Let’s discuss an important and perhaps the most common way of converting aldehydes and ketones to amines, known as reductive amination. Before going into the details of reductive amination, let’s recall that amines are not formed by “simply” reacting aldehydes and ketones with amines. Depending on the amine, the product of these reactions can be an imine or an enamine:

Now, imines are structurally similar to carbonyls – we have a C=N instead of C=O, and we know that the carbonyl bond can be reduced in a number of ways. So, why not isolate the imine and reduce it to an amine? The problem is that many imines are not stable to be isolated, but those that are can certainly be reduced to the corresponding amines.
The next question is then how to make this transformation more universal without worrying about the instability of the imide. Can we reduce the imine as it forms without isolating it? The question was a hot topic in organic chemistry of 1960s, and it was Richard F. Borch at the University of Minnesota who discovered that sodium cyanoborohydride (NaBH3CN) can be used to prepare amines in situ by reacting aldehydes and ketones at ~ pH ≤ 7 levels (Borch, R. F.; Bernstein, M. D.; Durst, H. D. J. Am. Chem. Soc. 1971, 93, 2897).
All we need is to mix an amine with an aldehyde or ketone in the presence of sodium cyanoborohydride (NaBH3CN) at a lower pH:

One question you may be wondering is why we cannot use LiAlH4 or NaBH4. The problem with LiAlH4 is that it requires dry conditions, and NaBH4 hydrolyzes at lower pH as well. So, how is NaBH3CN different? The role of the CN group in NaBH3CN is to reduce the electron density and thus the reducing power through inductive and resonance withdrawing effects:
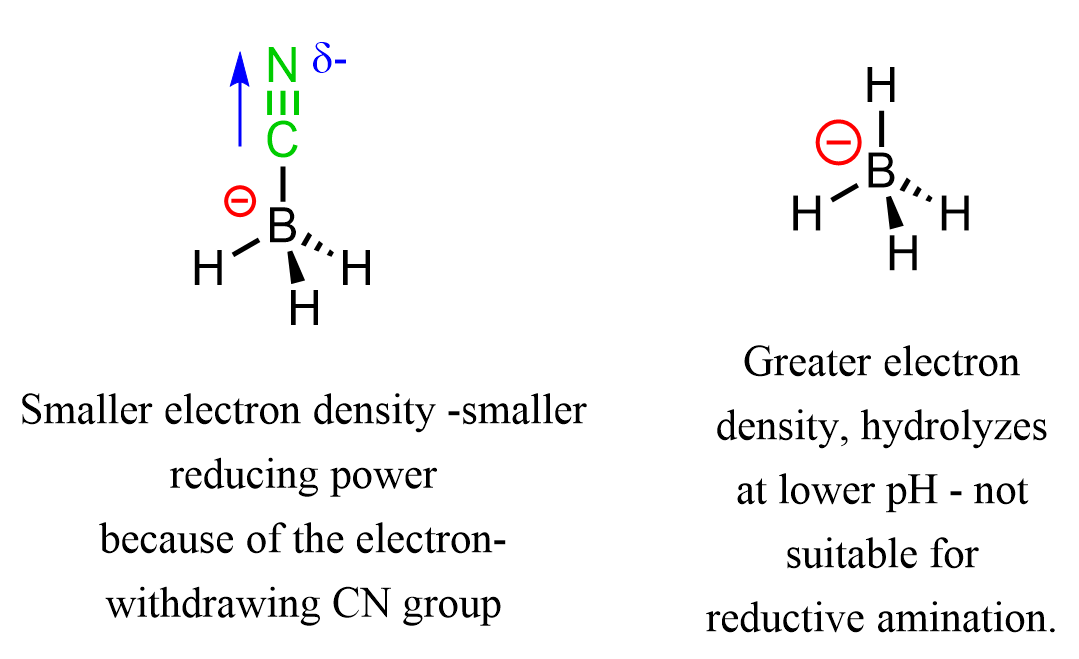
Another advantage of using NaBH3CN is that its reduction capabilities are pH dependent, and the conditions are adjusted such that the aldehyde and ketone are not reduced while the imine is. Overall, reductive amination using NaBH3CN allows for a one-pot synthesis without harsh conditions such as elevated pressure and temperature:

The Mechanism of Reductive Amination
The mechanism of reductive amination is not much different from what we have seen in the reduction of carbonyl compounds using NaBH4 and NaBH3CN. The carbon of the C=N bond has a partial positive charge because of the greater electronegativity of nitrogen, and this makes it an electrophilic center susceptible to nucleophilic attack by the hydride ion:

The last step, the reduction of iminium ion, is what we also see in the reduction of nitriles with LiAlH4.
Patterns in Reductive Amination
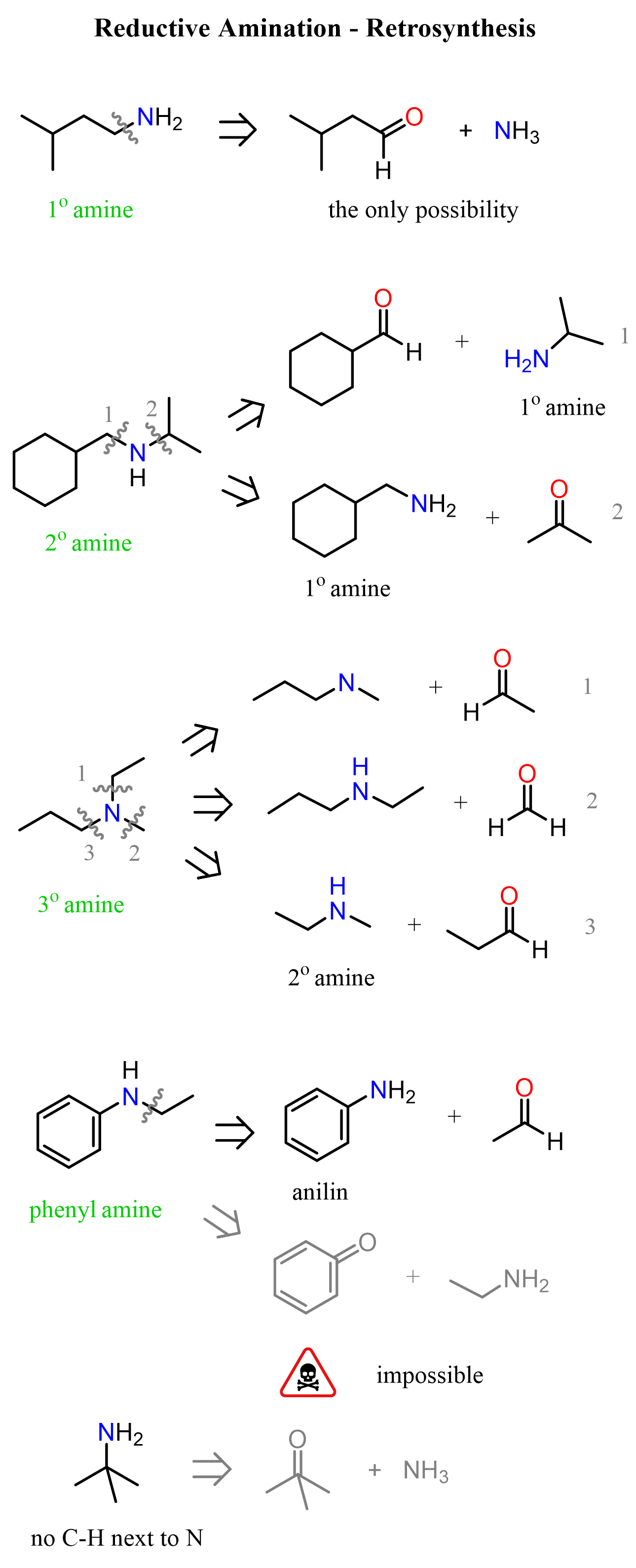
The main requirement for an amine to be used in reductive amination is the presence of a hydrogen on the nitrogen. This restricts the pool of amines to ammonia, primary, and secondary amines. Remember, primary amines give imines, and secondary amines give enamines, whereas tertiary amines do not undergo condensation with carbonyl compounds, and thus cannot be used in reductive amination.

Once again, notice that when a primary amine is used, the intermediate that undergoes reduction by NaBH3CN is the imine, whereas in the case of secondary amines, it is the iminium ion salt.
The only amine that would lead to a primary amine through reductive amination is ammonia. Most often, ammonium chloride or acetate is used as a source of ammonia. So, for any secondary and tertiary amine, we can retrosynthetically identify two pairs of amines and carbonyls as precursors:
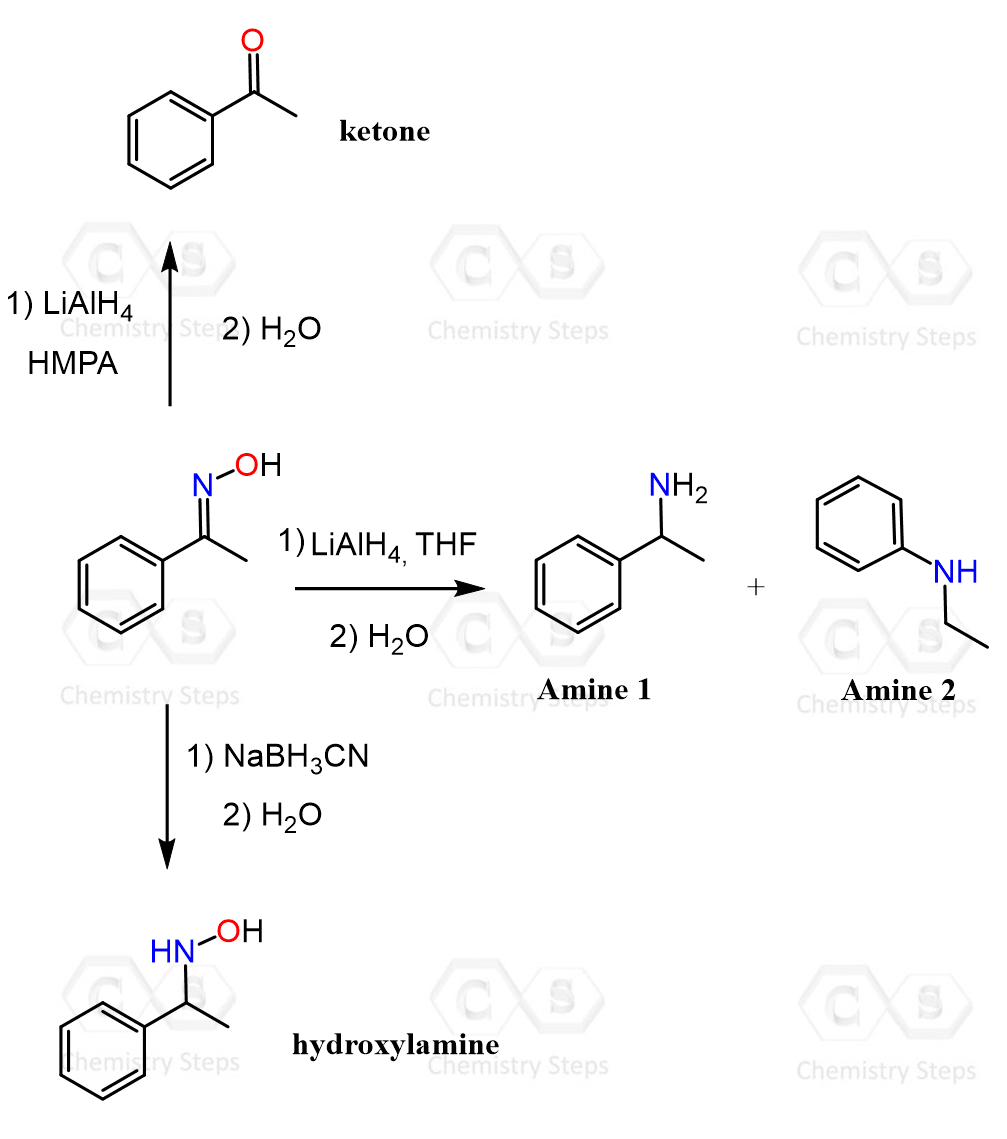
Amines via Reduction of Oximes
Another way of converting aldehydes and ketones to amines can be the preparation of the corresponding oximes, with their subsequent reduction using Na in ethanol or other reducing agents such as LiAlH₄ or catalytic hydrogenation. Oximes are easily formed by treating the carbonyl compound with hydroxylamine, and they are generally stable enough to be isolated and purified. The C=N–OH functionality can then be reduced to the amine, effectively replacing the carbonyl oxygen with an amino group.

This method is less commonly used than reductive amination because it requires an additional step to form the oxime; however, it provides a useful alternative when direct imine formation is difficult or when one wants to avoid issues related to imine stability.
These were the main points I wanted to include in this post on the conversion of aldehydes and ketones to amines. Let me know if you have questions or if there is anything you find that would be worth adding here.
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!