We have seen in many examples how Grignard and organolithium reagents react with various carbonyl compounds such as aldehydes, ketones, esters, and more. So, let’s discuss what happens when we react a Grignard or organolithium reagent with carbon dioxide. Although CO₂ may be seen as an unreactive gas because of its abundance in the atmosphere and its relative rarity in classic organic reaction textbooks, its central carbon atom is highly electrophilic.

Each C=O bond is a polarized π bond. Oxygen, being more electronegative, pulls electron density away from carbon, leaving it with a partial positive charge (δ⁺). This makes it a linear molecule primed for attack by strong nucleophiles such as the Grignard and organolithium reagents.
Remember, these are organometallics where the carbon is connected to a metal and, because of their low electronegativities, they render the carbon atom rich in electron density, thus making them strong bases and nucleophiles.
The Mechanism of Grignard and Organolithium Reagents with Carbon Dioxide
In the first step, the nucleophilic carbon chain of your organometallic reagent (R⁻ from RMgX or RLi) attacks the electrophilic carbon atom of CO₂, breaking one of its π bonds. That attack pushes the π electrons onto one oxygen, giving you a metal‑stabilized carboxylate ion. The carboxylate ion is simply treated with an aqueous acidic work‑up (H₃O⁺), converting the carboxylate into the corresponding carboxylic acid:
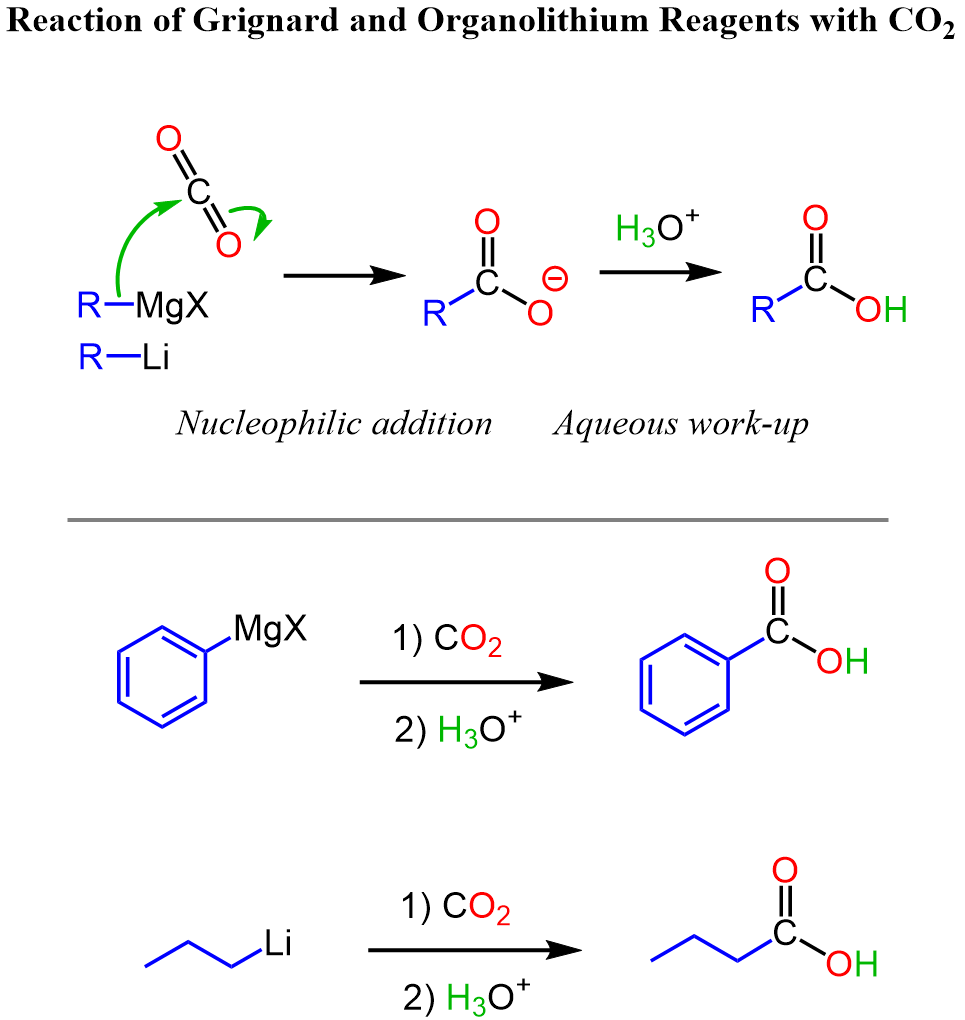
Notice that the carbon chain is extended by one carbon atom coming from the carbon dioxide, so this is a great synthetic strategy for introducing a new functional group and extending the carbon chain.
Check Also
- Organic Reaction Maps
- Grignard Reaction in Preparing Alcohols with Practice Problems
- Grignard Reaction in Organic Synthesis with Practice Problems
- The Grignard Reaction of Epoxides
- Preparation of Carboxylic Acids
- Naming Carboxylic Acids
- Naming Nitriles
- Naming Esters
- Naming Carboxylic Acid Derivatives – Practice Problems
- Fischer Esterification
- Ester Hydrolysis by Acid and Base-Catalyzed Hydrolysis
- What is Transesterification?
- Esters Reaction with Amines – The Aminolysis Mechanism
- Ester Reactions Summary and Practice Problems
- Preparation of Acyl (Acid) Chlorides (ROCl)
- Reactions of Acid Chlorides (ROCl) with Nucleophiles
- Reduction of Acyl Chlorides by LiAlH4, NaBH4, and LiAl(OtBu)3H
- Preparation and Reaction Mechanism of Carboxylic Anhydrides
- Amides – Structure and Reactivity
- Naming Amides
- Amides Hydrolysis: Acid and Base-Catalyzed Mechanism
- Amide Dehydration Mechanism by SOCl2, POCl3, and P2O5
- Amide Reduction Mechanism by LiAlH4
- Reduction of Amides to Amines and Aldehydes
- Amides Preparation and Reactions Summary
- Amides from Carboxylic Acids-DCC and EDC Coupling
- The Mechanism of Nitrile Hydrolysis To Carboxylic Acid
- Nitrile Reduction Mechanism with LiAlH4 and DIBAL to Amine or Aldehyde
- The Mechanism of Grignard and Organolithium Reactions with Nitriles
- The Reactions of Nitriles
- Converting Nitriles to Amides
- Carboxylic Acids to Ketones
- Esters to Ketones
- Carboxylic Acids and Their Derivatives Practice Problems
- Carboxylic Acids and Their Derivatives Quiz
- Reactions Map of Carboxylic Acid Derivatives
