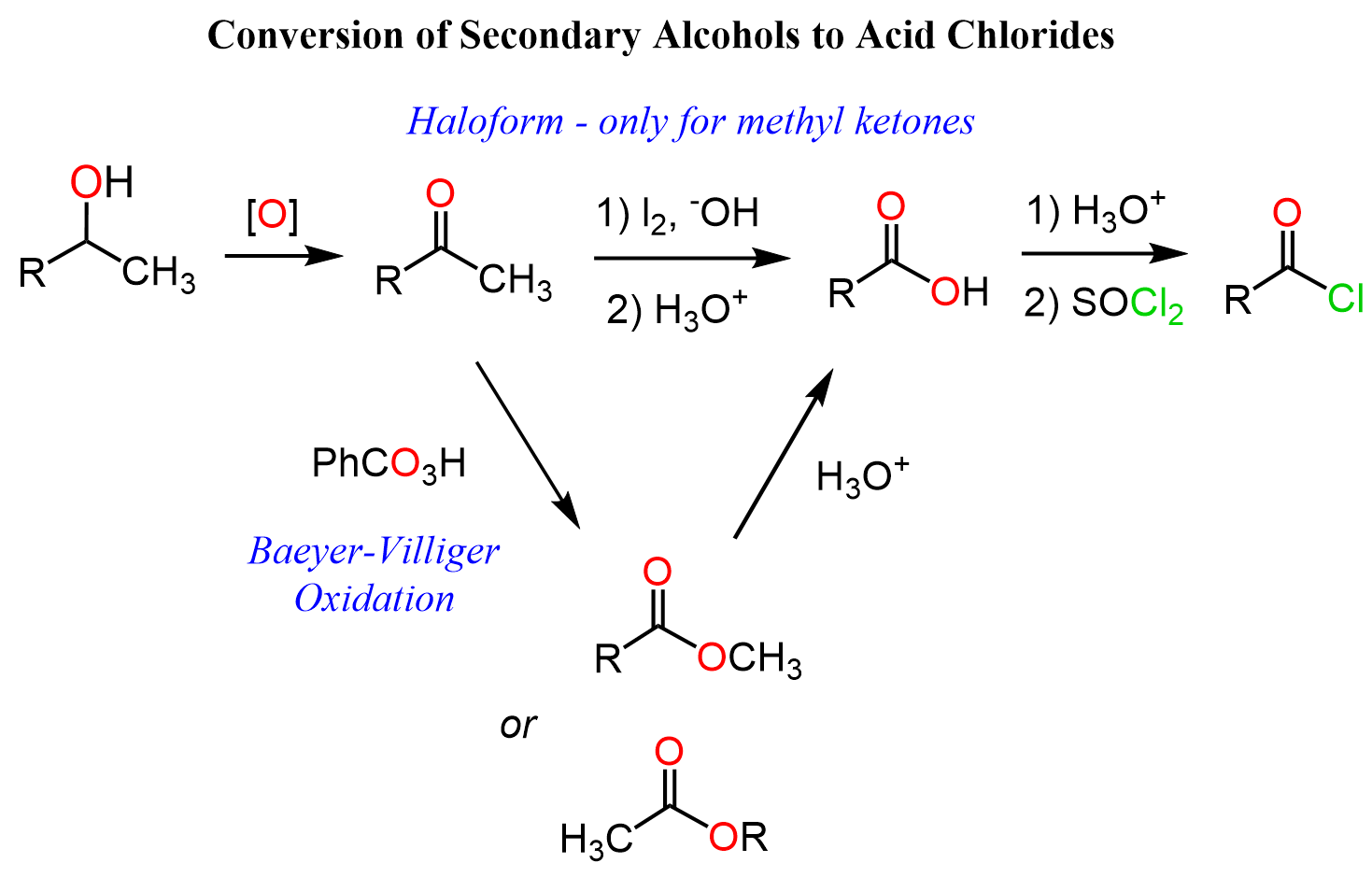
You can read the post on the conversion of aldehydes and ketones to carboxylic acids for details about the mentioned reactions.
Another strategy to convert secondary alcohols to acid chlorides is to first convert the alcohol to an alkene via dehydration and cleave the double bond with a strong oxidizing agent such as ozone with H2O2 or KMnO4 is acidic media:
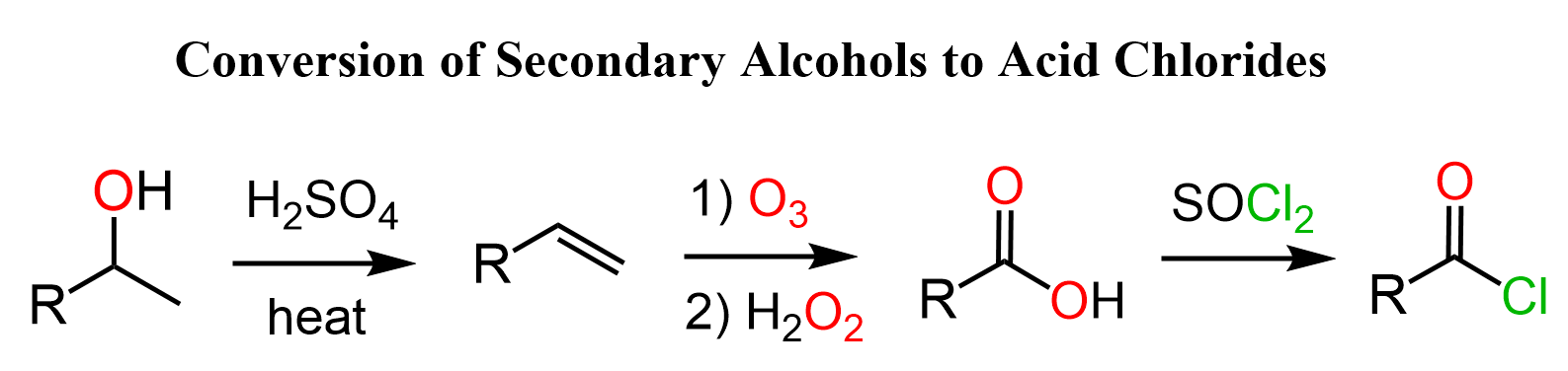
Remember that the acid-catalyzed dehydration of alcohols goes via a carbocation intermediate thus rearrangements are possible. To avoid rearrangements, you can convert the alcohol to an alkyl halide, mesylate, or a tosylate and do a Zaitsev or Hofmann elimination to prepare the needed alkene:
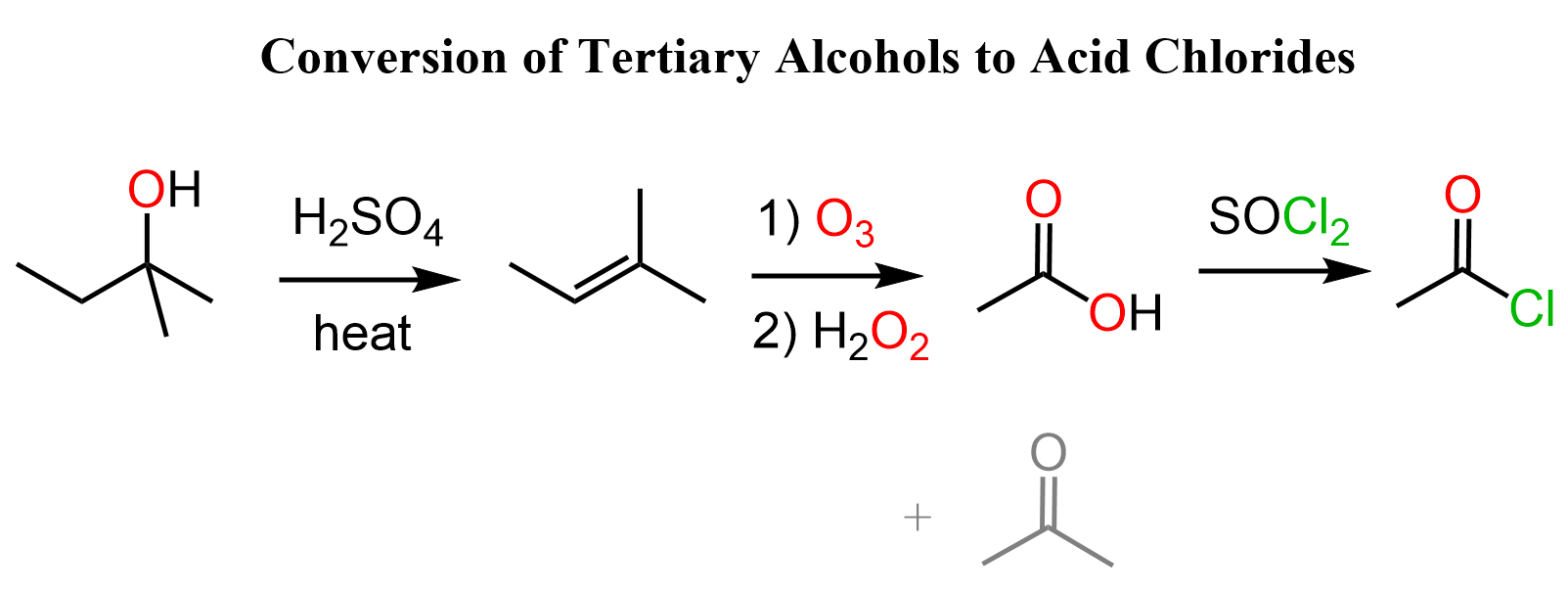
Tertiary Alcohols to Acid Chlorides
Tertiary alcohols have room for further oxidation, so the only strategy here would be converting them to alkenes and oxidizing the latter to a carboxylic acid.
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Preparation of Alcohols via Substitution or Addition Reactions
- Reaction of Alcohols with HCl, HBr and HI Acids
- Mesylates and Tosylates as Good Leaving Groups
- SOCl2 and PBr3 for Conversion of Alcohols to Alkyl Halides
- Alcohols in Substitution Reactions Practice Problems
- POCl3 for Dehydration of Alcohols
- Dehydration of Alcohols by E1 and E2 Elimination
- The Oxidation States of Organic Compounds
- LiAlH4 and NaBH4 Carbonyl Reduction Mechanism
- Alcohols from Carbonyl Reductions – Practice Problems
- Grignard Reaction in Preparing Alcohols with Practice Problems
- Grignard Reaction in Organic Synthesis with Practice Problems
- Protecting Groups For Alcohols in Organic Synthesis
- Oxidation of Alcohols: PCC, PDC, CrO3, DMP, Swern and All of That
- Diols: Nomenclature, Preparation, and Reactions
- NaIO4 Oxidative Cleavage of Diols
- The Pinacol Rearrangement
- The Williamson Ether Synthesis
- Alcohol Reactions Practice Problems
- Naming Thiols and Sulfides
- Reactions of Thiols
- Alcohols Quiz – Naming, Preparation, and Reactions