Alkyl halides can be converted to alkenes via elimination reactions using strong and weak bases. These conversions are also known as dehydrohalogenation reactions:
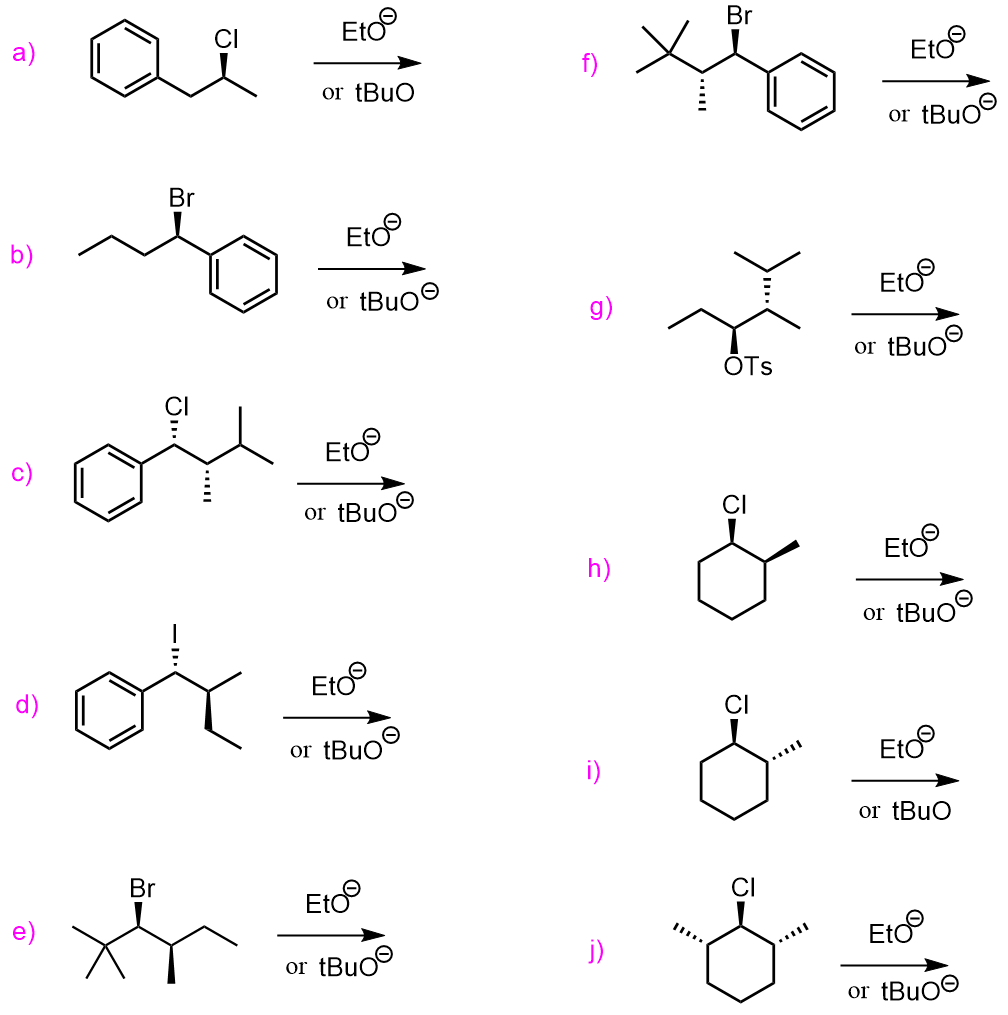
The elimination reactions are most often achieved by using strong bases, and there are several key factors such as the regio- and stereochemistry of these reactions that we need to address in this post. Therefore, let’s start with the eliminations using weak bases and then dive deeper into the examples of strong bases.
The E1 Elimination in Converting Alkyl Halides to Alkenes
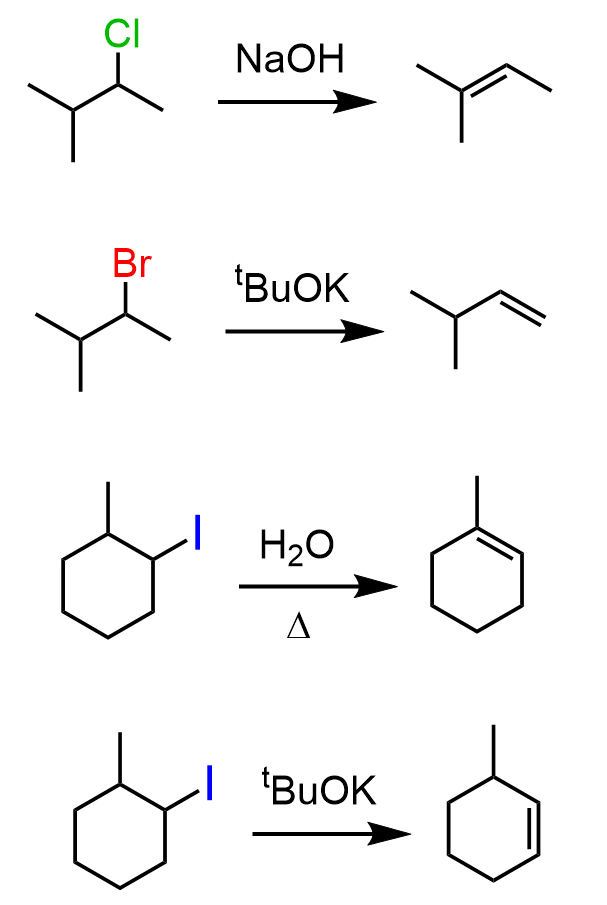
The most common weak bases you will encounter in elimination reactions of alkyl halides are water and alcohols. Recall that they are also poor nucleophiles and therefore, they only do SN1 and E1 reactions. Keep in mind also that the words “only”, “never”, and “always” are always associated with exceptions, so we are talking about the general patterns.

The competition between substitution and elimination reactions with weak bases is often determined by the temperature of the reaction. Recall that heat favors elimination so the higher the temperature, the more elimination product we’d normally get. One reason for this is the formation of an extra molecule in elimination reactions meaning that eliminations are entropically favored.
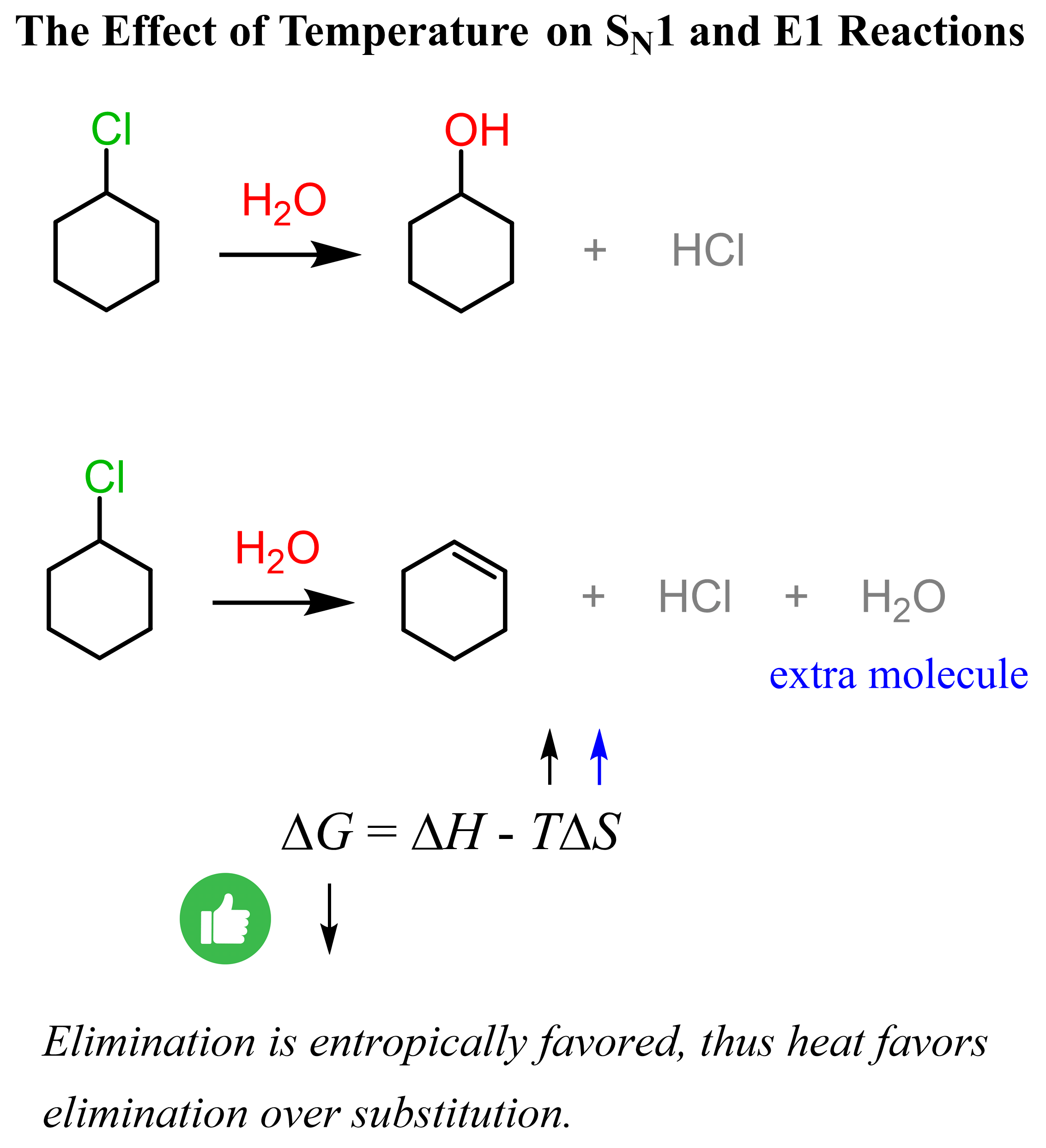
The extra molecule formed in the elimination reactions incenses the entropy, and the higher temperature makes it a larger (negative larger) factor, which decreases the Gibbs free energy of the reaction. Recall that negative ΔG indicates a spontaneous reaction, and in general, the smaller it is, the more favored the forward reaction. Check this article for more details about the effect of temperature on substitution and elimination reactions.
Another factor that favors elimination is the formation of substituted and conjugated alkenes (dienes). Recall that the stability of alkenes increases with the number of alkyl groups on the double bond, and conjugated systems are also associated with additional stabilization.
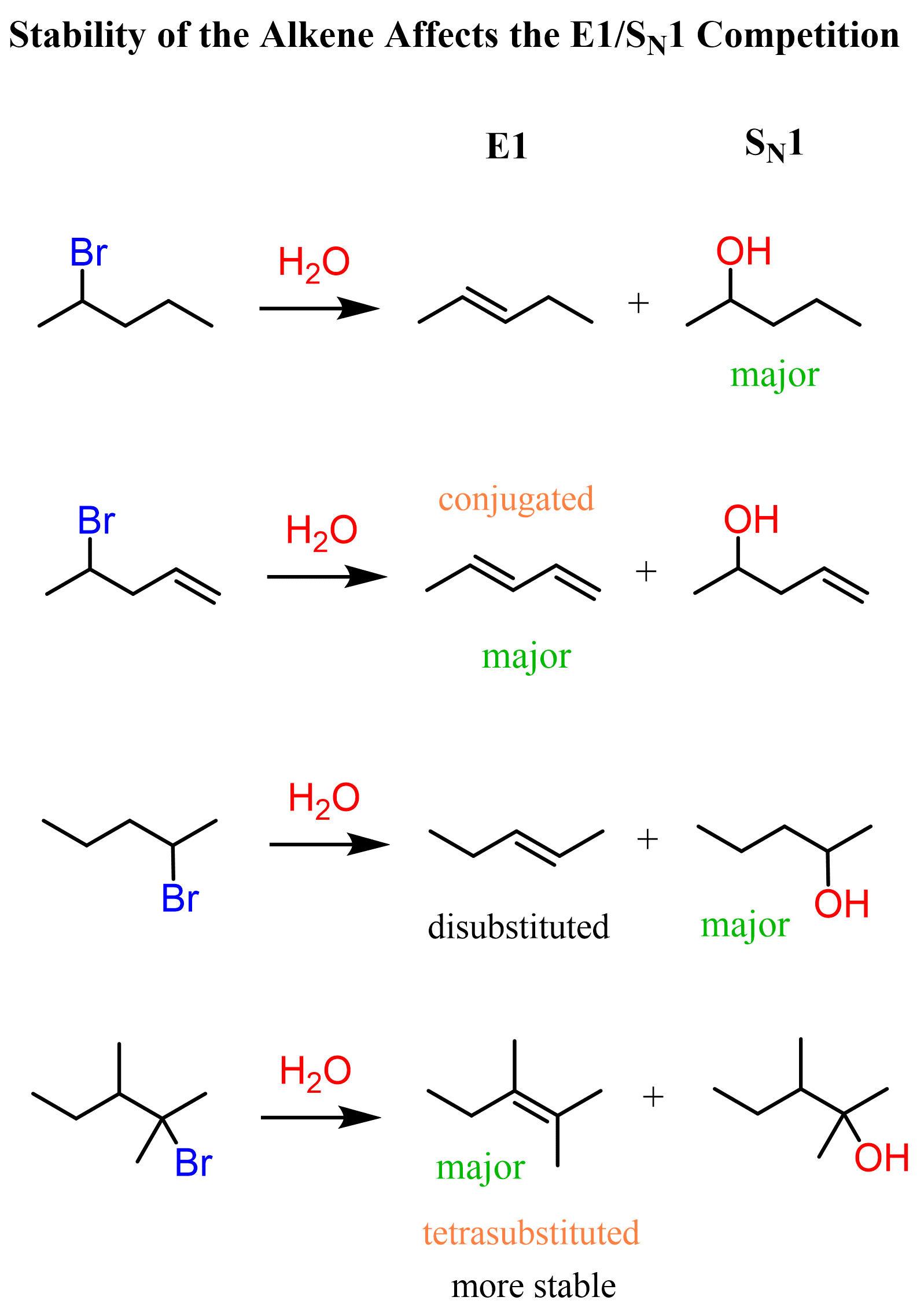
I want to mention that I did not search for experimental data to show the percentage of each product in these reactions. This is simply to explain what favors elimination and when we can expect it to be the major pathway of the reaction.
The Regiochemistry of E1 Elimination Reactions
E1 reactions are regioselective and just like in the E2 mechanism, the Zaitsev product predominates due to the higher stability of the more substituted alkene:

Alkyl Halides to Alkenes via E2 Elimination
The E2 elimination of alkyl halides is achieved by using strong bases such as hydroxides (–OH), alkoxides (RO–), organometallics, etc. Keep in mind that primary alkyl halides undergo SN2 substitution with –OH and RO– and other non-bulky bases (nucleophiles if it is a substitution reaction):
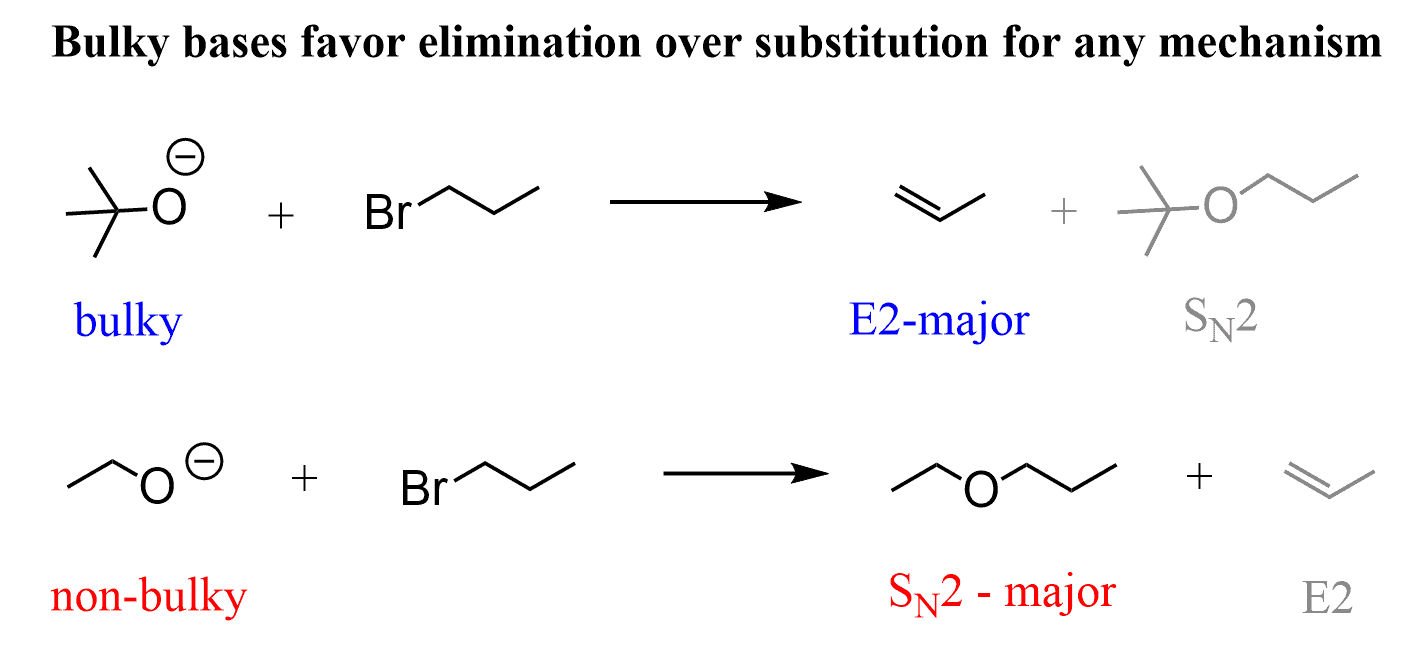
In general, remember that bases with charged atoms are strong bases and they favor the E2 mechanism:

Check this post for more details on deciding between SN1, SN2, E1, and E2 reactions.
The Regiochemistry of E2 Reactions
Unsymmetrical alkyl halides can be converted to alkenes with a more substituted double bond or less substituted double bond. Relatively small bases (sterically non-hindered or non-bulky) such as hydroxide ions favor the formation of more substituted alkenes. This is known as Zaitsev’s rule. On the contrary, bulky bases (sterically hindered) such as potassium tert-butoxide (tBuOK) favor the formation of less substituted alkenes – Hofmann elimination:
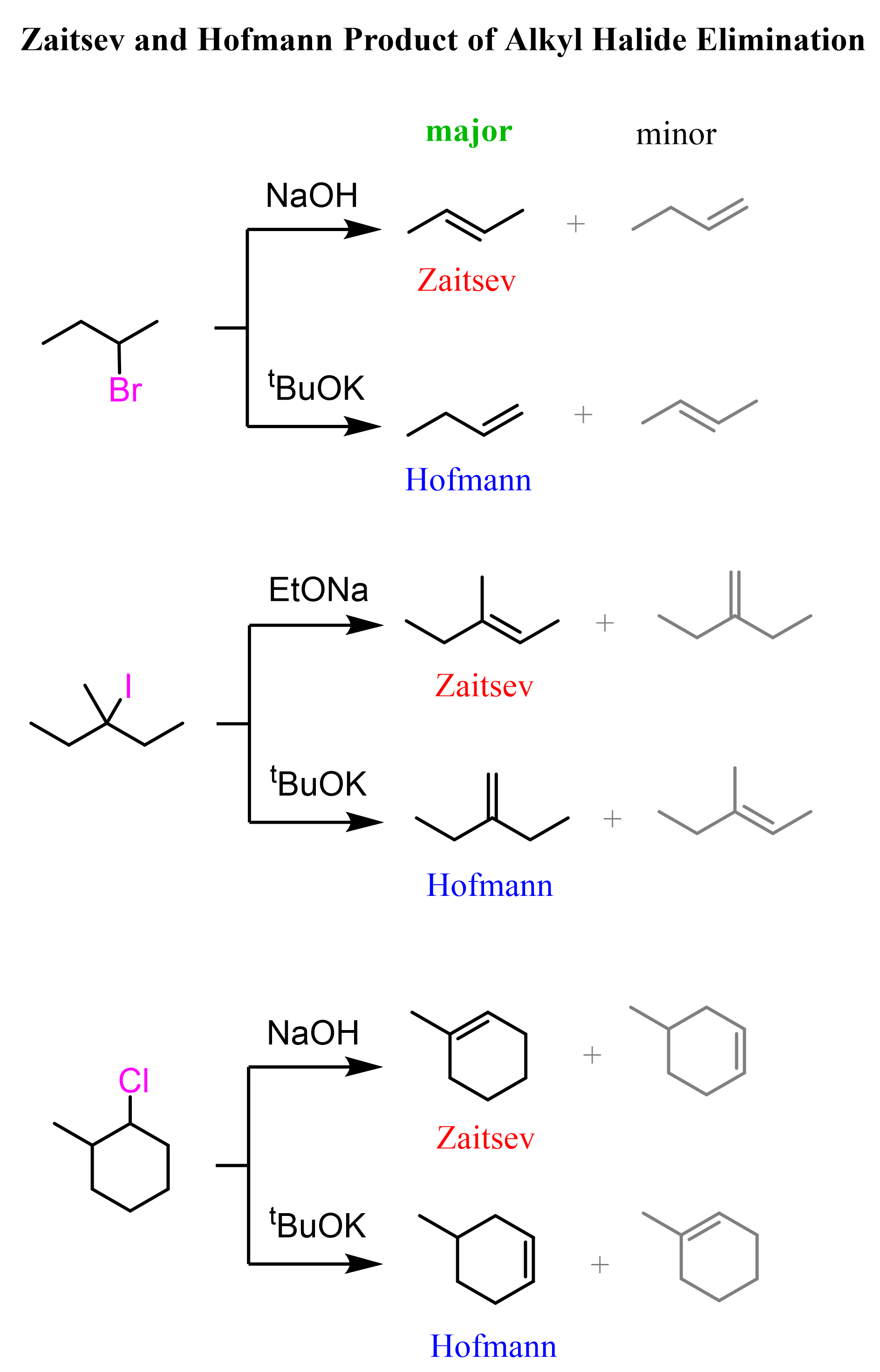
Cyclohexanes in Elimination Reactions
All the E1 and E2 reactions are examples of β elimination. The term β means the hydrogen is in the β position of the leaving group. There is an important requirement regarding the orientation of the β hydrogen and the leaving group that makes the E2 elimination possible: The β hydrogen must be anti-periplanar to the leaving group (parallel and at 180o).
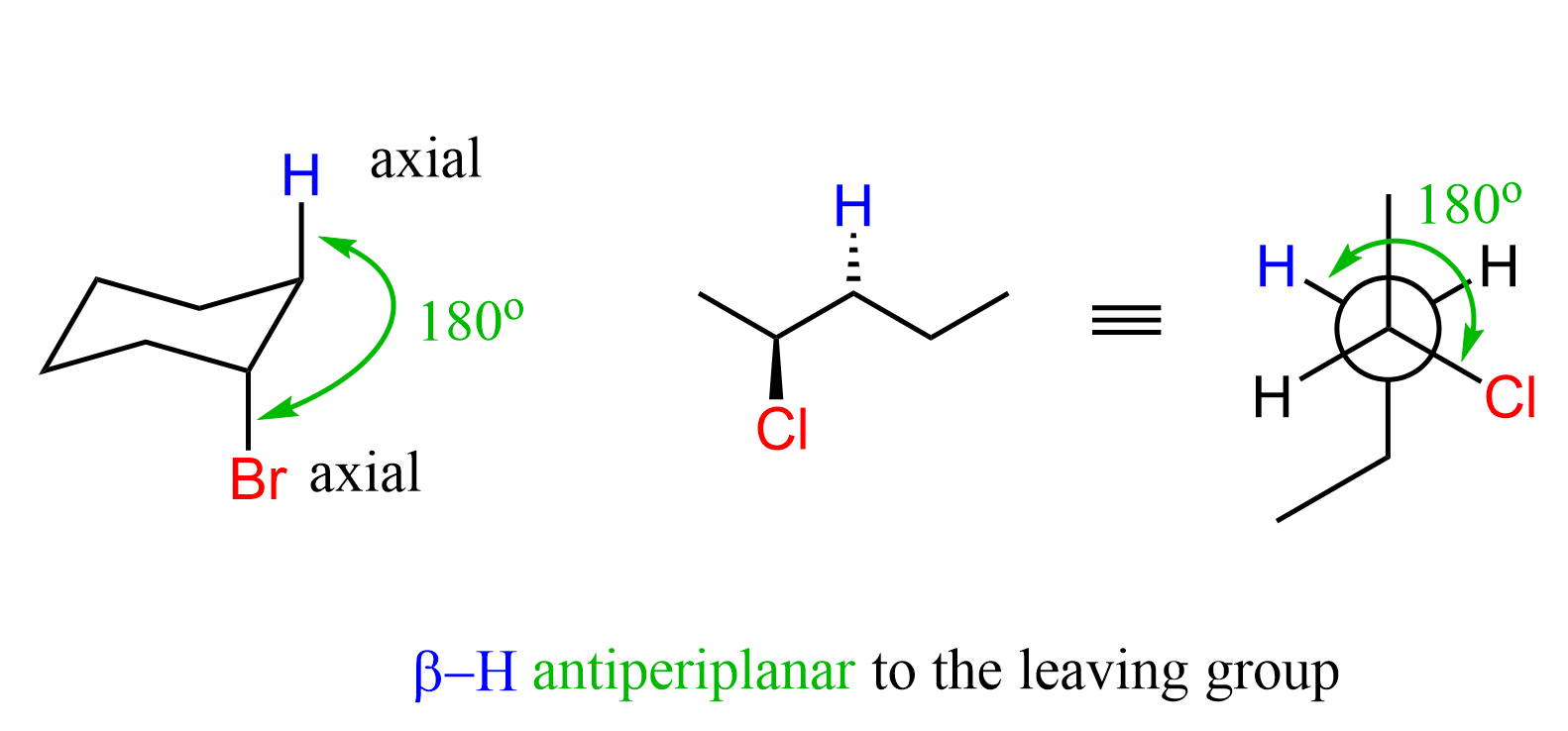
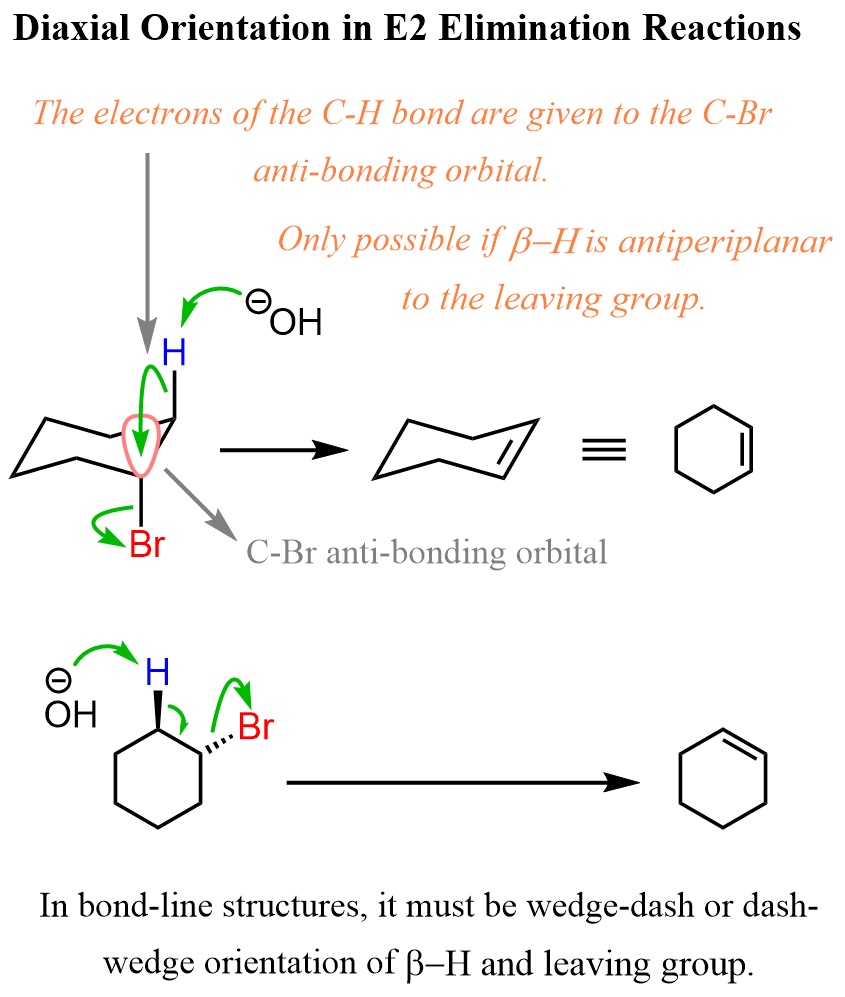
The reason for this is that when the β hydrogen is abstracted by the base, it gives the electrons of C-H bond to the antibonding orbital of the C-Halogen bond, and this is only possible when the orbitals are anti-periplanar:
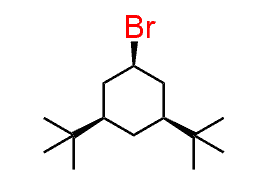
The significance of this is that sometimes only one alkene can be formed regardless if we use a bulky or non-bulky base. For example, the following alkyl halide, which is a substituted cyclohexane gives the Hofmann product even when treated with a nun-bulky base such as sodium ethoxide:
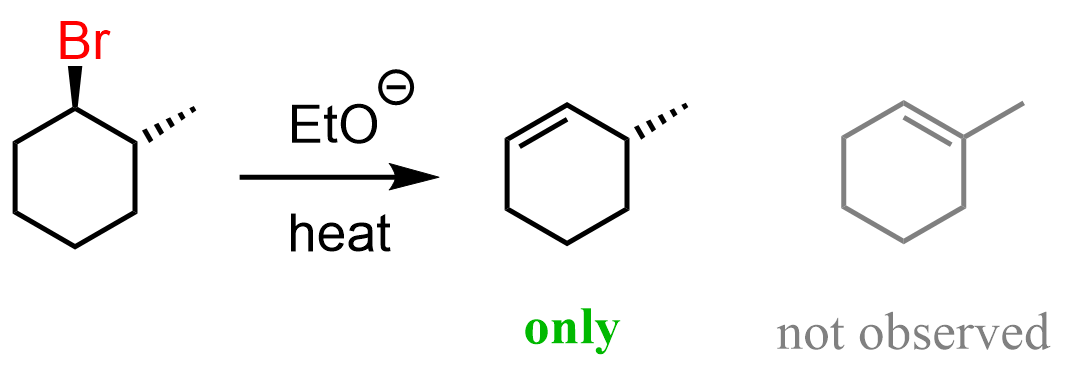
To understand why this happens, we need to convert the cyclohexane to a chair conformation. Importantly, out of the two chair conformations, it must be the one where the β hydrogen and the leaving group are axial. This is the only conformation that allows for the E2 elimination as the β hydrogen and the leaving group are anti-periplanar.

Notice that it is only the hydrogen on C6 that can be anti-periplanar to the Br, and therefore, the only product that can be obtained is the one with less substituted double bond.
We have a separate article on the elimination reactions of cyclohexanes, so feel free to check that out as well.
Stereoselectivity and Stereospecificity of Elimination Reactions
So far, we have been talking about the regioselectivity of elimination reactions. Very often, the product of the elimination can be a cis or a trans alkene (E or Z in a broader definition), and, if possible, the trans alkene is the major stereoisomer because of its higher stability:
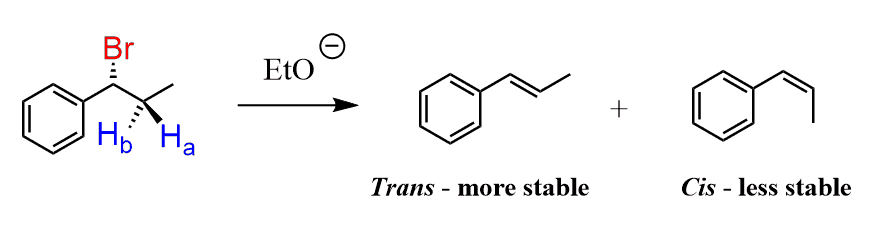
Let’s also understand we use the term “if possible”. When the alkyl halide has two β hydrogens, it adopts a conformation that forms the more stable alkene. We say the reaction is stereoselective: the substrate “selects” which hydrogen participates in the elimination to form the more stable product.
If the alkyl halide has only one β hydrogen, there can only be one conformation when it is anti-periplanar to the leaving group, thus only one stereoisomer can be formed which may or may not be the most stable. This is an example of a stereospecific reaction: the substrate “has no choice” and the stereochemistry of the reaction is determined by the stereochemistry of the substrate.

You can read about the stereospecificity of E2 reactions here, but as a shortcut, remember that the alkene is formed by eliminating a wedge β hydrogen with a dash leaving group or a dash β hydrogen with a wedge leaving group. If there are two beta hydrogens, you can draw the structure of the alkene based on the zig-zag geometry of the alkyl halide – the more stable alkene is formed. If there is only one beta hydrogen, and it is not anti to the leaving group, you need to rotate the zig-zag to get the only possible alkene.

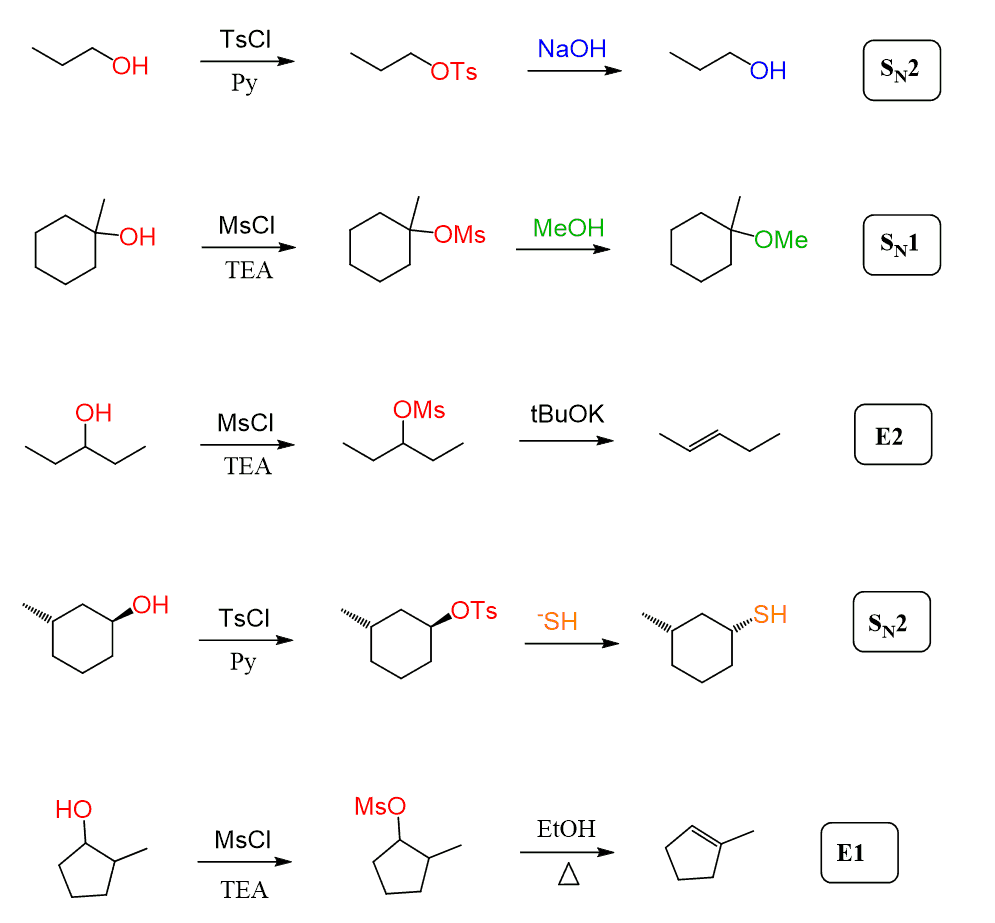
The elimination reactions are a comprehensive chapter, and we cannot address all the features in one post. However, this was a summary of how alkyl halides are converted to alkenes by E1 and E2 eliminations. Keep in mind that other substrates with good leaving groups such as mesylates and tosylates follow the same patterns, so do not be confused when seeing OMs and OTs notation in substitution and elimination reactions.
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!