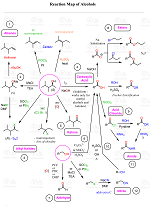
Alkenes to Aldehydes and Ketones via Ozonolysis
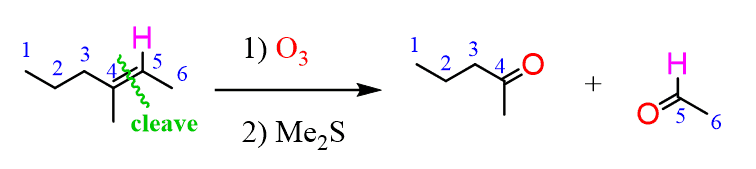
Alkenes can be converted to aldehydes or ketones via ozonolysis, which is an oxidative cleavage reaction converting the C=C bond to a C=O bond. Whether an aldehyde or a ketone is obtained depends on the structure of the alkene. If there is a hydrogen on any or both of the C=C carbons, an aldehyde is obtained:
One of the advantages of ozonolysis compared to other oxidative cleavage reactions is that it does not over-oxidize the alkene to carboxylic acid, unlike, for example, the potassium permanganate (KMnO4):

Even for terminal alkenes, it is possible to obtain formaldehyde instead of carbon dioxide, which is formed when strong oxidizing agents are used:

The same principle applies to cyclic compounds. Whenever you need to determine the product of an ozonolysis reaction, number the carbons (numbering carbons will help you a lot, no matter the exercises!) and replace the C=C double bonds with carbonyls:
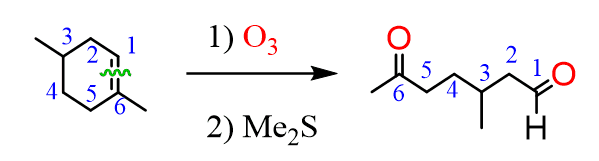
The mechanism and more examples of ozonolysis are covered in this article, as well as the multiple-choice quiz on the reactions of alkenes.
Alkenes to Aldehydes and Ketones Via Hydration and Oxidation
Another approach to convert an alkene to an aldehyde or a ketone is to oxidize the alcohol formed by the hydration of the alkene.

Notice that acid-catalyzed hydration of the alkenes adds the OH to the more substituted carbon (Markovnikov’s rule), while hydroboration-oxidation places the OH group on the less substituted carbon of an alkene.
The mechanism and the radiochemistry of these reactions are covered in the corresponding linked articles.
The primary alcohol formed via hydroboration-oxidation is converted to an aldehyde using mild reagents/conditions such as PCC, PDC, Dess-Martin, etc. The secondary alcohol can be oxidized to a ketone with either a strong or mild oxidizing agent since the ketone cannot be oxidized any further.
Below is the list of the strong and mild oxidizing agents, and you can read about them in more detail in the article “Oxidation of Alcohols”.

If you did not remember about the regioselective hydration of alkenes discussed above, you could also do a similar hydrohalogenation to obtain an alkyl halide, which then can be converted to an alcohol via SN2, followed by oxidation of the alcohol:

The first hydrohalogenation is according to Markovnikov’s rule, while the second is an anti-Markovnikov addition via Free-Radical Addition of HBr in the presence of a peroxide.
This involves an extra step for converting the alkene to an alcohol; however, it still gets to the desired transformation of the alkene to an aldehyde or a ketone.
More examples of alkene reactions are covered in the multiple-choice quiz and the practice problems after the corresponding articles.
Alkene Addition Reactions Practice Quiz
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Electrophilic Addition Reactions to Alkenes
- Markovnikov’s Rule
- Markovnikov’s Rule with Practice Problems
- Addition of Water to Alkenes
- Acid-Catalyzed Hydration of Alkenes with Practice Problems
- Rearrangements in Alkene Addition Reactions
- Oxymercuration-Demercuration
- Addition of Alcohols to Alkenes
- Free-Radical Addition of HBr: Anti-Markovnikov Addition
- Hydroboration-Oxidation: The Mechanism
- Hydroboration-Oxidation of Alkenes: Regiochemistry and Stereochemistry with Practice Problems
- Halogenation of Alkenes and Halohydrin Formation
- The Regiochemistry of Alkene Addition Reactions
- The Stereochemistry of Alkene Addition Reactions
- Cis product in an anti Addition Reaction of Alkenes
- Ozonolysis of Alkenes with Practice Problems
- Syn Dihydroxylation of Alkenes with KMnO4 and OsO4
- Anti Dihydroxylation of Alkenes with MCPBA and Other Peroxides with Practice Problems
- Oxidative Cleavage of Alkenes with KMno4 and O3
- Alkene Reactions Practice Problems
- Changing the Position of a Double Bond
- Changing the Position of a Leaving Group
- Alkenes Multi-Step Synthesis Practice Problems
- Alkene Addition Reactions Practice Quiz