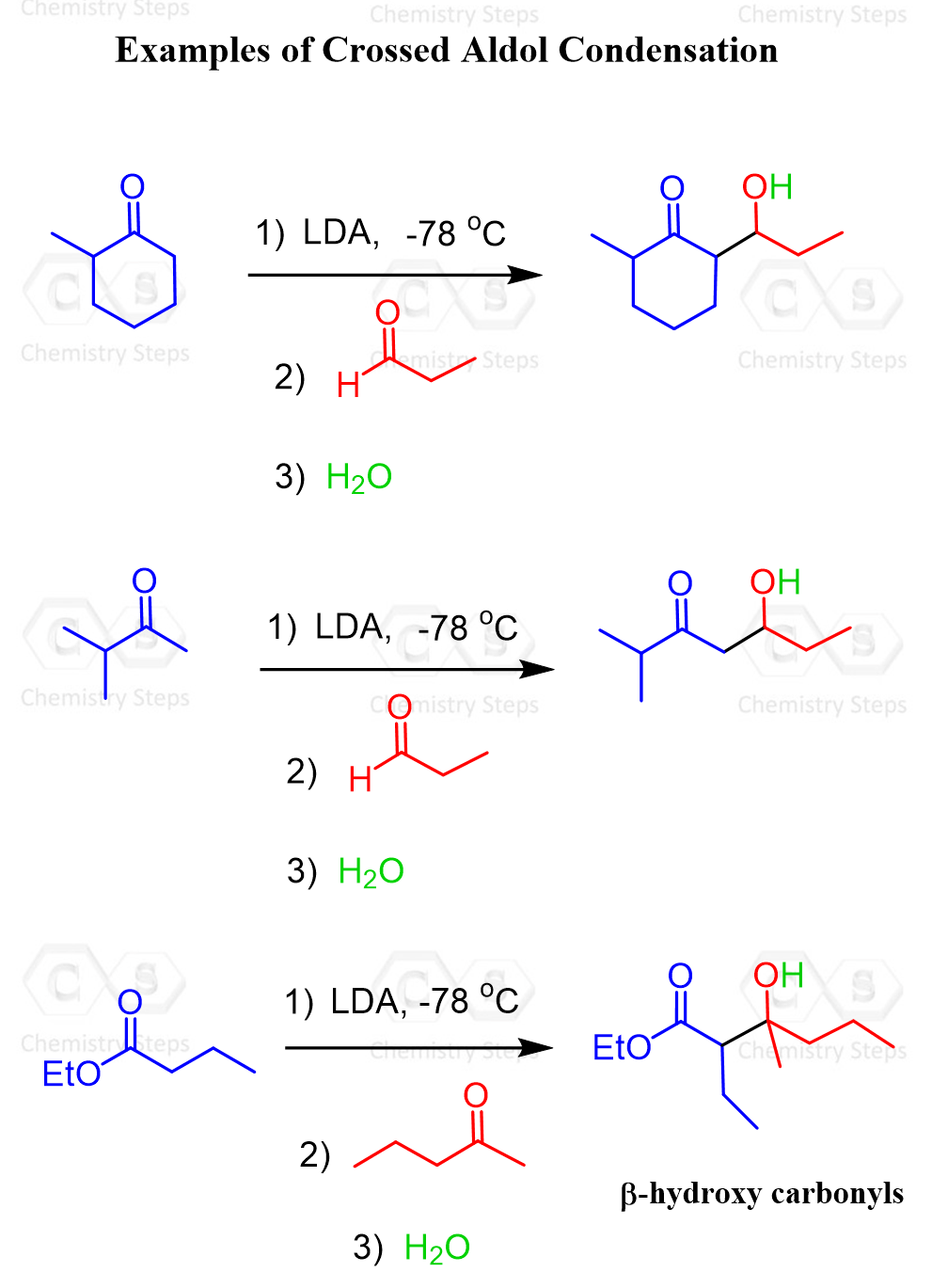
We have seen in the previous post that crossed aldol reactions allow us to prepare β-hydroxy carbonyl compounds by reacting an enolate (formed from one carbonyl compound) with another aldehyde or ketone, followed by protonation.
A nice variation/type of the crossed aldol condensation is the Reformatsky reaction, where an α-halo ester is reacted with different carbonyl compounds, such as aldehydes and ketones, to form a β-hydroxy ester.
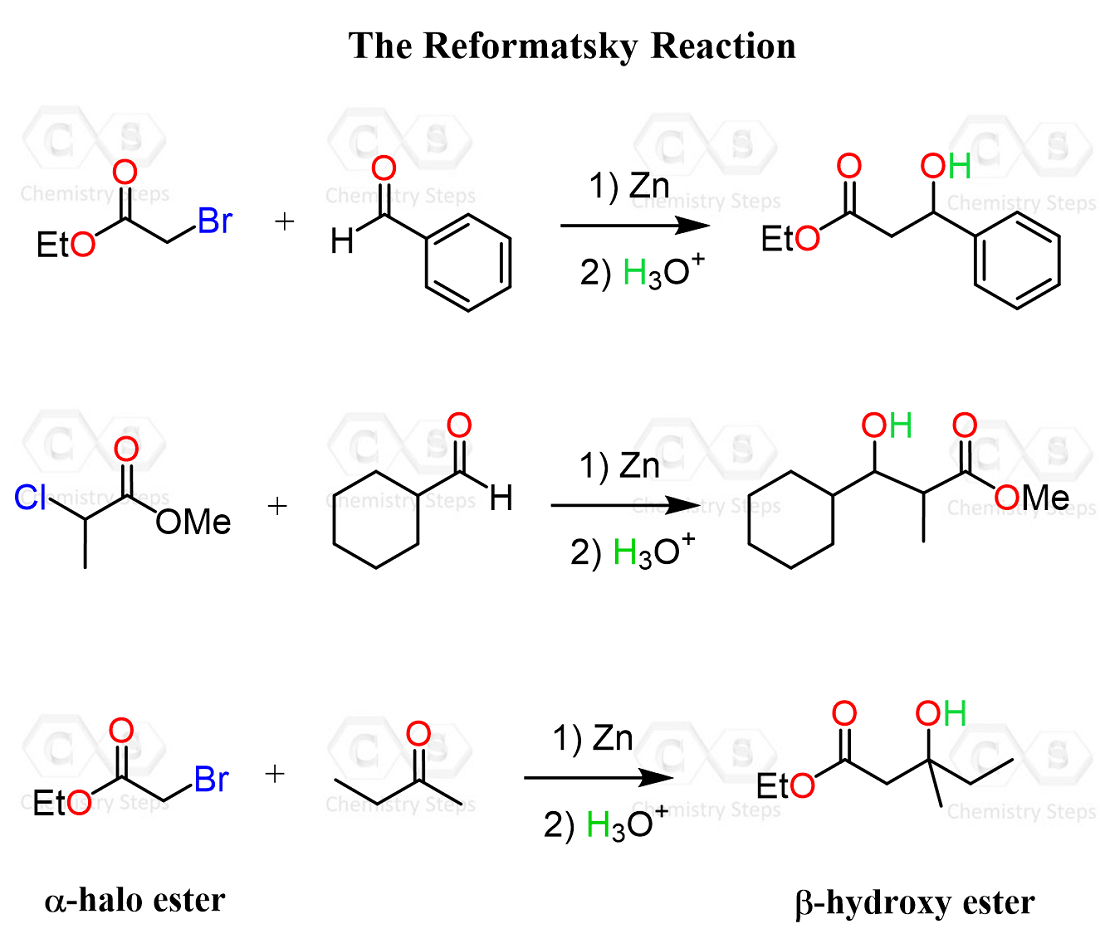
The idea here is to convert the halo ester to an enolate, which then attacks the aldehyde or ketone to form the needed β-hydroxy carbonyl. So, compared to a regular crossed aldol, we can see that instead of the typical lithium enolates that are prepared using, for example, LDA, metallic zinc is used here.
Overall, this can be viewed as a more controlled alternative to crossed aldol reactions. Of course, the halogen on the α-position also plays a key role, as that is what helps the Zn to form the needed enolate.
The Formation of the Zn Enolate
The Zn enolate is formed by insertion of zinc metal into the C–X bond of the α-halo ester. This generates an organozinc intermediate, often referred to as a Zn enolate, which behaves similarly to a regular enolate.
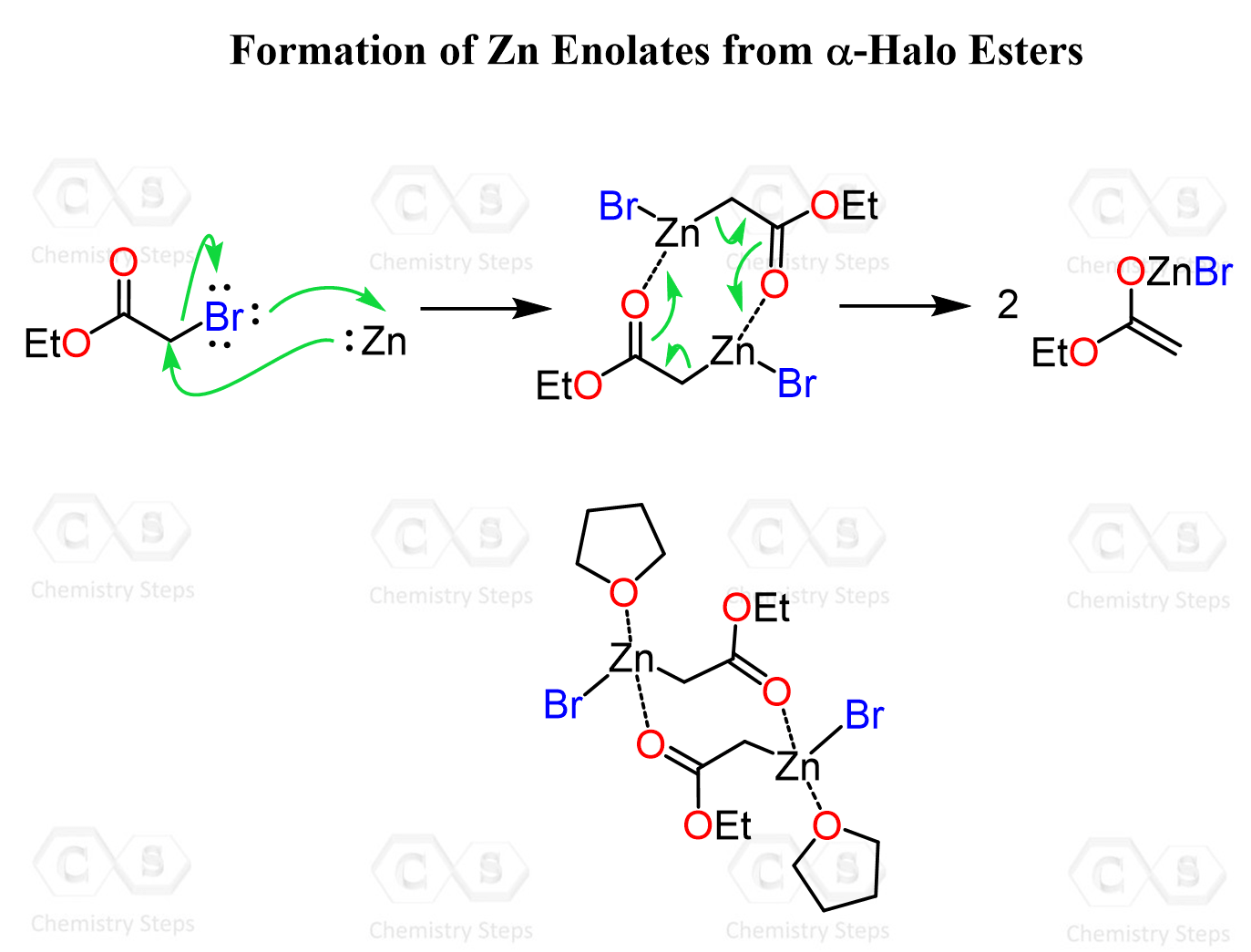
The cyclic molecule is coordinated to Zn in tetrahydrofuran (THF), which is a polar aprotic solvent. It stabilizes the organozinc intermediate by donating electron density through the oxygen lone pairs and coordinating to the metal center.
This is a common pattern in the behavior of organometallics – solvents like THF or diethyl ether act as ligands, helping to stabilize and solubilize reactive intermediates such as organozinc, Grignard, and organolithium reagents.
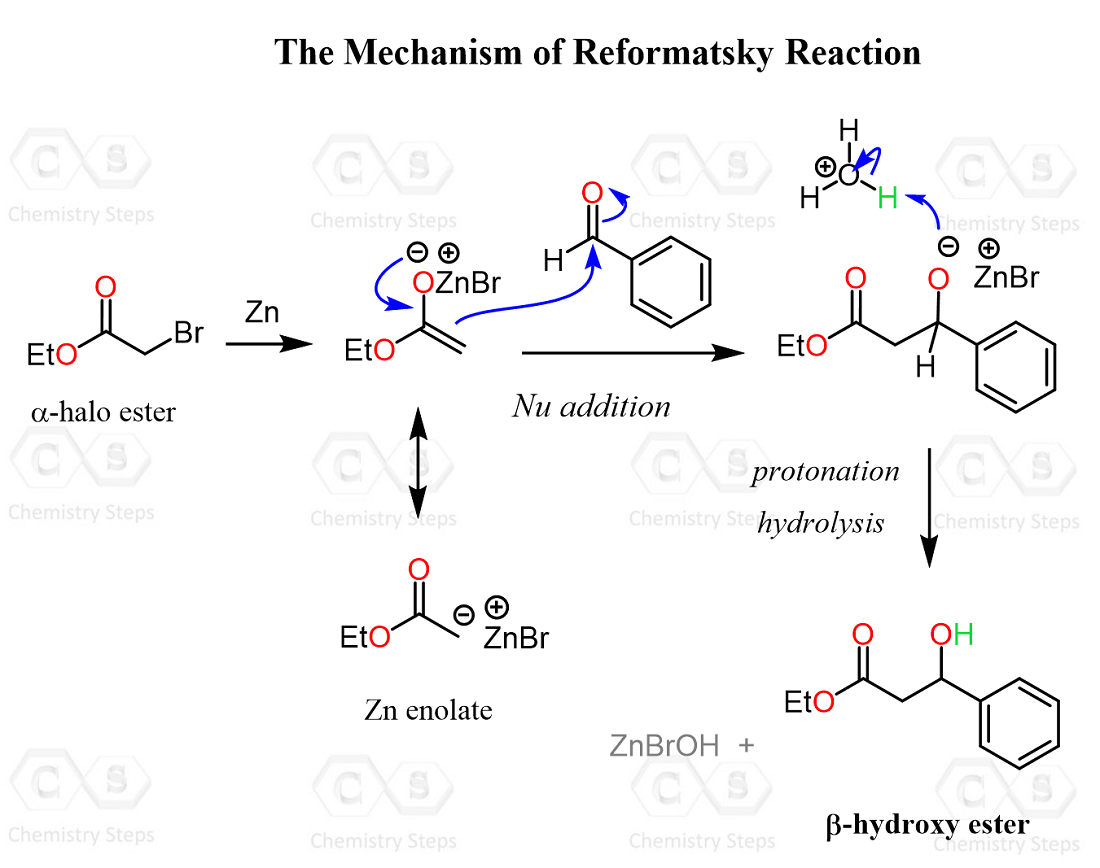
The Condensation of the Zn Enolate with Carbonyls
Once the Zn enolate is formed, it reacts in the same way as a typical enolate does. Remember, we first have a nucleophilic addition to the carbonyl group, forming an alkoxide intermediate, which is then protonated upon acidic workup, and the desired β-hydroxy ester is formed.
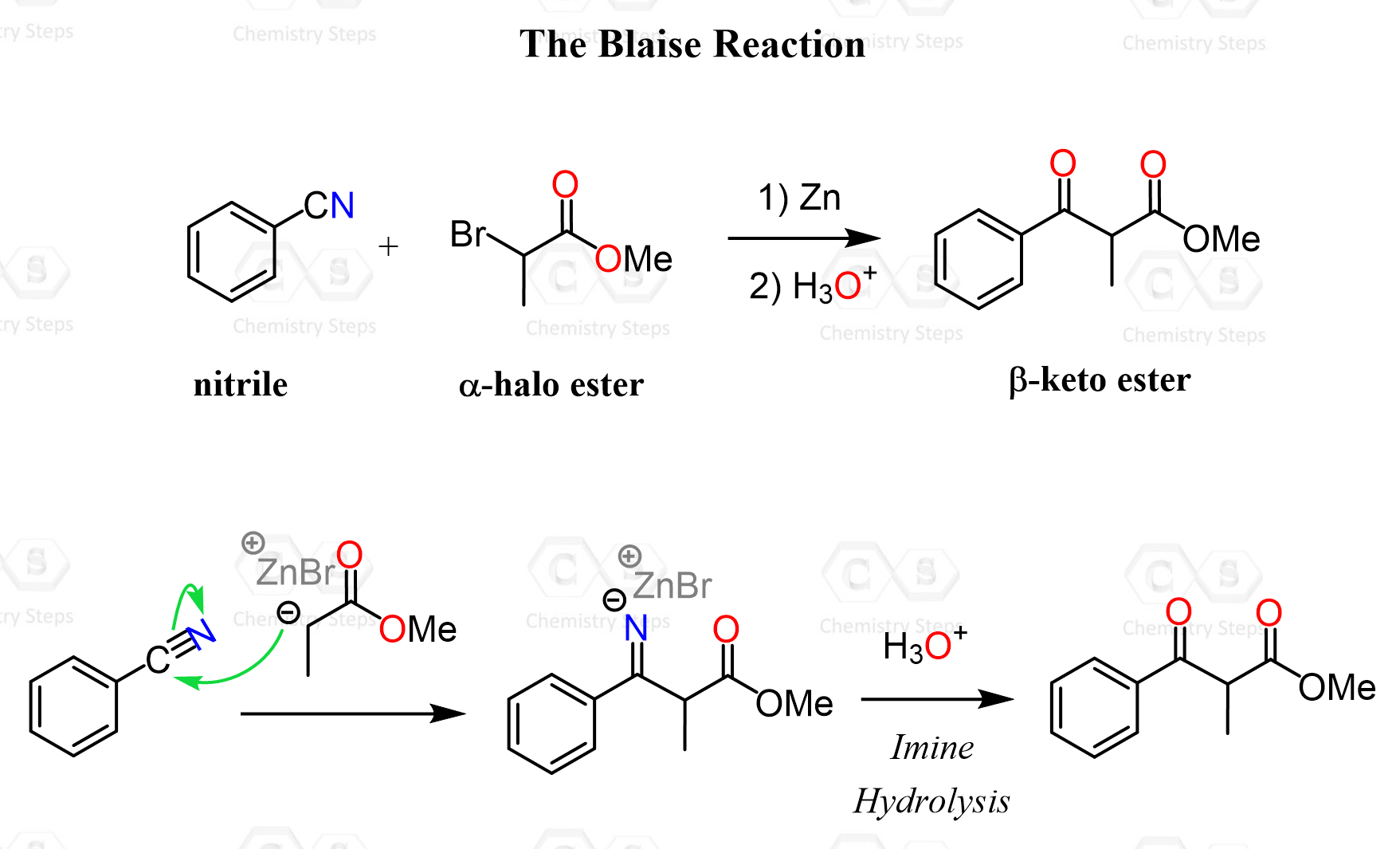
The Blaise Reaction
The Blaise reaction is a variation of the Reformatsky reaction, where instead of a typical carbonyl group, a nitrile is used as the electrophilic species.
Just like we have seen in the reactions of nitriles with Grignard and organolithium reagents, the Zn enolate attacks the carbon, forming an iminium intermediate. This intermediate is no longer electrophilic enough to undergo another nucleophilic addition, which explains why the reaction stops at this stage. Therefore, upon acidic workup, the imino group is hydrolyzed to the corresponding ketone, forming the β-keto ester.
Other Electrophiles in the Reformatsky Reaction
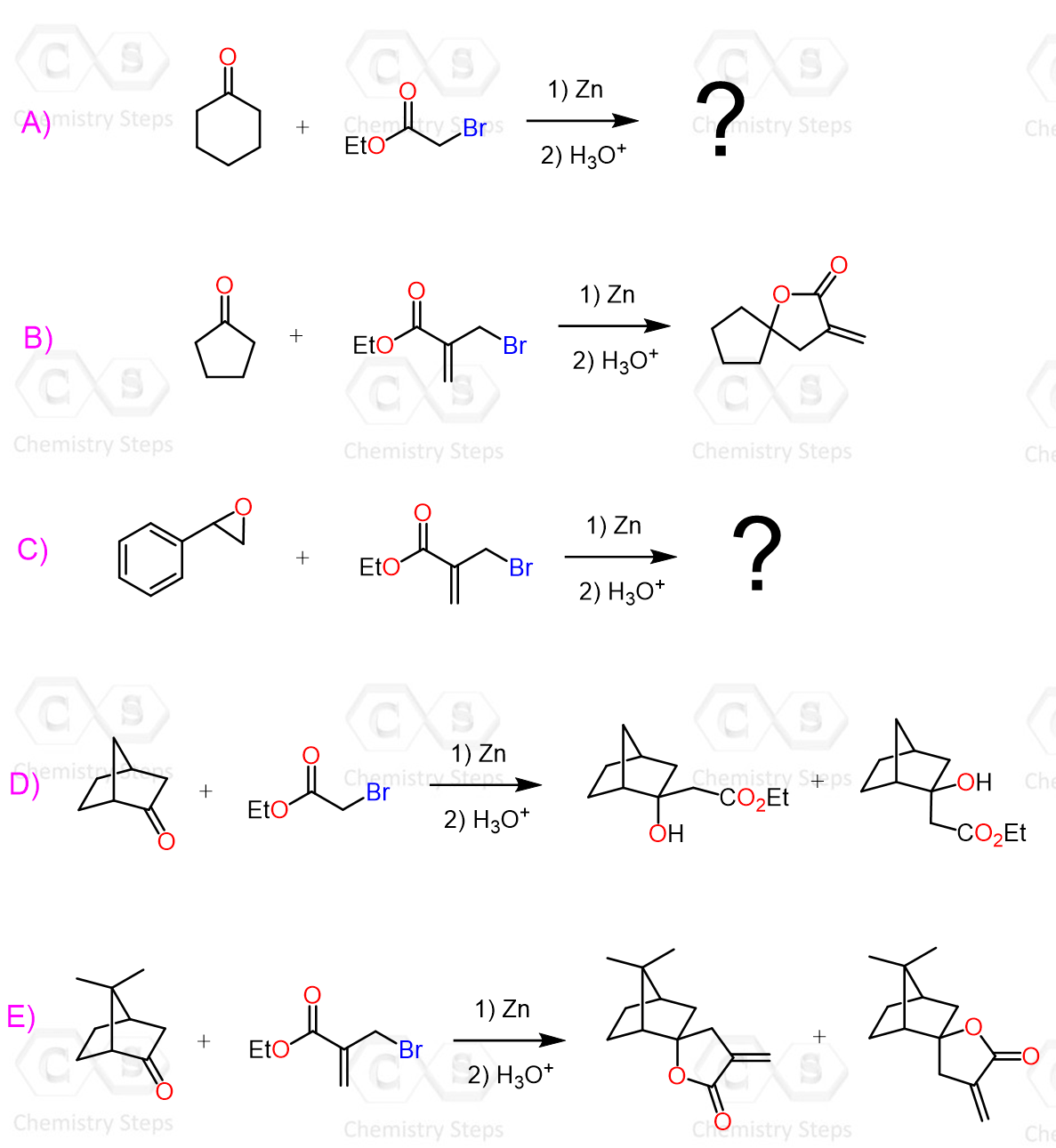
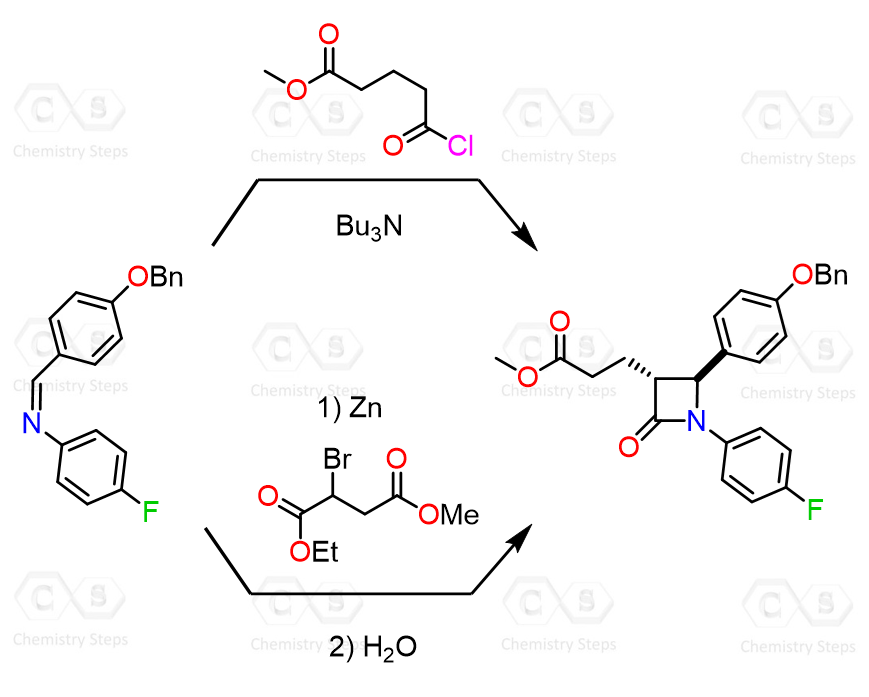
The Reformatsky reaction is not limited to aldehydes and ketones. In fact, it can react with a wide range of so-called “non-classical” electrophiles, including imines, nitriles, acid chlorides, anhydrides, esters, orthoformates, epoxides, azirines, and related systems. In all of these cases, the Reformatsky reagent behaves in the same general way – it acts as a nucleophile and adds to the electrophilic center.
In some ways, Gilman reagents are great alternatives to Grignard and organolithium reagents as they are milder and allow for some selective conversions, such as the acid chloride to ketone conversion.
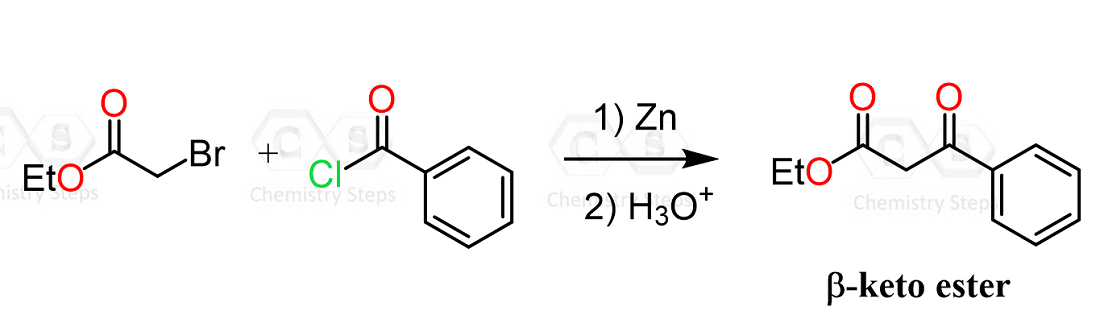
Remember, Grignard and organolithium reagents perform two nucleophilic additions to acid chlorides, converting them into tertiary alcohols.
Another important point is that the Reformatsky reaction is not limited to zinc only. While zinc is the classical metal, other metals, such as chromium-based systems and related low-valent metals, can also be used to change how the enolate is formed and how reactive it is. On top of that, transition metal additives, including palladium-based catalysts, can also be introduced in some variants to open up new pathways and improve reactivity or selectivity
Unfortunately, or not, all of this is beyond the scope of this post, but if interested, refer, among others, to these sources for a more advanced understanding of the Reformatsky reaction:
1) The Reformatsky reaction in organic synthesis. Recent advances, Tetrahedron, 2004, (60) 9325–9374
2) Recent Advances in the Reformatsky Reaction, Synthesis 1989 (8) 571–590,
3) Clayden, Organic Chemistry
Summarizing the Reformatsky Reaction
The Reformatsky reaction is a valuable alternative to classical crossed aldol reactions, offering a more controlled way to generate β-hydroxy carbonyl compounds, especially β-hydroxy esters.
- One of its key advantages is that it proceeds under neutral or near-neutral conditions, in contrast to aldol reactions that typically require strong bases or acids. This makes it particularly useful for base-sensitive substrates.
- Another important advantage is the high chemoselectivity of the zinc enolate formation: the reaction occurs specifically at the carbon bearing the halogen, and in classical cases, no competing O-functionalization products are observed.
- The use of zinc also suppresses self-condensation, since the organozinc species does not react with esters in the same way as strongly basic enolates do.
In addition, Reformatsky-type chemistry is quite versatile, having been extended to a wide range of haloesters, different electrophiles beyond aldehydes and ketones, and even other metals or catalytic systems that can mimic zinc insertion, making it a broadly useful C–C bond-forming strategy.