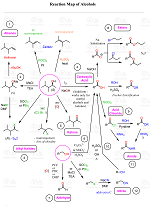
Let’s discuss a few strategies for converting alkenes to carboxylic acids that allow to increase the length of the carbon chain, decrease it, and keep it the same as well:

Alkenes to Carboxylic Acids: Keeping the Length of the Carbon Chain
To prepare a carboxylic acid from an alkene without changing the length of the carbon chain, we can first convert it to primary alcohol via anti-Markovnikov hydration, and then oxidize the alcohol to carboxylic acid:

Alkenes to Carboxylic Acids: Increasing the Length of the Carbon Chain
Many reactions you will see in Organic Chemistry 1 when the length of the carbon chain is increased, involve using the nitrile (CN) group. For example, here, we can convert the alkene to an alkyl halide, replace the halogen with a CN group via an SN2 substitution, and finally hydrolyze the nitrile to a carboxylic acid:

Notice that we can orient the position of the nitrile group by doing a Markovnikov or anti-Markovnikov hydrohalogenation of the alkene. Remember that the anti-Markovnikov hydrohalogenation works only with HBr in the presence of a radical catalyst.
Alkenes to Carboxylic Acids: Decreasing the Length of the Carbon Chain
Alkenes can also be converted to carboxylic acids by an oxidative cleavage of the double using ozonolysis or KMnO4:

Let’s also put together all these strategies for converting alkenes to carboxylic acids.

Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Electrophilic Addition Reactions to Alkenes
- Markovnikov’s Rule
- Markovnikov’s Rule with Practice Problems
- Addition of Water to Alkenes
- Acid-Catalyzed Hydration of Alkenes with Practice Problems
- Rearrangements in Alkene Addition Reactions
- Oxymercuration-Demercuration
- Addition of Alcohols to Alkenes
- Free-Radical Addition of HBr: Anti-Markovnikov Addition
- Hydroboration-Oxidation: The Mechanism
- Hydroboration-Oxidation of Alkenes: Regiochemistry and Stereochemistry with Practice Problems
- Halogenation of Alkenes and Halohydrin Formation
- The Regiochemistry of Alkene Addition Reactions
- The Stereochemistry of Alkene Addition Reactions
- Cis product in an anti Addition Reaction of Alkenes
- Ozonolysis of Alkenes with Practice Problems
- Syn Dihydroxylation of Alkenes with KMnO4 and OsO4
- Anti-Dihydroxylation of Alkenes with MCPBA and Other Peroxides with Practice Problems
- Oxidative Cleavage of Alkenes with KMno4 and O3
- Alkene Reactions Practice Problems
- Changing the Position of a Double Bond
- Changing the Position of a Leaving Group
- Alkenes Multi-Step Synthesis Practice Problems
- Alkene Addition Reactions Practice Quiz