We know that aldehydes and ketones react with one equivalent of Grignard reagents, producing secondary and tertiary alcohols, respectively. Esters, on the other hand, react with two equivalents of Grignard reagents, producing tertiary alcohols:

What happens is we have two nucleophilic additions to the carbonyl. The first is an addition-elimination where the alkoxy group of the ester is expelled from the tetrahedral intermediate. This restores the carbonyl group in the form of a ketone, which then reacts with another equivalent of the Grignard reagent, forming a tertiary alcohol:

A question you may be wondering here is why the Grignard reagent doesn’t react with the ester instead of the newly formed ketone. Remember that aldehydes and ketones are more reactive than esters since the electrophilicity of the carbon atom of the ester is partially suppressed by the lone pair of the oxygen through resonance stabilization:

This effect is more profound in amides which makes them the least reactive among carboxylic acid derivatives:

Therefore, once the ketone is formed, it is going to react with the Grignard reagent faster than the ester, so we cannot stop the reaction at this stage.
This demonstrates why two equivalents of Grignard are needed when reacted with esters. If one equivalent were used, a mixture of a tertiary alcohol, ketone, and unreacted ester would have been obtained after the acidic work-up:
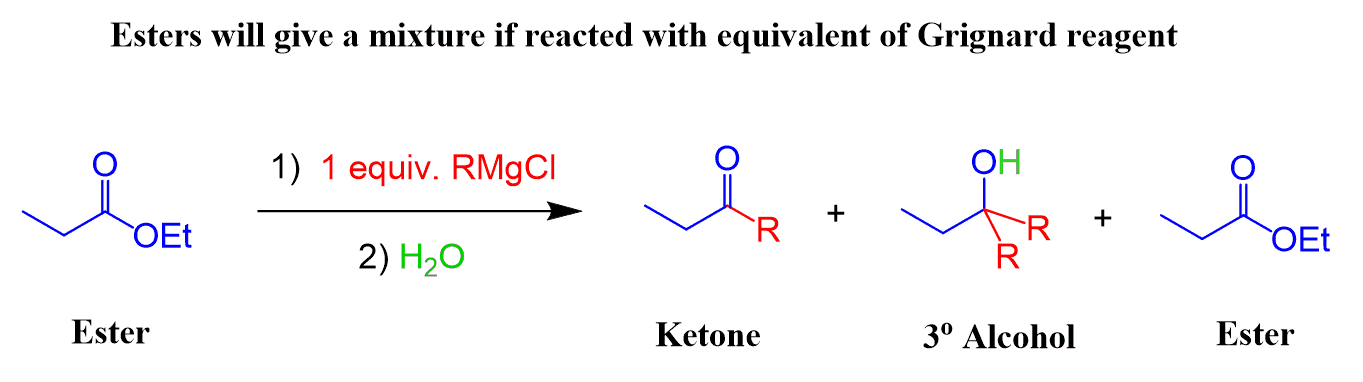
Therefore, we always use an excess of Grignard reagent with esters, to ensure one product, and that is the tertiary alcohol, is obtained.
Esters can be converted to ketones, but we need to first hydrolyze them to carboxylic acids. These do not undergo nucleophilic additions by Grignard reagent; however, organolithiums are powerful enough to do a nucleophilic attack after the deprotonation of the acid:

There are other ways too for converting carboxylic acids to ketones, and we mention them in a separate post, which you can find here.
Organolithiums with Esters
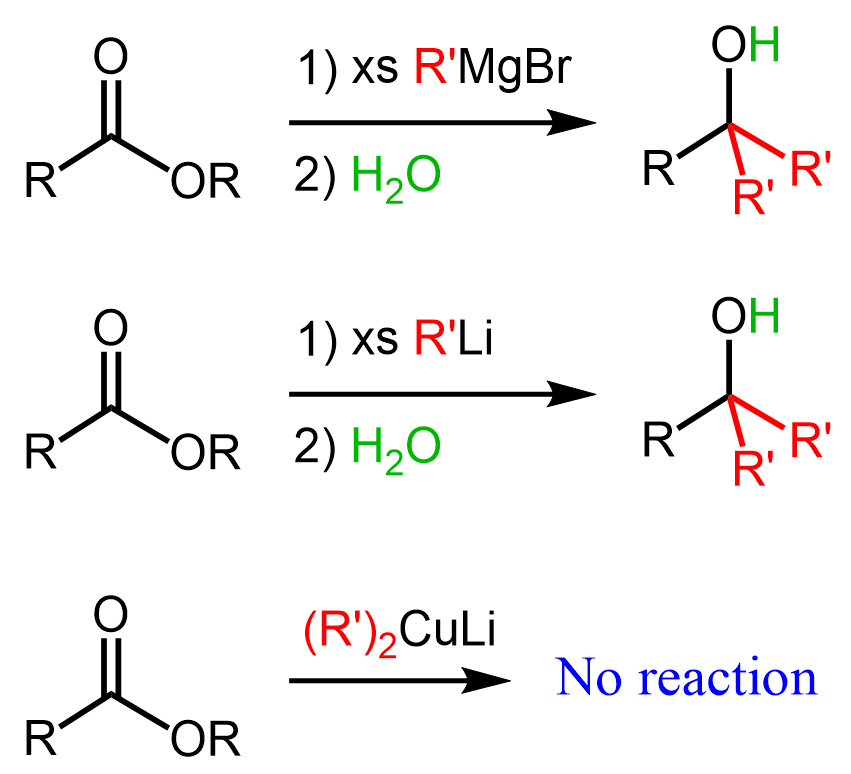
The same pattern and outcome are observed with esters when reacted with organolithiums. Notice that organocuprates (Gilman reagent) are less reactive and do not react with esters:
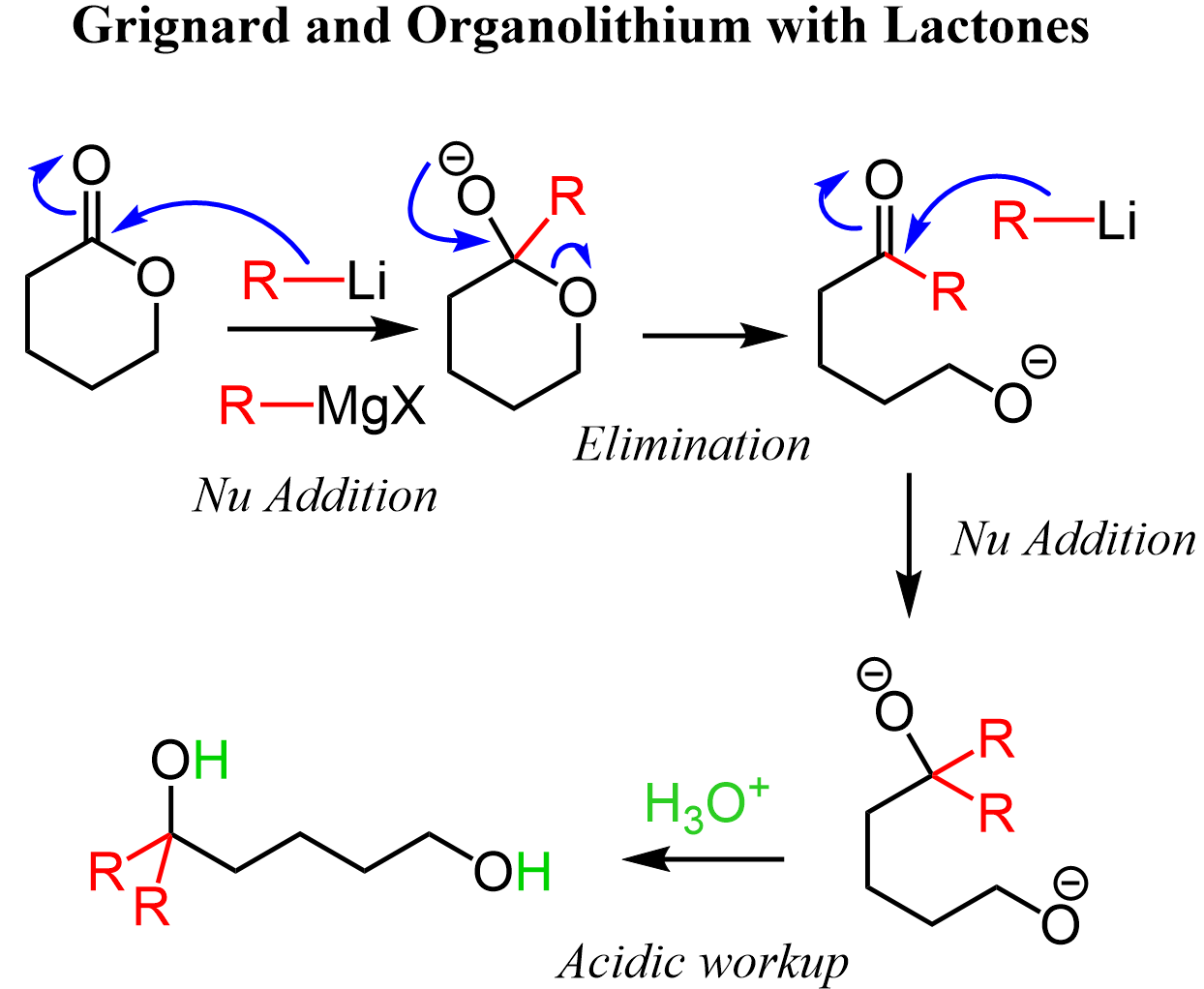
Let’s show the mechanism of an organolithium with cyclic esters known as lactones. The steps are the same: we have a nucleophilic addition-elimination, which forms a ketone and opens the ring. The ketone is then attacked by the carbon nucleophile, and after the workup, two alcohol groups are formed:
Check Also
- LiAlH4 and NaBH4 Carbonyl Reduction Mechanism
- Alcohols from Carbonyl Reductions – Practice Problems
- Grignard Reaction in Preparing Alcohols with Practice Problems
- Grignard Reaction in Organic Synthesis with Practice Problems
- Fischer Esterification
- Ester Hydrolysis by Acid and Base-Catalyzed Hydrolysis
- What is Transesterification?
- Esters Reaction with Amines – The Aminolysis Mechanism
- Ester Reactions Summary and Practice Problems
- Preparation of Acyl (Acid) Chlorides (ROCl)
- Reactions of Acid Chlorides (ROCl) with Nucleophiles
- Reaction of Acyl Chlorides with Grignard and Gilman (Organocuprate) Reagents
- Reduction of Acyl Chlorides by LiAlH4, NaBH4, and LiAl(OtBu)3H
- Preparation and Reaction Mechanism of Carboxylic Anhydrides
- Amides – Structure and Reactivity
- Naming Amides
- Amides Hydrolysis: Acid and Base-Catalyzed Mechanism
- Amide Dehydration Mechanism by SOCl2, POCl3, and P2O5
- Amide Reduction Mechanism by LiAlH4
- Amides Preparation and Reactions Summary
- Amides from Carboxylic Acids-DCC and EDC Coupling
- The Mechanism of Nitrile Hydrolysis To Carboxylic Acid
- Nitrile Reduction Mechanism with LiAlH4 and DIBAL to Amine or Aldehyde
- The Mechanism of Grignard and Organolithium Reactions with Nitriles
- Carboxylic Acids to Ketones
- Esters to Ketones
- Carboxylic Acids and Their Derivatives Practice Problems
- Carboxylic Acids and Their Derivatives Quiz

Thank you so much for this post. I was looking everywhere for this mechanism and I could not find it. Here is so well explained. You saved my organic chemistry exam.
I’m really glad it helped you. Thanks for the kind words and good luck on your organic chemistry exam!