There are direct ways of converting alkenes to amines covered in undergraduate organic chemistry textbooks, so a question like this is most likely meant to test your knowledge on multiple functional group transformations.

The strategy here is to convert the alkene to an alcohol or an alkyl halide and then introduce an azide group via SN2 substitution which can then be reduced to the corresponding amine. We can convert the alkene to an alcohol via Markovnikov and anti-Markovnikov addition of water. If the alkene is susceptible to a rearrangement, you can use the oxymercuration-demercuration reaction.

Notice that the alcohol needs to be first converted into a good leaving group such as a mesylate and tosylate which then can be substituted by an azide salt in a polar aprotic salt.
We can also convert the alkene to an alkyl halide and react the latter with an azide.
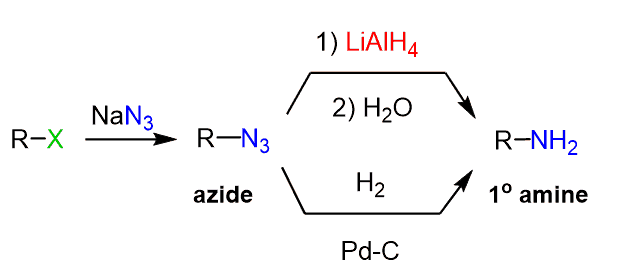
The problem of a rearrangement is still addressed by converting the alkene to an alcohol via oxymercuration-demercuration:

The alkyl halides can also be reacted with a cyanide salt to prepare the corresponding nitrile and then reduce it an amine:
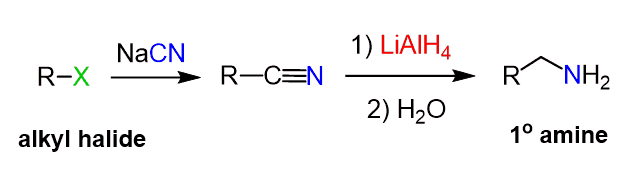
Notice that in this case, an extra carbon is added to the chain which comes from the cyanide ion (–CN).
You may be wondering why are we not reacting the mesylate or the alkyl halide with ammonia or another amine to prepare a new or another amine.

This can be done and nothing conceptually wrong with it. However, the problem is that the amine formed by nucleophilic substitution still has a lone pair of electrons, thus it can serve as a nucleophile or a base reacting with the alkyl halide. This gives a mixture of 1°, 2°, and 3° amines which limits its application.

In addition to the substitution, the elimination reactions become an issue, too, as the alkyl groups keep adding, resulting in secondary and especially tertiary amines.
The Ritter Reaction for Converting Alkenes to Amines
This is the third reaction we have in the first image, and it is the Ritter reaction which is especially useful for preparing tertiary amines.
Although the Ritter reaction is not covered in many textbooks, it is a great combination of principles and reactions such as carbocations, loss of a leaving group, nucleophilic addition, and hydrolysis, so you should be good to use it for a variety of functional group transformations without violating what you are allowed to use in your assignments.
The Ritter reaction can be used for preparing amines from alkenes, alkyl halides, and alcohols. All of these are used as the source of carbocation that is attacked by a nitrile, followed by hydrolysis to obtain an amide that can further be hydrolyzed into the needed amine.
We only need a precursor to generate a carbocation, which is reacted with a nitrile nucleophile:
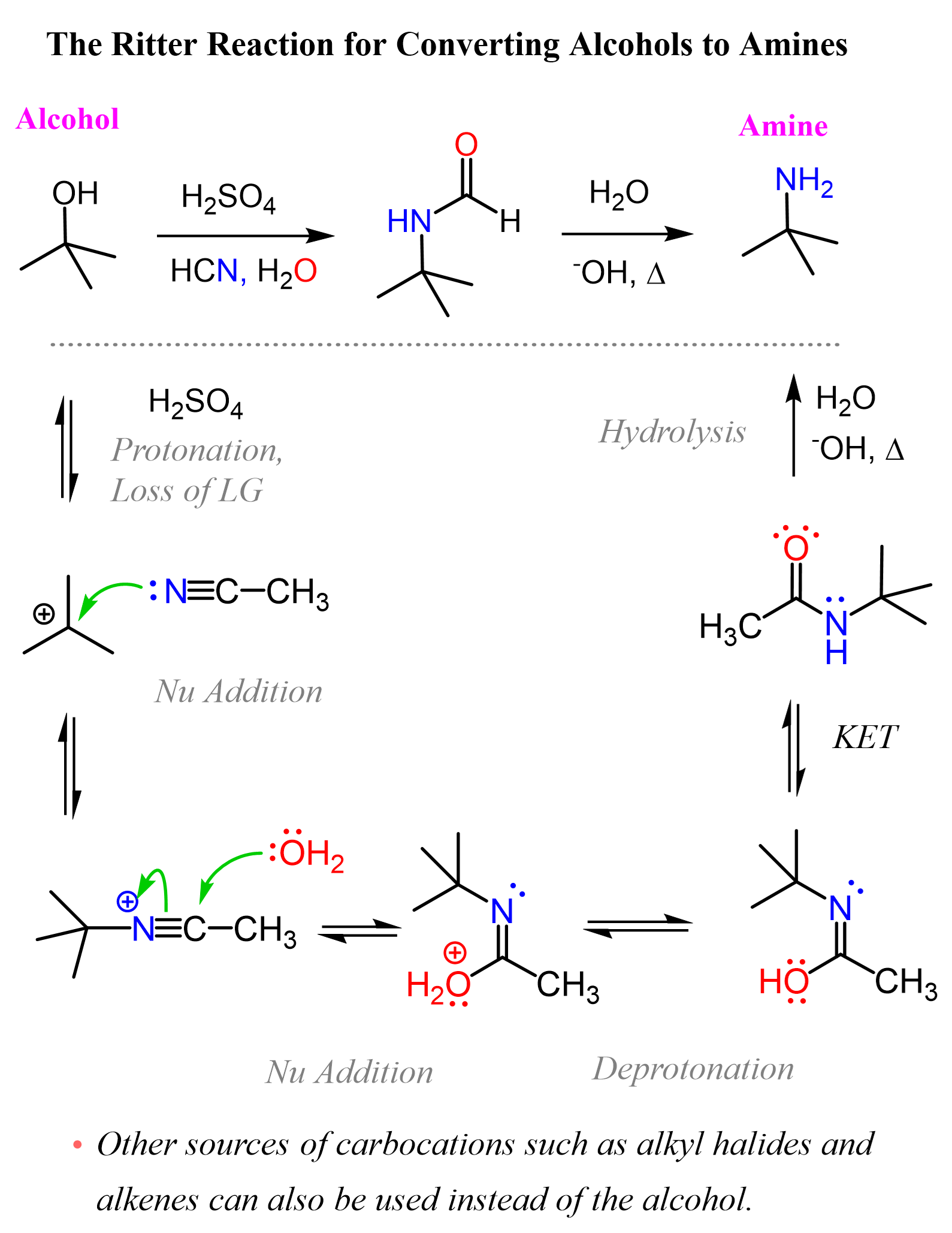
A quick note here: if you are in the first semester of Organic Chemistry and you have not covered keto-enol tautomerization, the reactions of nitriles and amides such as their hydrolysis, then do not worry about understanding what is going on in this transformation.
Check the article “Preparation of Amines” for more details about the Ritter reaction.
Hydroamination
In some ways, this is not a conversion of an alkene to an amine since an amine is a reagent, and you will most likely be asked to convert the alkene to a primary amine. However, it is a well-known and applicable reaction for many synthetic purposes.
Hydroamination is definitely outside of the curriculum in most undergraduate courses, so do not show it as a solution unless previously discussed with your instructor. It is transition and lanthanide metal catalyzed coupling of C and N atoms of an alkene/alkyne and amine to prepare a new amine.

Hydroamination mostly does Markovnikov addition of amines to alkenes and alkynes, and as mentioned in the article cited below, quote “The catalytic anti-Markovnikov addition of H-NR2 to olefins was listed as one of the so-called “Ten Challenges for Catalysis””.
There are an endless number of metal-catalyzed coupling reactions that defeat the purpose of undergraduate educational organic chemistry, so I am adding this only as an FYI.
Here is a nice review on all types of details in hydroamination reactions, so I will add that here too: Chem. Rev. 2008, 108, 3795–3892
Check Also
- Electrophilic Addition Reactions to Alkenes
- Markovnikov’s Rule
- Markovnikov’s Rule with Practice Problems
- Addition of Water to Alkenes
- Acid-Catalyzed Hydration of Alkenes with Practice Problems
- Rearrangements in Alkene Addition Reactions
- Oxymercuration-Demercuration
- Addition of Alcohols to Alkenes
- Free-Radical Addition of HBr: Anti-Markovnikov Addition
- Hydroboration-Oxidation: The Mechanism
- Hydroboration-Oxidation of Alkenes: Regiochemistry and Stereochemistry with Practice Problems
- Halogenation of Alkenes and Halohydrin Formation
- The Regiochemistry of Alkene Addition Reactions
- The Stereochemistry of Alkene Addition Reactions
- Cis product in an anti Addition Reaction of Alkenes
- Ozonolysis of Alkenes with Practice Problems
- Syn Dihydroxylation of Alkenes with KMnO4 and OsO4
- Anti Dihydroxylation of Alkenes with MCPBA and Other Peroxides with Practice Problems
- Oxidative Cleavage of Alkenes with KMno4 and O3
- Alkene Reactions Practice Problems
- Changing the Position of a Double Bond
- Changing the Position of a Leaving Group
- Alkenes Multi-Step Synthesis Practice Problems
- Alkene Addition Reactions Practice Quiz
