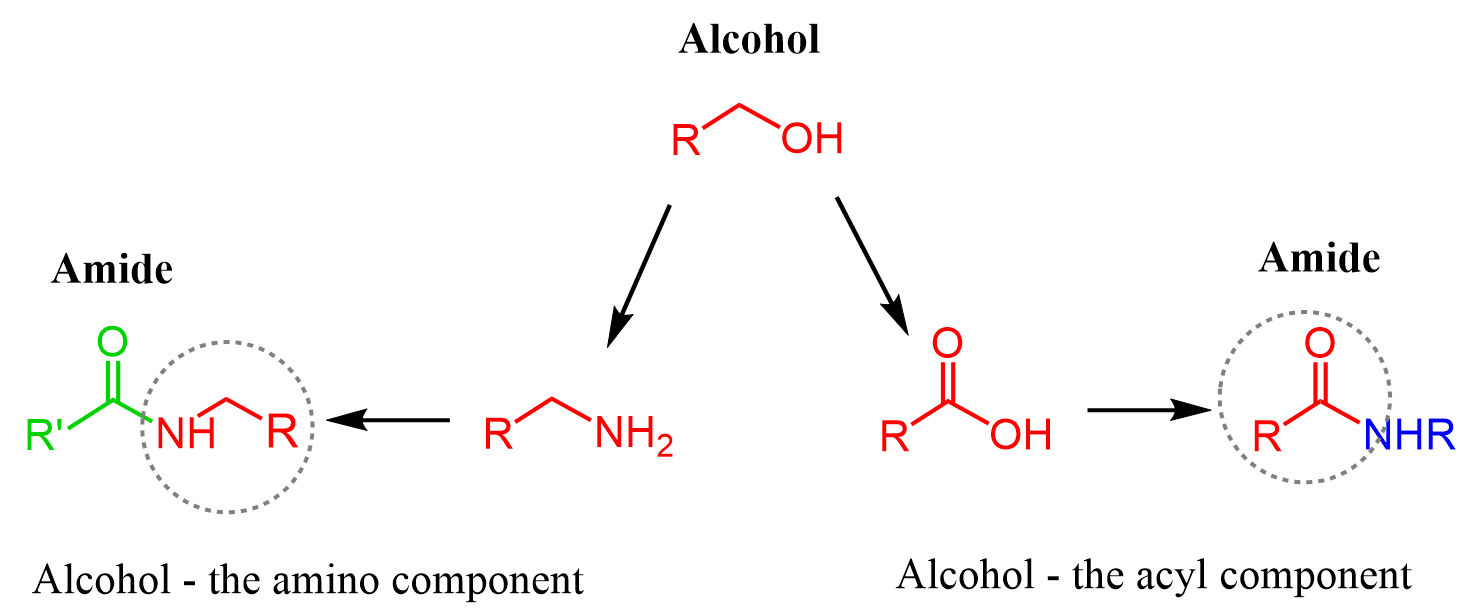
There are a few strategies to convert alcohols to amides, and to discuss them, we first need to decide whether the alcohol is going to be the acyl or amino part of the amide:
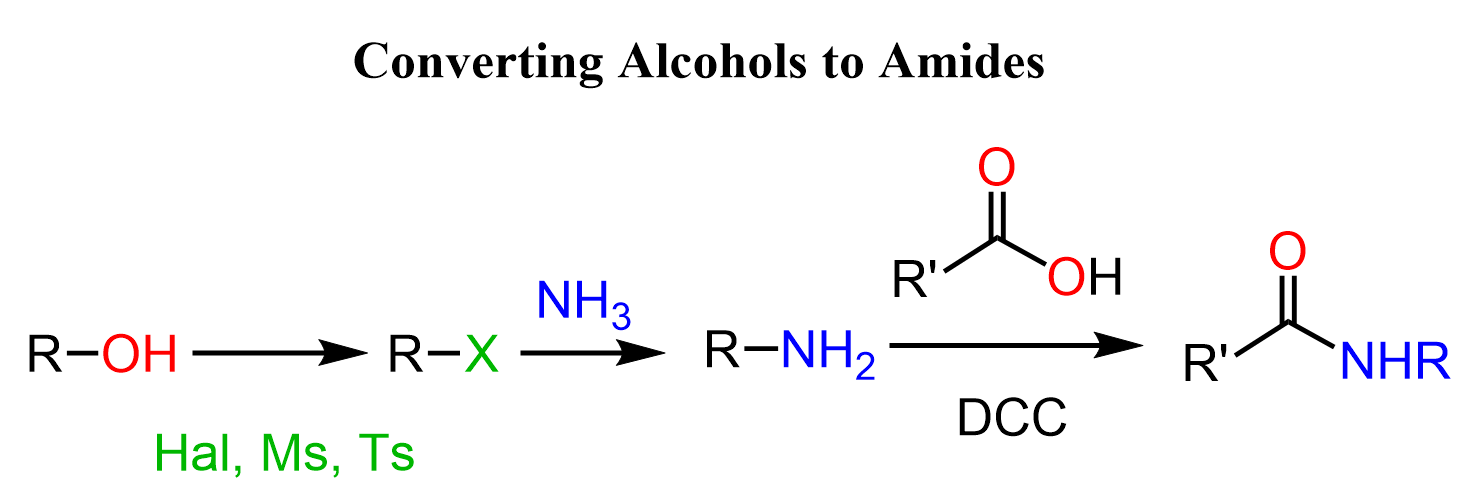
Alcohols to Amides via Amines
Let’s start with the one where we want the alcohol to be the amino part of the amide. For this, we’ll need to convert the alcohol to an amine via activation of the OH which can be achieved by mesylation, tosylation, and converting to a halide using HX acids, PBr3, or SOCl2.
Once we have the amine, we can react with a carboxylic acid using a coupling agent such as DCC or EDC. These reactions are at times challenging too, so the alternative of converting the acid to acid chloride first is also shown in the synthetic scheme. Recall that acid chlorides are the most reactive derivatives of carboxylic acids, and they readily react with all types of nucleophiles including amines.
Let’s now consider the possibilities of incorporating the alcohol as the acyl component of the amide:
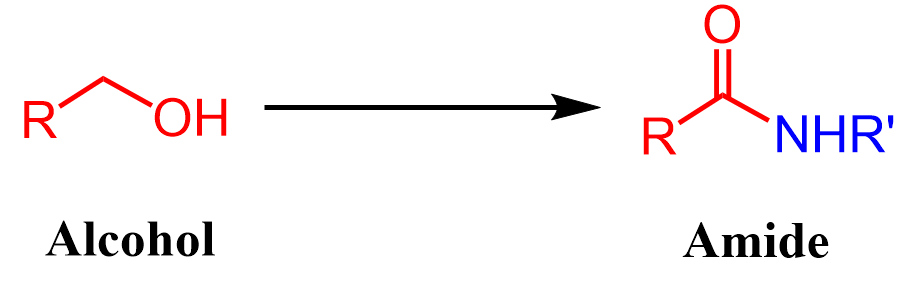
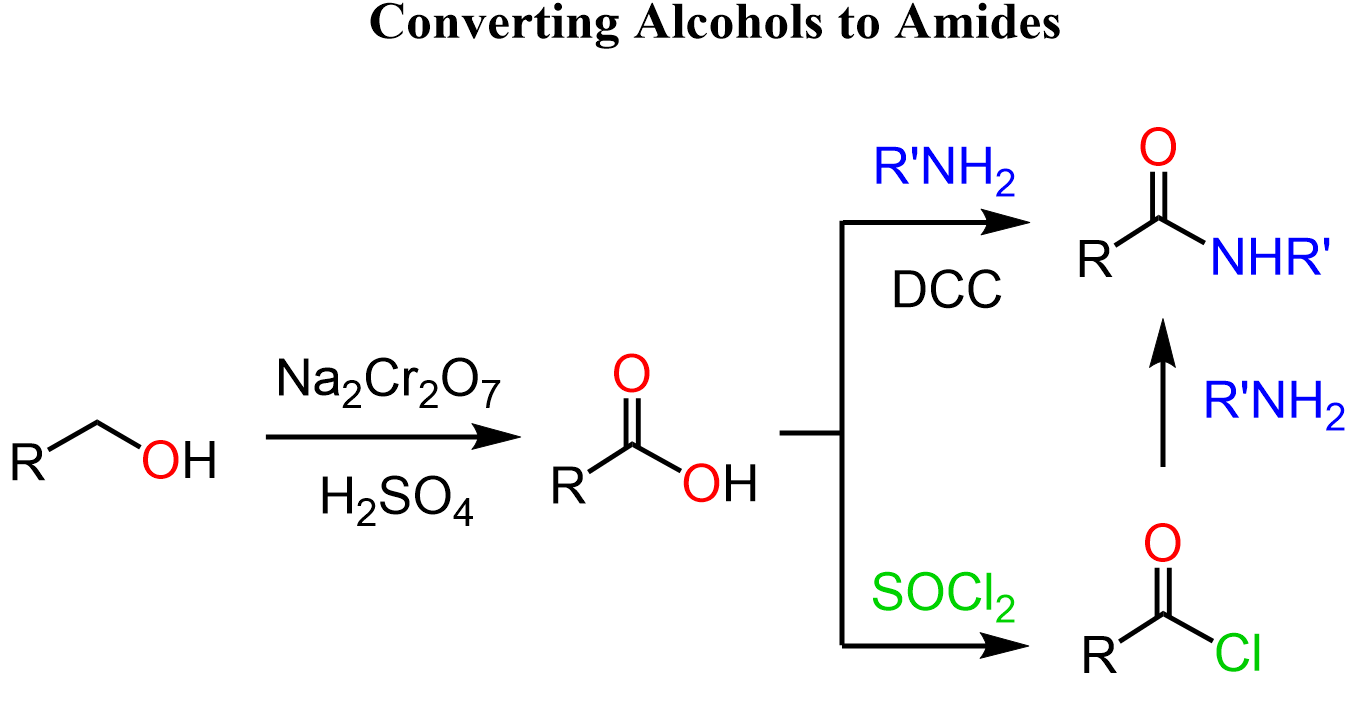
Alcohols to Acyl Component of Amides
The first thing that comes to mind is oxidizing the alcohol to a carboxylic acid with subsequent condensation with an amine by the methods mentioned earlier:
Alcohols to Amides vis Nitriles
Another strategy, which also adds an extra carbon to the molecule, is the conversion of the alcohol to a nitrile, and the latter to an amide.
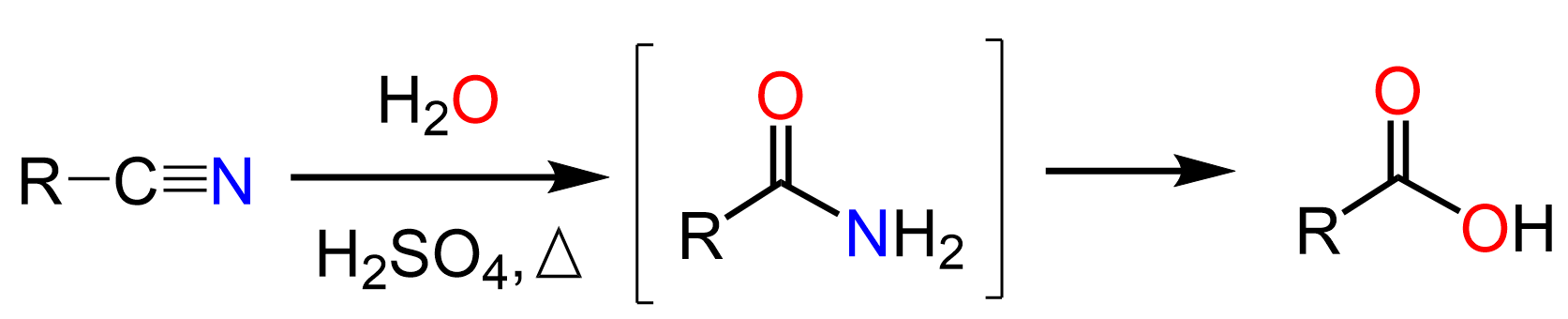
There are a few ways of converting nitriles to amides. One of them is the sure way as it imposes no risk of your instructor counting it as invalid and taking some or all your points off. This is the hydrolysis of the nitrile to carboxylic acid under acidic or basic conditions at elevated temperatures followed by a condensation of the acid with an amine:
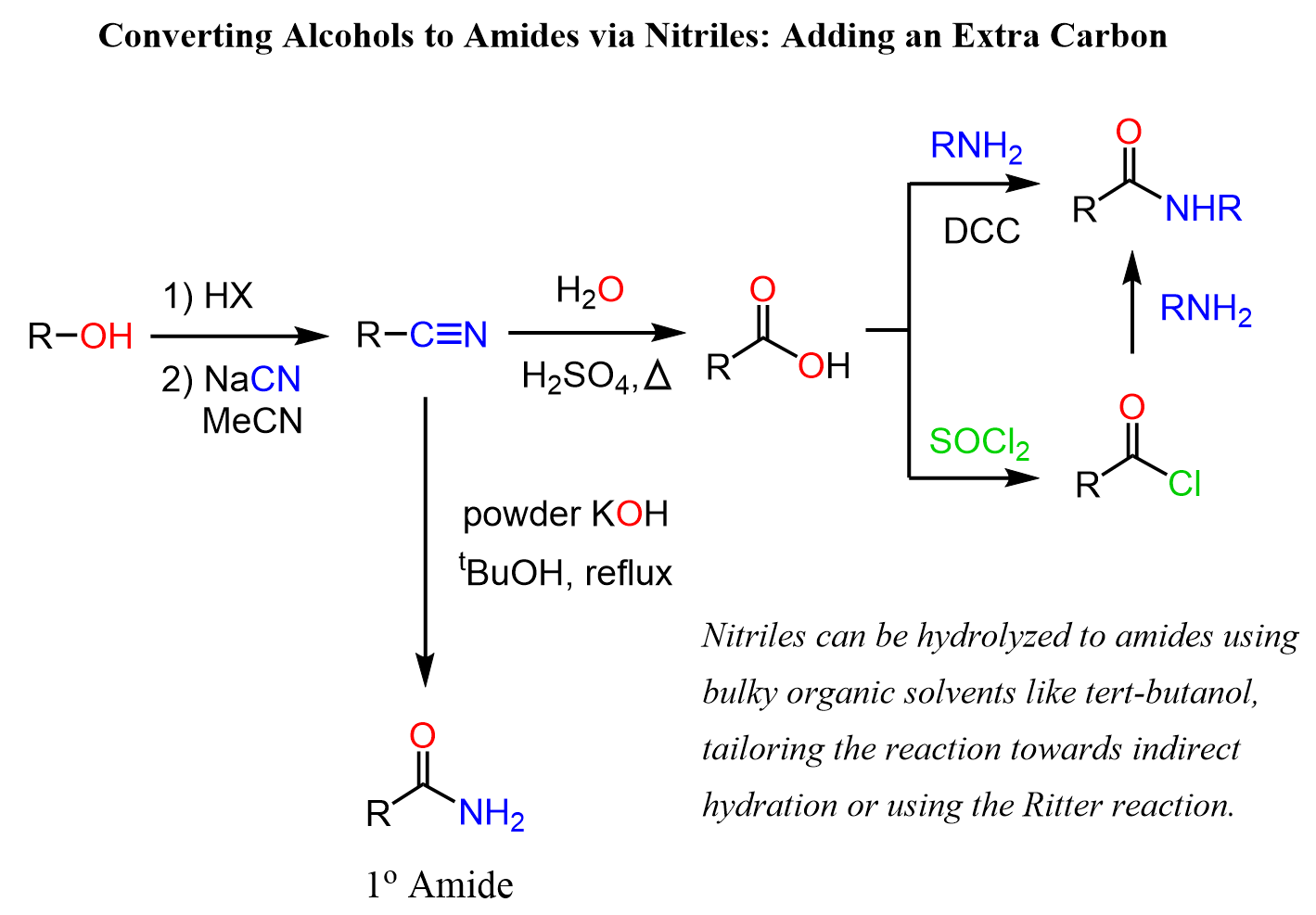
Branching down, we also have a direct conversion of the nitrile to an aldehyde. We know that nitriles are difficult to hydrolyze and harsh conditions of an acid and heat are needed to convert them to carboxylic acids. Therefore, the hydrolysis to amides is not as straightforward because it is difficult to stop the reaction at this stage.
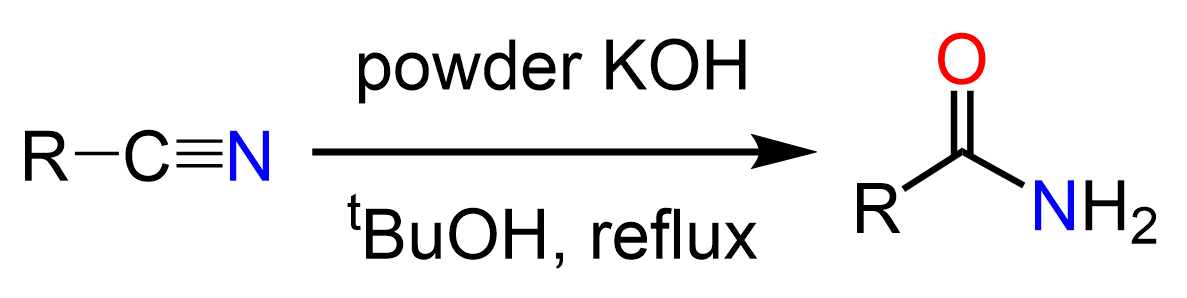
There are different ways of overcoming this challenge such as using bulky organic solvents like tert-butanol, tailoring the reaction towards indirect hydration, or using the Ritter reaction.
Check the post “Converting Nitriles to Amides” for more details.
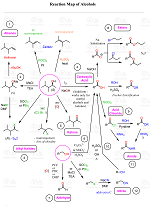
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Amides Hydrolysis: Acid and Base-Catalyzed Mechanism
- Amides Preparation and Reactions Summary
- Amides from Carboxylic Acids-DCC and EDC Coupling
- The Mechanism of Nitrile Hydrolysis To Carboxylic Acid
- Nitrile Reduction Mechanism with LiAlH4 and DIBAL to Amine or Aldehyde
- Nomenclature of Alcohols: Naming Alcohols based on IUPAC Rules with Practice Problems
- Preparation of Alcohols via Substitution or Addition Reactions
- Reaction of Alcohols with HCl, HBr and HI Acids
- Mesylates and Tosylates as Good Leaving Groups
- SOCl2 and PBr3 for Conversion of Alcohols to Alkyl Halides
- Alcohols in Substitution Reactions Practice Problems
- POCl3 for Dehydration of Alcohols
- Dehydration of Alcohols by E1 and E2 Elimination
- The Oxidation States of Organic Compounds
- LiAlH4 and NaBH4 Carbonyl Reduction Mechanism
- Alcohols from Carbonyl Reductions – Practice Problems
- Grignard Reaction in Preparing Alcohols with Practice Problems
- Grignard Reaction in Organic Synthesis with Practice Problems
- Protecting Groups For Alcohols in Organic Synthesis
- Oxidation of Alcohols: PCC, PDC, CrO3, DMP, Swern and All of That
- Diols: Nomenclature, Preparation, and Reactions
- NaIO4 Oxidative Cleavage of Diols
- The Pinacol Rearrangement
- The Williamson Ether Synthesis
- Alcohol Reactions Practice Problems
- Naming Thiols and Sulfides
- Reactions of Thiols
- Alcohols Quiz – Naming, Preparation, and Reactions