We have seen that alcohols can be converted to amines by converting the OH into a good leaving group and reacting the substrate with an amine:

The reverse process is more challenging because NH2 is even a worse leaving group than OH, and there are no straightforward ways of converting it into a good leaving group and performing a substitution. So, for starters, let’s leave this here:

It is true that there are some literature data for direct conversion of amines to alcohols at > 200 oC ( Org. Lett. 2000, 2, 18, 2893–2895), but our discussion is for undergraduate educational purposes, and besides, not every molecule is going to survive 200 oC in basic conditions.
We will discuss two approaches to converting amines to alcohols: 1) Diazotization – converting to a diazonium salt, 2) Converting the amine to a quaternary ammonium salt.

Amines to Alcohols via Diazonium Salts
Diazonium salts have a general formula of R-N2+, and they undergo various reactions because the leaving group here is nitrogen gas. Diazonium salts are prepared by reacting the amine with NaNO2 and HCl which produces HNO2 and subsequently +NO electrophile:

Arene diazonium salts (Ar-N2+) are by far the most common diazonium salts because they are the most stable, while primary aliphatic diazonium salts spontaneously react with water giving a variety of substitution and elimination reactions.
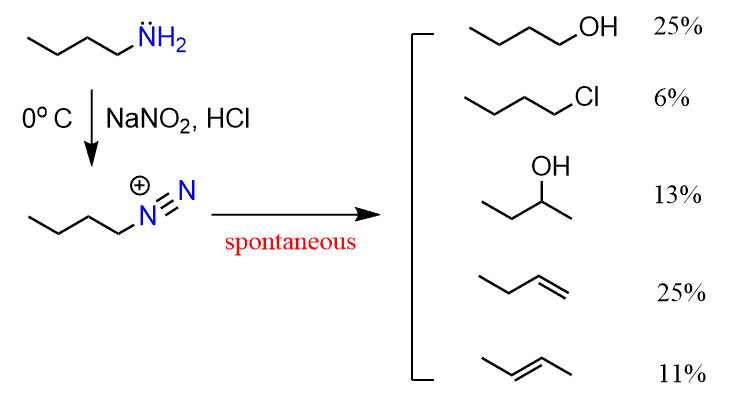
To convert the amine to an alcohol, this may not be the worst option, however, there is little control over the course of the reaction and the yields are not going to be great either.
Arene diazonium salts, on the other hand, are relatively stable and they can be converted into a different functional group in a controlled manner. Acidic or basic hydrolysis can be used to replace the N2 group with an OH thus we can use this method to prepare phenols from aniline derivatives:

It is worth mentioning that secondary amines with nitrous acid stop at N-nitrosamines (R2NN=O) and diazonium salts are not obtained.
Amines to Alcohols via Elimination and Hydration
The amino group can also be converted into a good leaving group by reacting methyl iodide. The product of this reaction is a quaternary ammonium salt and it is a good leaving group because the nitrogen is positively charged.

When heated with an aqueous solution of Ag2O, an E2 elimination occurs forming the Hofmann product – that is the less substituted alkenes:

What happens is –OH is formed in situ and it attacks the less hindered beta hydrogen producing the corresponding alkene. Unfortunately for today’s purpose, SN2 substitution still does not occur because of the steric hindrance associated with the size of the ammonium group.
So, if the alkene is unsymmetrical, we need to hydrate it in a Markovnikov or anti-Markovnikov fashion.
For example, let’s convert the following amine into an alcohol via Hofmann elimination:

In the first part of the reaction, we are going to convert the amino group into a quaternary ammonium salt, and obtain the terminal alkene upon heating the salt with Ag2O solution:

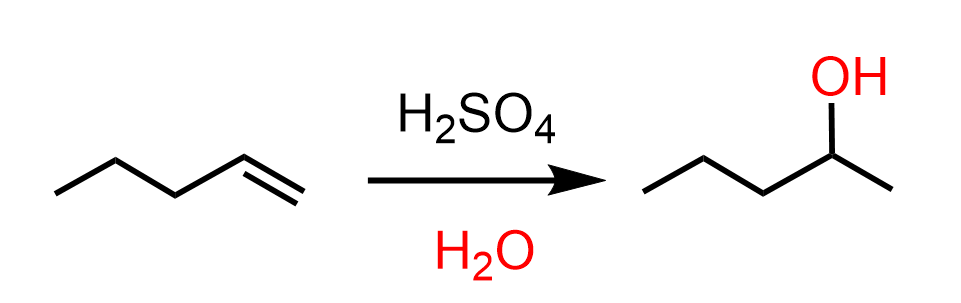
At this point, we need to use a hydration of alkene that follows Markovnikov’s rule. We can do this by reacting it with dilute sulfuric acid or oxymercuration-demercuration if there is a risk of a rearrangement:
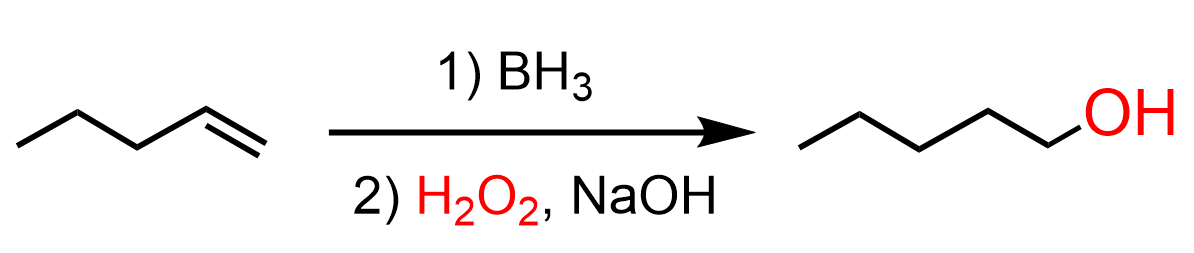
If it was a primary amine, then in the last step, we’d use hydroboration-oxidation to add the OH to the anti-Markovnikov position of the double bond:
Let’s summarize all the steps of these two reactions in one synthetic scheme for conversion of amines to alcohols:

This is what we had to share about converting amines to alcohols. Let me know in the comments if there are any methods that would work along with the undergraduate coursework of organic chemistry.
One approach that also may work is converting the amine into an imine via oxidation, then hydrolyzing the imine to the corresponding carbonyl followed by a reduction to alcohol.

The problem here would be finding a suitable way of oxidizing the amine to imine, preventing the condensation of the two amines (https://doi.org/10.1002/chem.201803251).
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Naming Amines: Systematic and Common Nomenclature
- Preparation of Amines
- The Gabriel Synthesis of Primary Amines
- Imines from Aldehydes and Ketones with Primary Amines
- Enamines from Aldehydes and Ketones with Secondary Amines
- The Hofmann Elimination of Amines and Alkyl Fluorides
- The Reaction of Amines with Nitrous Acid
- Reactions of Amines Practice Problems
- The Cope elimination
- Basicity of Amines
- Boc Protecting Group for Amines