We know that the hydrohalogenation of alkynes produces a vinyl halide which is an alkene that is converted into a geminal dihalide upon the second electrophilic addition of HX:

So, how can we convert the alkyne into an alkyl halide?
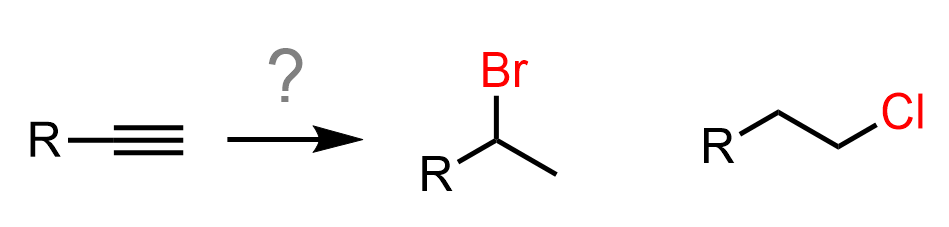
There is no direct way of achieving this, so what we can do is reduce the alkyne to an alkene and hydrohalogenate the alkene by Markovnikov and anti-Markovnikov addition:
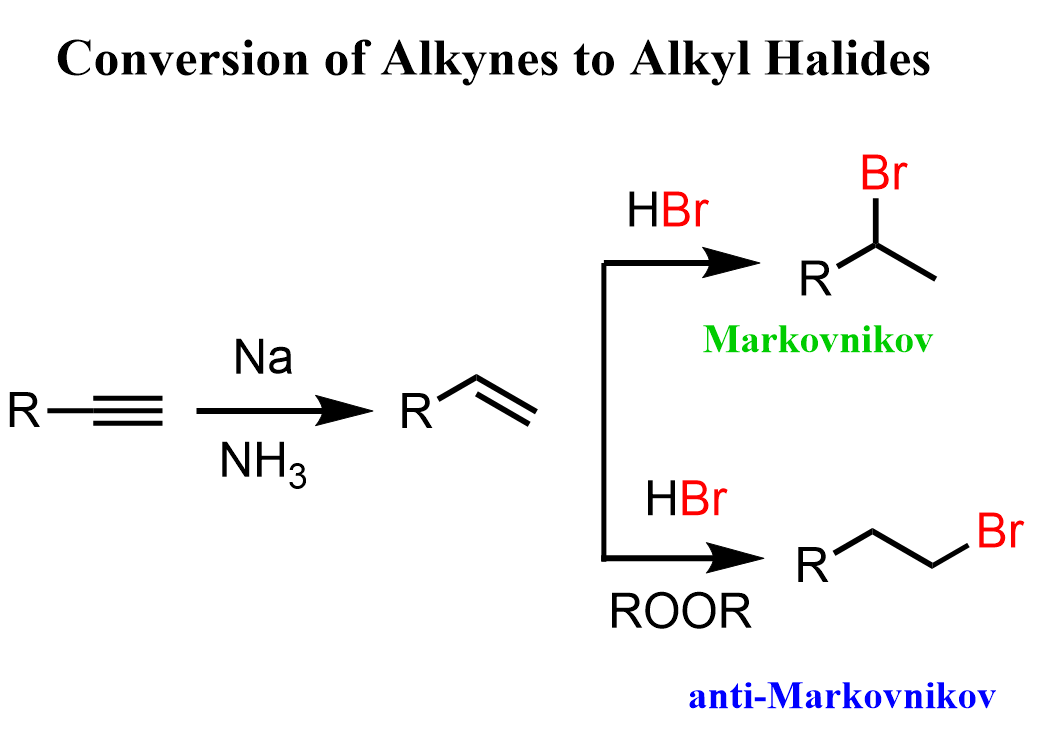
Notice that the anti-Markovnikov hydrohalogenation is possible only with HBr in the presence of a peroxide catalyst.
Alternatively, and this is a longer route, however maybe helpful in recognizing some patterns and solving practice problems, we can convert the alkyne into an alcohol, and then replace the OH with the needed halogen:
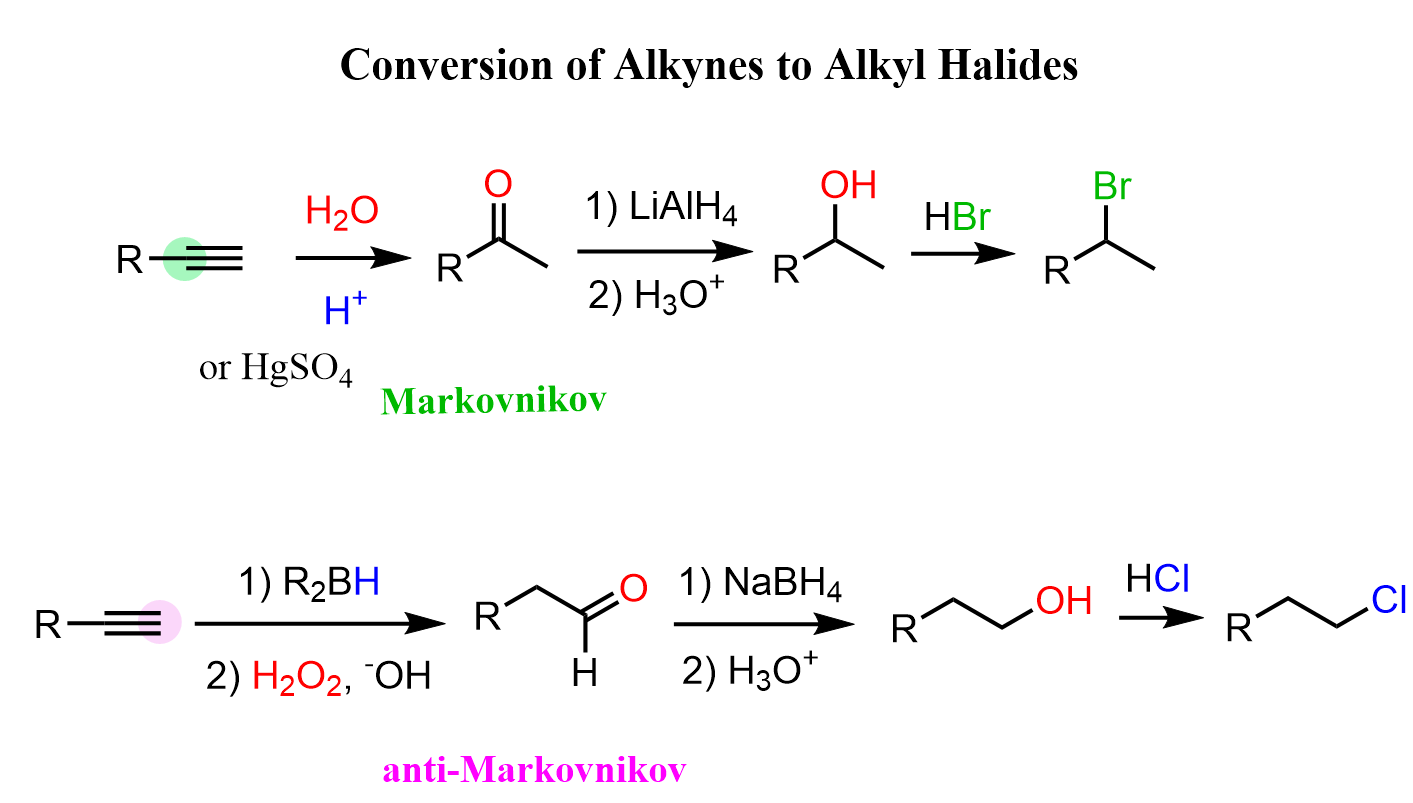
The first step is the hydration of the alkyne which can be a Markovnikov or anti-Markovnikov addition of water. This forms the corresponding carbonyl compound because the intermediate enol is unstable and tautomerizes to the corresponding aldehyde or ketone.

In the next step, we can reduce the aldehyde or the ketone with NaBH4, LiAlH4, catalytic hydrogenation, etc.
The conversion of the alcohol can be done in different ways such as the reaction with HX acid, using SOCl2 or PBr3, or converting the OH into a mesylate or tosylate first if, for example, there is a risk of a rearrangement in the reaction with acids. Check the linked posts for more details about each of these methods for converting alcohols to alkyl halides.
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Introduction to Alkynes
- Naming Alkynes by IUPAC Nomenclature Rules – Practice Problems
- Preparation of Alkynes by Elimination Reactions
- Hydrohalogenation of Alkynes
- Acid Catalyzed Hydration of Alkynes with Practice Problems
- Reduction of Alkynes
- Halogenation of Alkynes
- Hydroboration-Oxidation of Alkynes with Practice Problems
- Ozonolysis of Alkynes with Practice Problems
- Alkylation of Terminal Alkynes in Organic Synthesis with Practice Problems
- Alkyne reactions summary practice problems
- Alkyne Synthesis Reactions Practice Problems
- Alkyne Naming and Reactions Practice Quiz