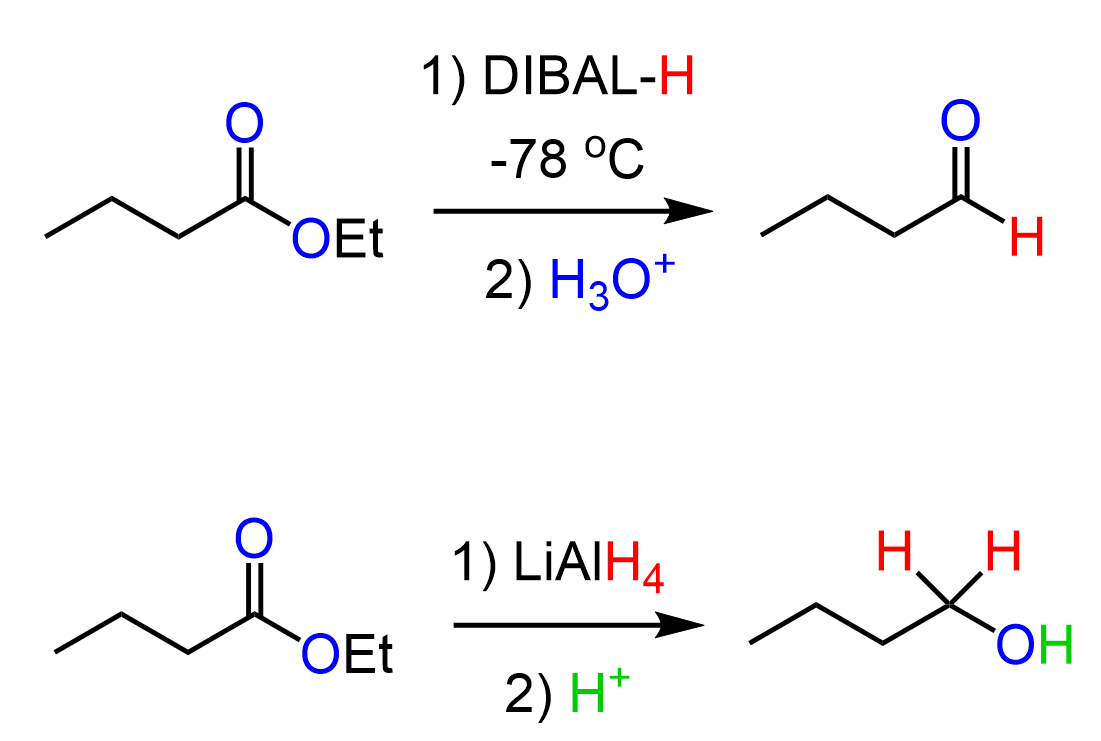
Let’s discuss how esters can be converted to aldehydes. In the previous post, we saw that esters can be reduced to primary alcohols using a strong reducing agent such as LiAlH4:

Now, we can of course oxidize the primary alcohol to the corresponding aldehyde using mild oxidizing agents such as PCC, PDC, Swern, or Dess Martin:

However, let’s why we cannot achieve this with LiAlH4 itself, and what is the way of doing this.
Overall the transformation is two hydride additions to the carbonyl. The first one converts the ester to an aldehyde, and the second reduces the aldehyde to the corresponding primary alcohol:

It would be nice if we could stop the reaction after the first addition, however, remember that aldehydes are more reactive than esters and once formed, it is quickly reduced to the corresponding alcohol by another hydride addition.
Reduction of Esters to Aldehydes using DIBAL
Fortunately, there is another reducing agent known as DIBAL or DIBAL-H that can reduce esters to aldehydes if the reaction is carried out at lower temperatures (typically -78 oC as that is the temperature of dry ice):
The mechanism is not very different from what we have seen in typical hydride reductions of acids and esters. It starts with the coordination of the carbonyl oxygen to the Al which has an empty p orbital and thus acts as a Lewis acid. This activates the carbonyl by withdrawing some electron density, and the hydride addition occurs forming a hemiacetal intermediate:
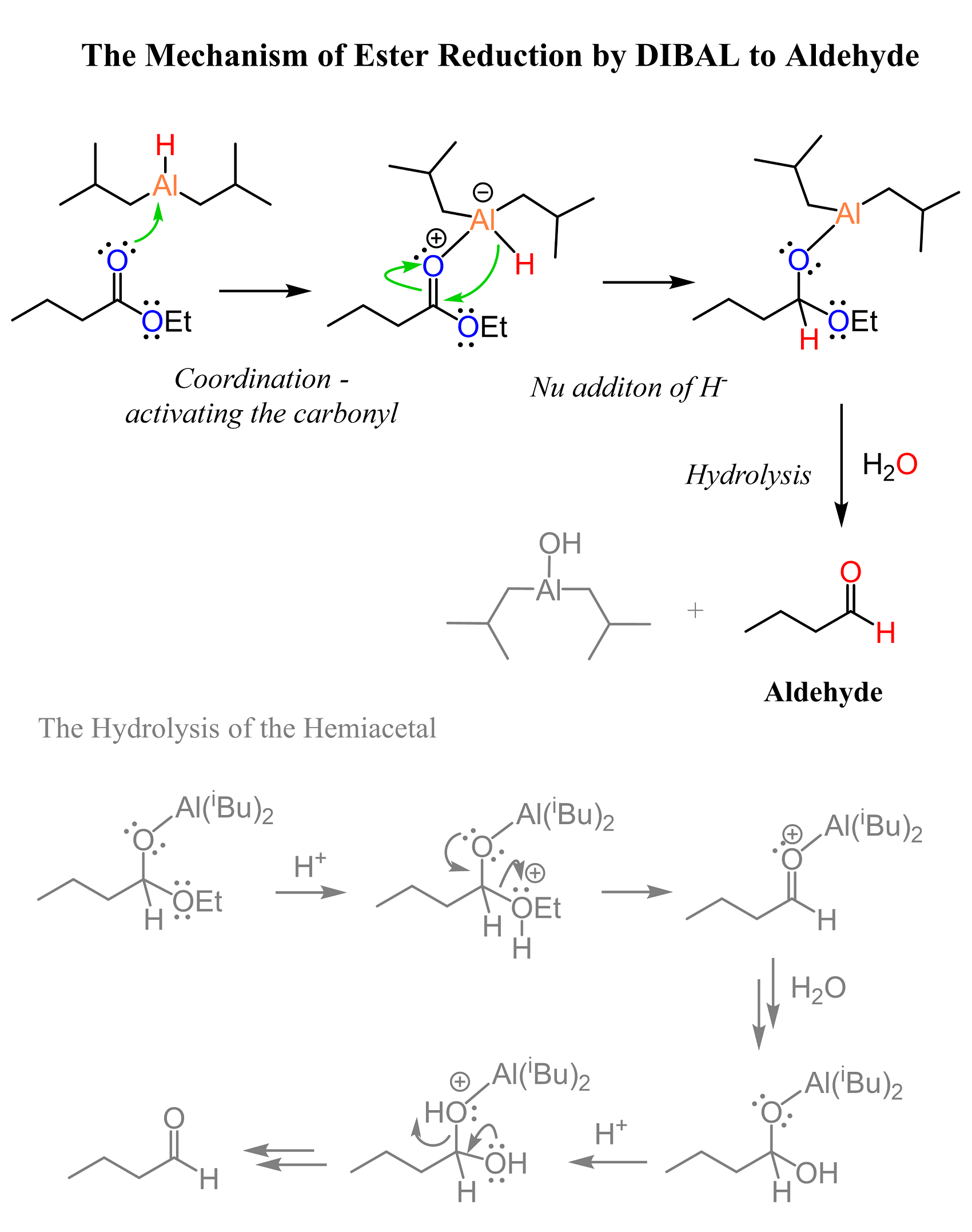
Like other hemiacetals, we can hydrolyze it to the corresponding aldehyde by an aqueous or acidic workup.
Although this looks easier on paper than it is in the lab, DIBAL can also be used to reduce nitriles to aldehydes in a similar manner:

The intermediate in this reduction is an imine, which is then hydrolyzed to an aldehyde.
In the next article, we will discuss the strategies for converting esters to ketones.
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- LiAlH4 and NaBH4 Carbonyl Reduction Mechanism
- Alcohols from Carbonyl Reductions – Practice Problems
- Grignard Reaction in Preparing Alcohols with Practice Problems
- Grignard Reaction in Organic Synthesis with Practice Problems
- Fischer Esterification
- Ester Hydrolysis by Acid and Base-Catalyzed Hydrolysis
- What is Transesterification?
- Esters Reaction with Amines – The Aminolysis Mechanism
- Ester Reactions Summary and Practice Problems
- Preparation of Acyl (Acid) Chlorides (ROCl)
- Reactions of Acid Chlorides (ROCl) with Nucleophiles
- Reaction of Acyl Chlorides with Grignard and Gilman (Organocuprate) Reagents
- Reduction of Acyl Chlorides by LiAlH4, NaBH4, and LiAl(OtBu)3H
- Preparation and Reaction Mechanism of Carboxylic Anhydrides
- Amides – Structure and Reactivity
- Naming Amides
- Amides Hydrolysis: Acid and Base-Catalyzed Mechanism
- Amide Dehydration Mechanism by SOCl2, POCl3, and P2O5
- Amide Reduction Mechanism by LiAlH4
- Amides Preparation and Reactions Summary
- Amides from Carboxylic Acids-DCC and EDC Coupling
- The Mechanism of Nitrile Hydrolysis To Carboxylic Acid
- Nitrile Reduction Mechanism with LiAlH4 and DIBAL to Amine or Aldehyde
- The Mechanism of Grignard and Organolithium Reactions with Nitriles
- Carboxylic Acids to Ketones
- Esters to Ketones
- Carboxylic Acids and Their Derivatives Practice Problems
- Carboxylic Acids and Their Derivatives Quiz