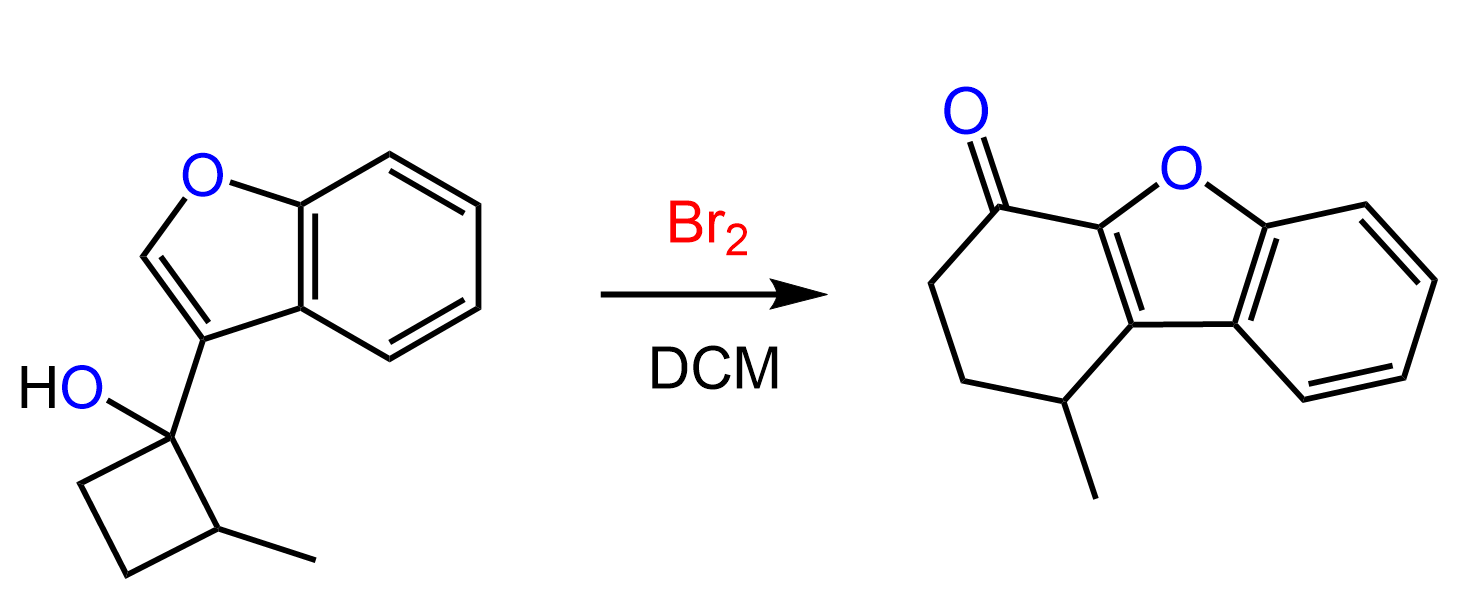
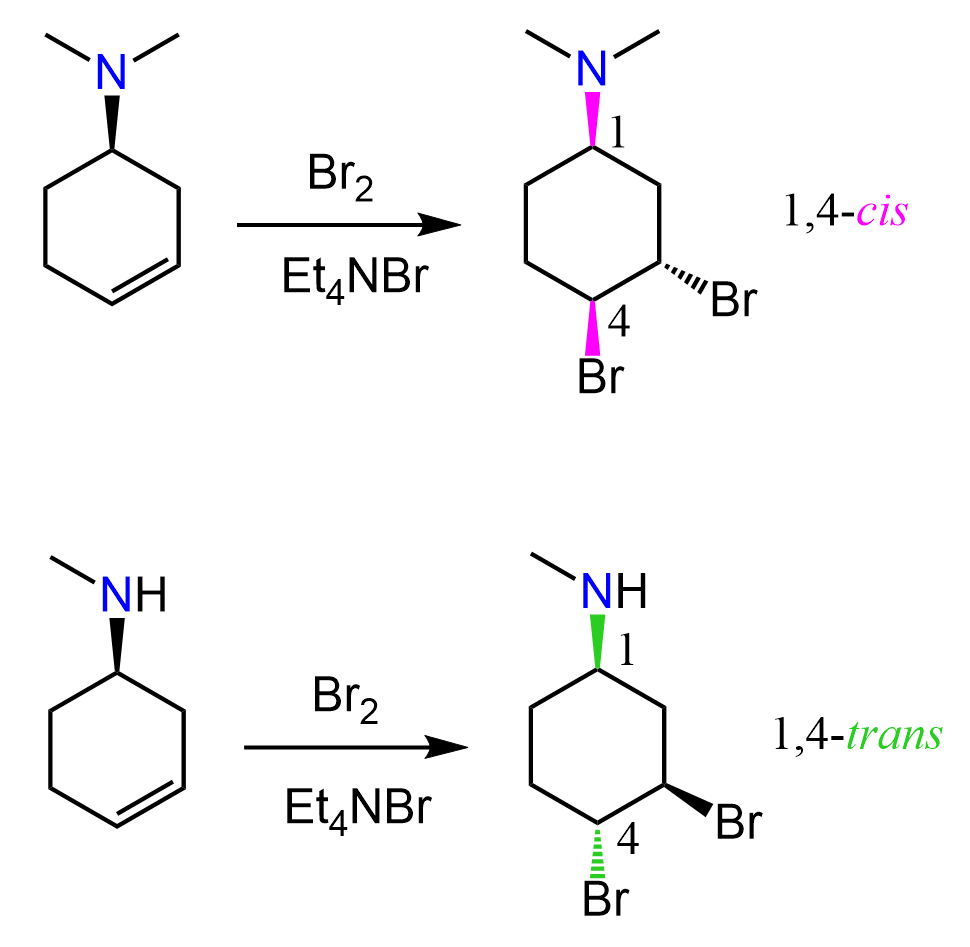
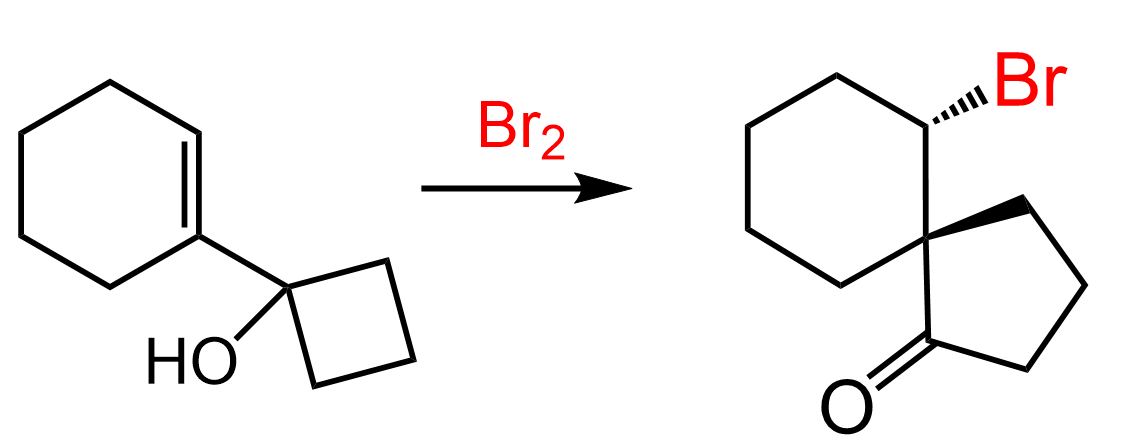
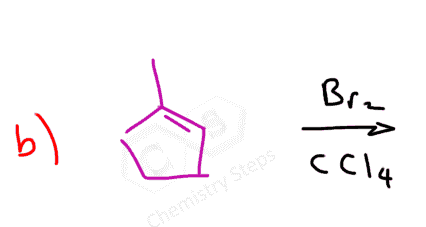Halonium ions are structures where a halogen atom is connected to two alkyl groups:

In your organic chemistry class, you are only going to worry about a specific type of halonium ion where the halogen is part of a three-membered ring:

Halonium ions are formed as a result of the electrophilic addition of halogens to alkenes. Most of the time, it is going to be bromine, in which case, we call it a bromonium ion.
Formation of Halonium Ions
Let’s recall that alkenes are electron-rich molecules because their double bonds contain an extra pair of electrons compared to single bonds. These extra electrons are found in the π bond, which is weaker and more reactive than a σ bond.
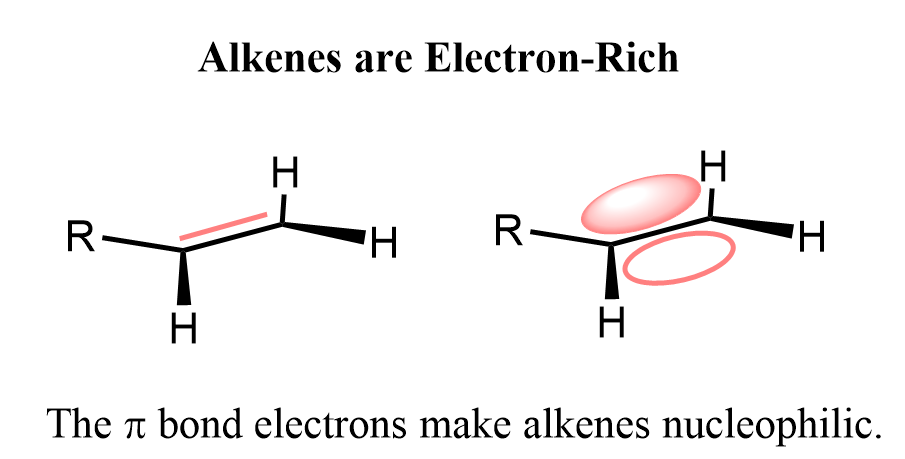
This makes the π electrons more available to react with electron-deficient species, or electrophiles. In other words, alkenes tend to act as nucleophiles in reactions because they can donate electrons from their π bond to electrophiles that are looking to accept them.
For example, here’s how the electrophilic addition of hydrohalic acids (like HBr or HCl) to alkenes takes place:
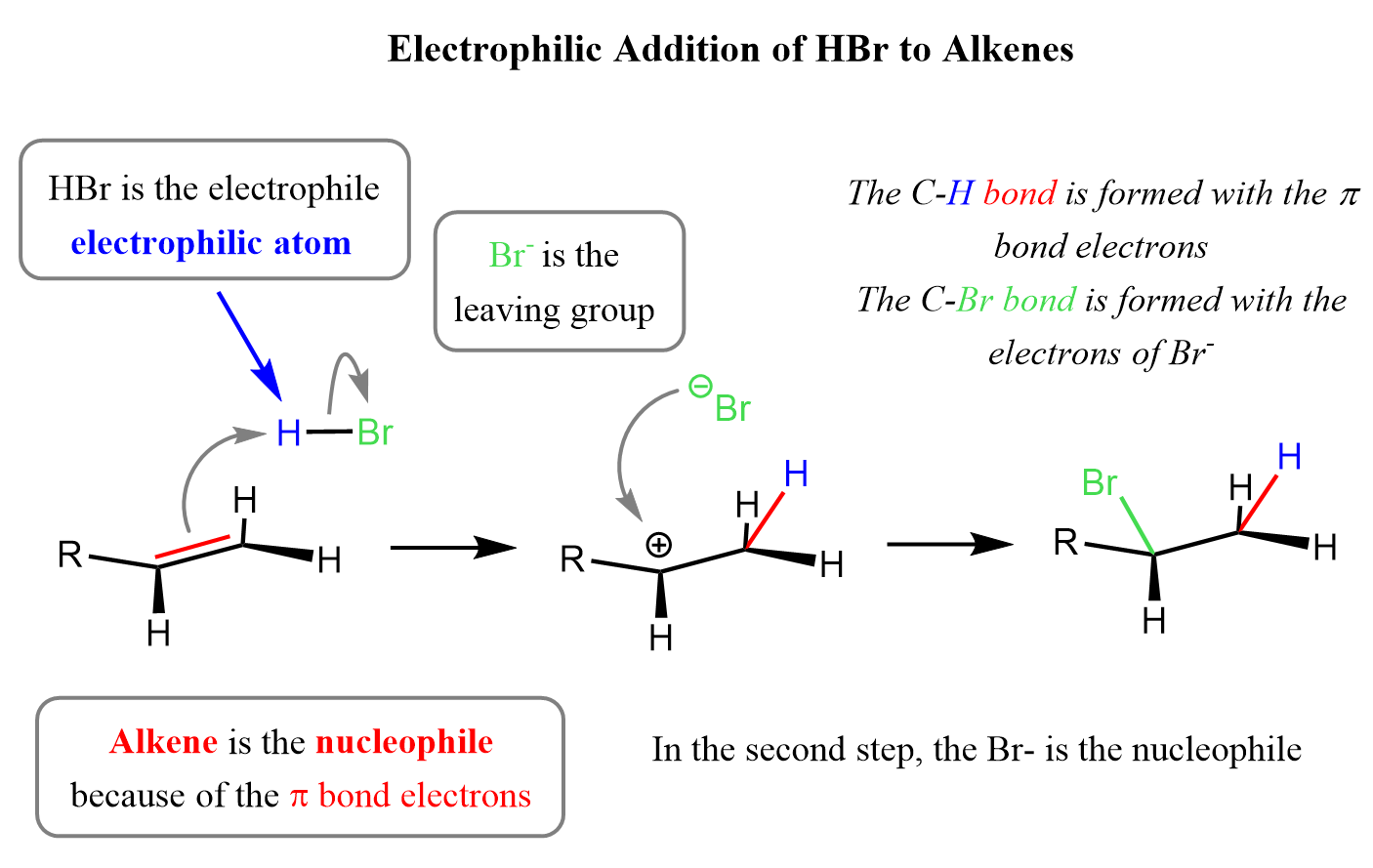
Keeping this pattern in mind, we can see the similarities with the addition reactions of alkenes. When an alkene reacts with a halogen molecule like Br₂, the first thing that happens is polarization of the Br–Br bond.
Even though Br₂ is a nonpolar molecule, the double bond in the alkene has a region of high electron density (the π bond). As the electrons from the double bond get close to the Br₂ molecule, they push electrons away from the nearby bromine atom toward the other one. This makes one bromine slightly positive (δ⁺) and the other slightly negative (δ⁻).
This temporary charge separation polarizes the Br–Br bond.
Now that one of the Br atoms has a slight positive charge, the alkene’s π electrons can attack it, forming a bond. At the same time, the Br–Br bond breaks, and the other Br leaves as a Br⁻ ion (a free halide ion).
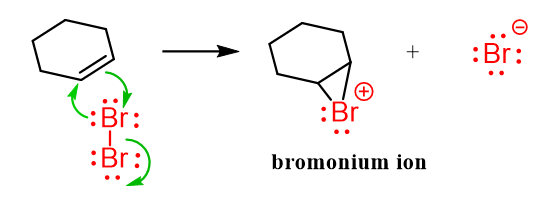
Notice that instead of forming a regular carbocation on one of the carbons, the positively charged bromine atom forms a three-membered ring with both carbon atoms of the former double bond. This is the halonium ion.
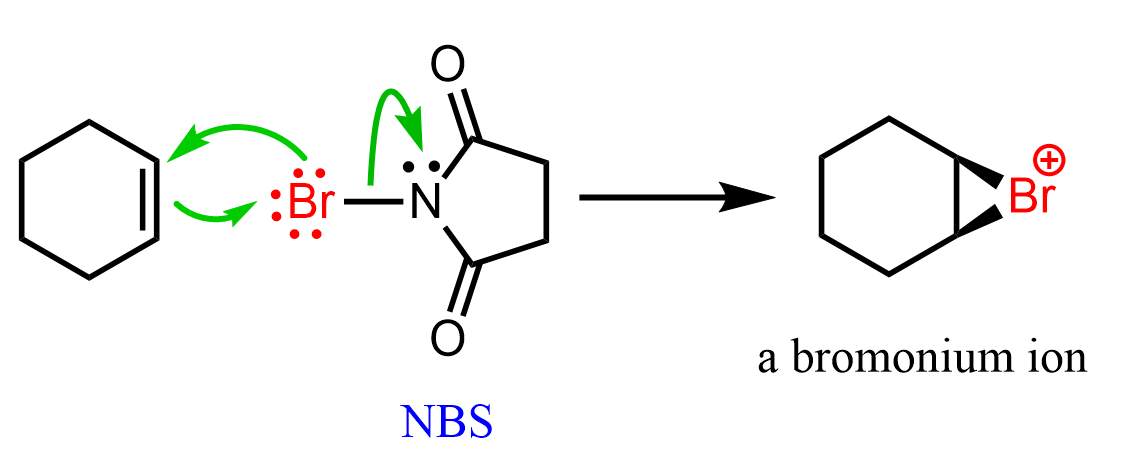
Another method for preparing halonium ions is the use of N-bromosuccinimide (NBS) instead of molecular bromine:
Reactions of Halonium Ions
What do we notice about the halonium ion? It is a three-membered ring, and they are also charged. Three-membered rings are associated with high ring strain, and charged species are not particularly stable in organic chemistry. If you have already covered epoxides, recall their reactivity towards almost any nucleophile due to their great ring strain:

Because of this high reactivity, the halonium ion is immediately attacked by the bromide ion formed during the addition to the double bond:

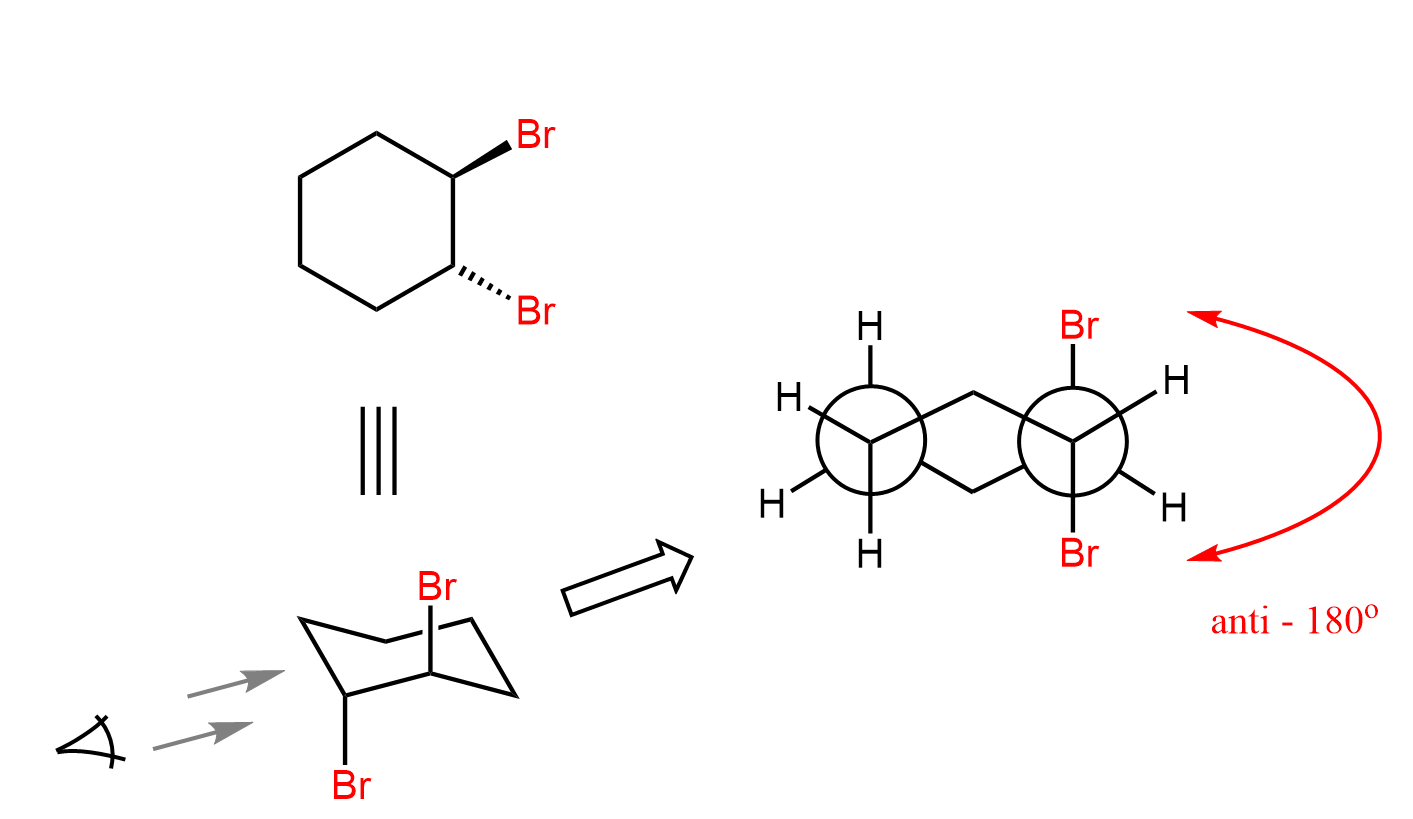
So, the bromine in the bromonium ion is the leaving group, and the bromide ion is the nucleophile, and overall, we have an SN2 substitution where the two species are at 180o. As a result, the two bromines have an anti orientation in the ring. Recall from Newman projections that anti conformation is when two groups of interest anti-periplanar, i.e., the dihedral angle between them is 180o:

We can also draw the Newman projection of this cyclohexane’s chair conformation to see the anti orientation of the two bromines:
Chlorine reacts the same way with alkenes, forming chloronium ion, which in general is called halonium ion. F2 and I2 are not synthetically useful for this reaction as F2 reacts explosively with the alkene, while the reaction with I2 does not proceed to a significant extent:

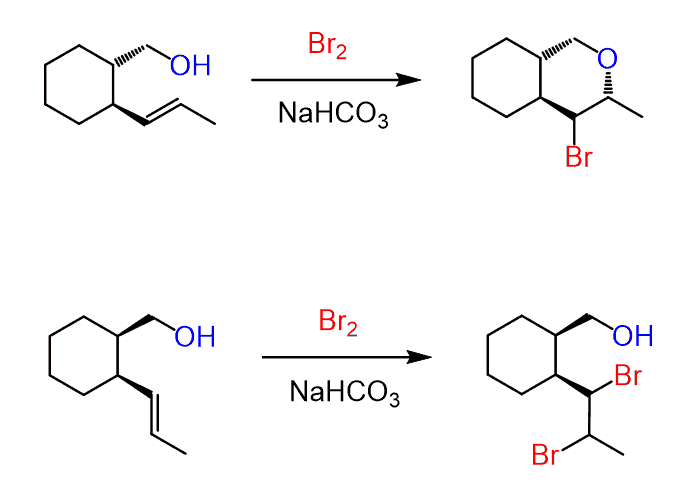
If other nucleophiles are present in the solution, the halonium ion is attacked by them in anti-addition, forming the corresponding trans product. For example, here is how the reaction proceeds when water is present in large quantities:
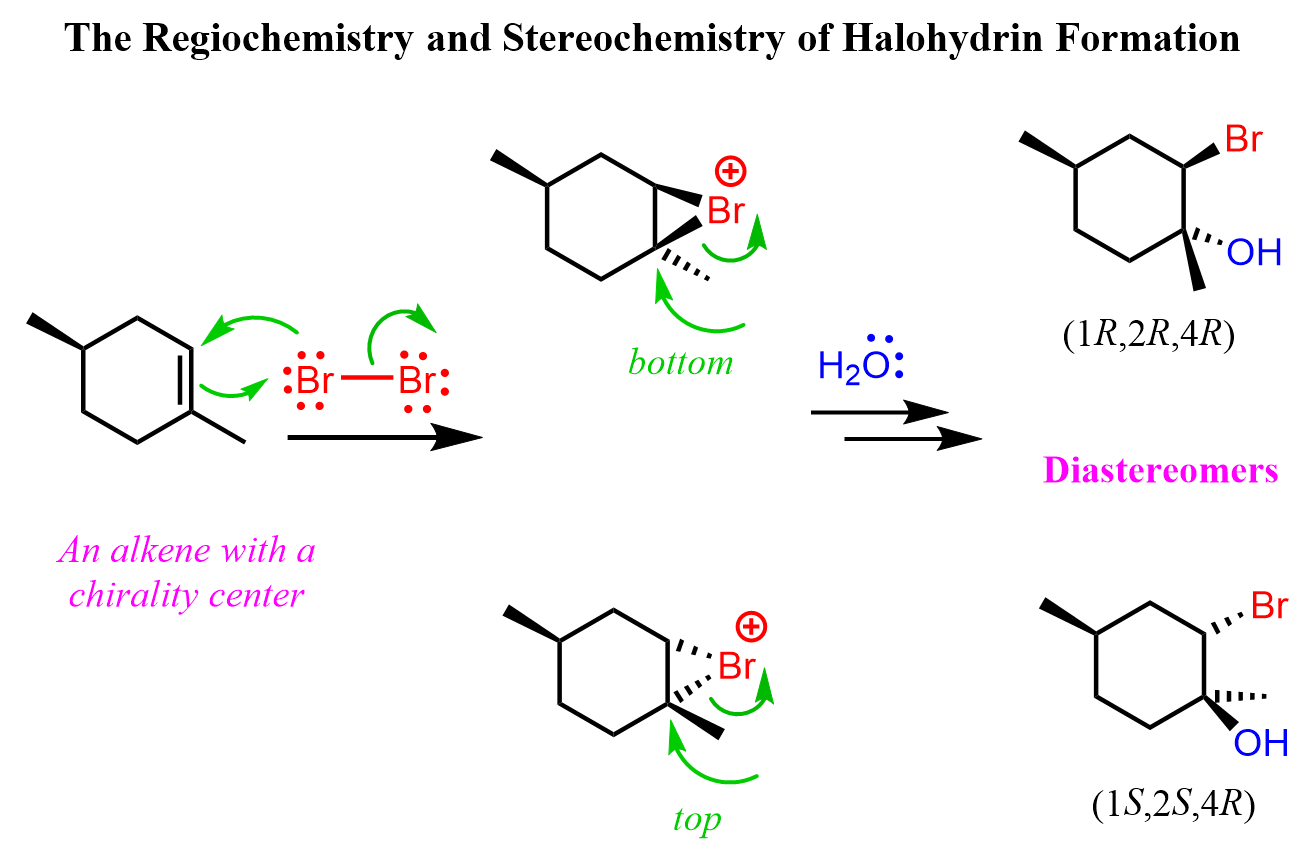
Notice that the chiral center in the starting material is not involved in the reaction, and the addition of water to both faces of the carbocation leads to the formation of diastereomers. Typically, in this type of reaction, when the starting material is achiral, the product is a racemic mixture of enantiomers.
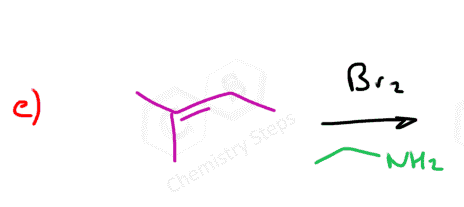
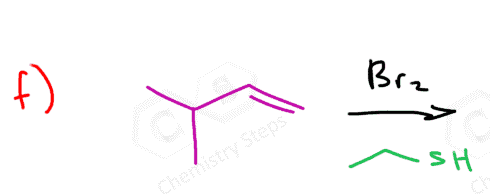
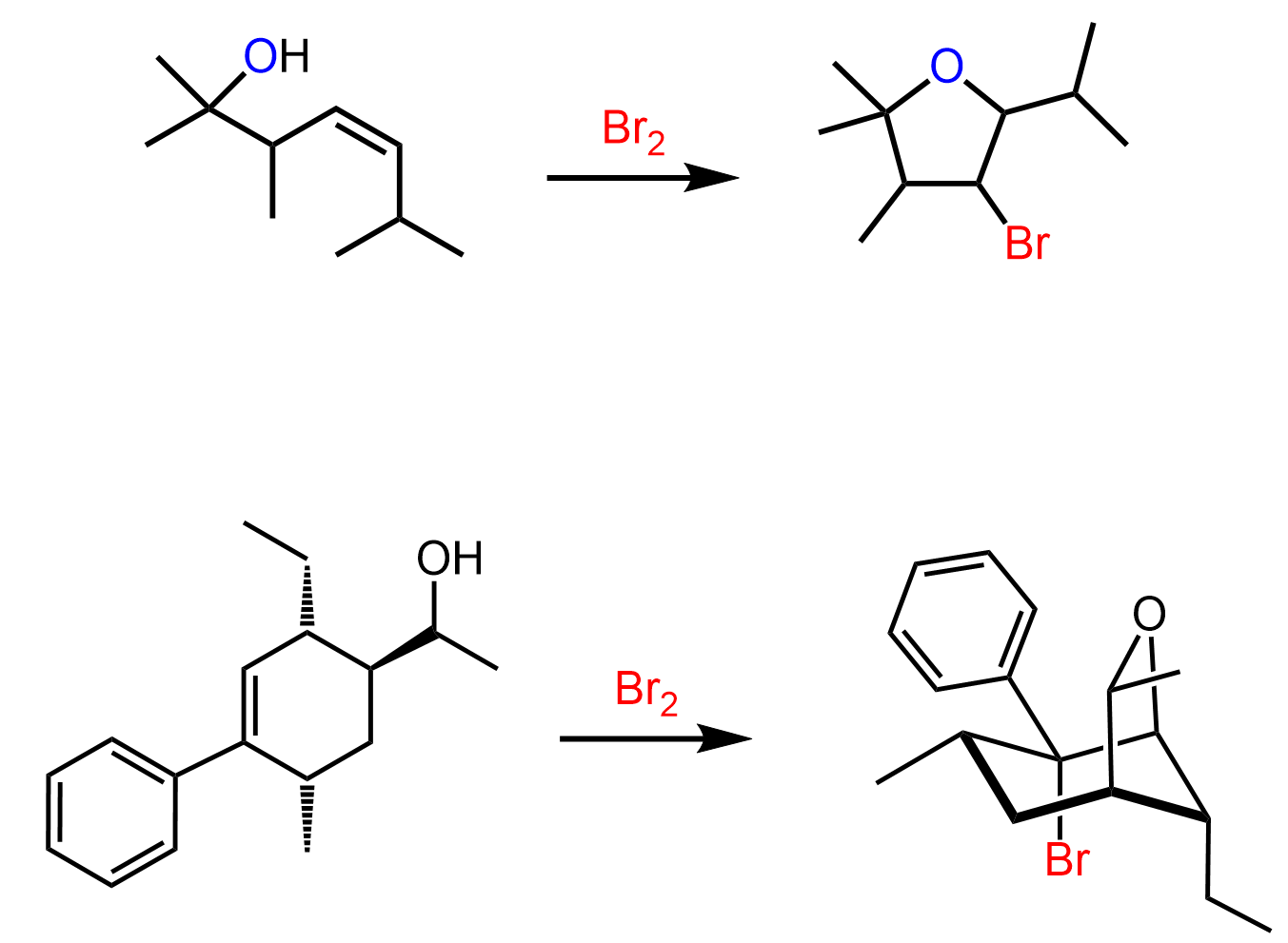
Instead of water, we can also use alcohols, thiols, and amines:
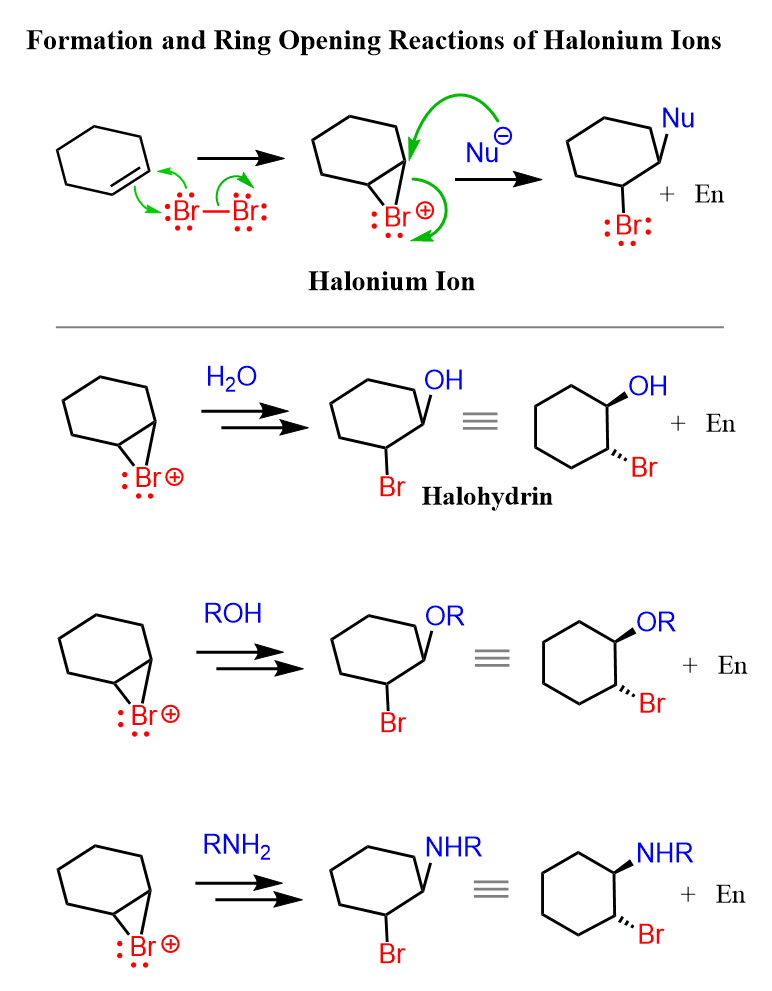
Notice that the nucleophile adds to the more substituted carbon, and the halogen ends up being on the less substituted carbon:

This is explained by the stability of the energy differences between the two possible transition states:
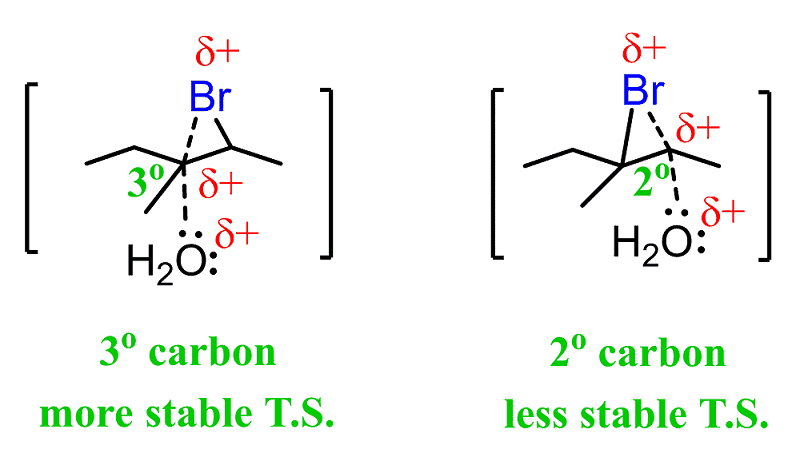
The first transition state has a partial positive charge on a more substituted carbon, making it more stable.
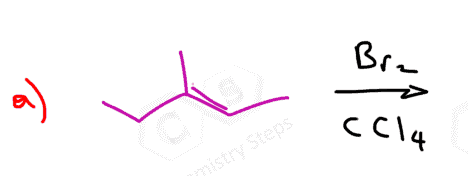
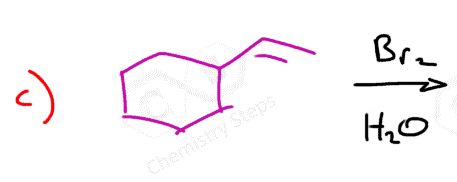
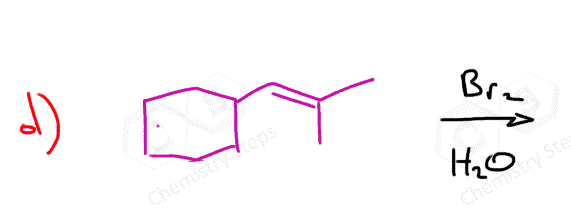
Here are some practice problems on addition reactions of alkenes that proceed through halonium ion intermediates.
We also have separate posts on the halogenation of alkenes and halohydrin formation, so feel free to check those out for more detailed explanations and mechanisms.