The alkylation of ketones is achieved through enolate intermediates, which are formed by deprotonating the alpha position (the carbon next to the carbonyl group):
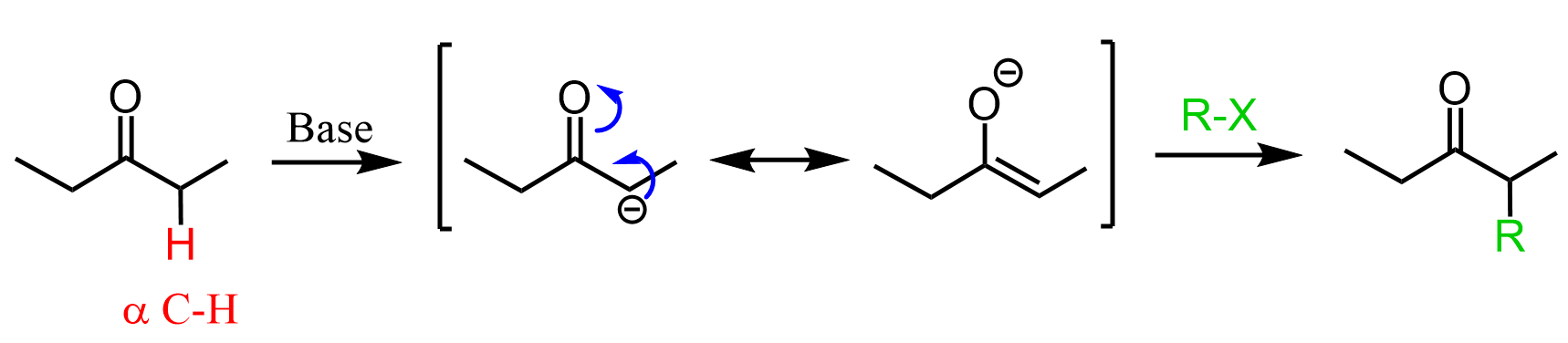
If you are still in the chapter on aldehydes and ketones, this may look foreign to you since all the reactions you have seen so far were directed to the C=O electrophilic carbon. So, yes, this is different because it starts with an acid-base reaction rather than a nucleophilic attack on the carbonyl. The acidity of the alpha hydrogen is very important, and it opens a whole new chapter of Alpha Carbon Chemistry.
Preparation of Enolates
One of the most common bases for preparing enolates from aldehydes and ketones is diisopropylamide, commonly used in the form of lithium diisopropylamide (LDA). This is a strong, non-nucleophilic base, which makes it ideal for deprotonating α-hydrogens without competing side reactions such as nucleophilic addition to the carbonyl.
LDA is typically generated in situ by reacting diisopropylamine with an organolithium reagent such as n-butyllithium. The resulting base is highly reactive and is therefore used under strictly anhydrous conditions.
The strength of LDA can be understood by comparing pKa values. The conjugate acid of LDA, diisopropylamine, has a pKa of about 36, while typical alpha hydrogens of carbonyl compounds have pKa values in the range of about 19 to 25. Because LDA is associated with a much weaker conjugate acid, it is strong enough to deprotonate these alpha carbons, making enolate formation both feasible and efficient.

Notice that in the unsymmetrical ketone, the methyl carbon is deprotonated instead of the one next to the R group. This is a key feature of LDA: its steric bulk, which prevents it from acting as a nucleophile. Instead of attacking the carbonyl carbon, it selectively removes the α-proton, forming the corresponding carbonyl or other C-H acid, such as for exokae nitreiels, in a clean and controlled manner.

The reaction is most commonly carried out in tetrahydrofuran (THF), which serves as an aprotic, coordinating solvent. THF stabilizes the lithium cation through coordination, helping to solvate the LDA and the resulting enolate, and ensuring efficient deprotonation. It also remains liquid at low temperatures, which is important because LDA reactions are often performed at −78 °C (dry ice-acetone bath) to control reactivity and favor formation of the kinetic enolate (see below).
The Reactions of Enolates
The enolate intermediate is very nucleophilic and undergoes a variety of reactions, including alkylations:
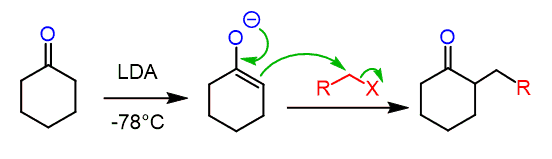
Like any other SN2 reaction, the alkylation of enolates works best with 1o, 1o benzylic, and 1o allylic substrates. Secondary and tertiary substrates would mostly undergo an E2 elimination, forming an alkene.
The alkylation works also with other functional groups such as esters, nitriles, and nitro compounds since they also have an acidic ɑ position:
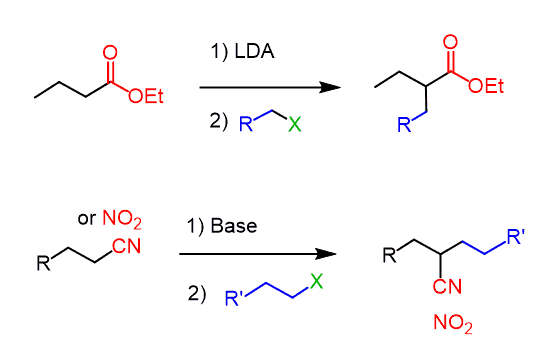
Aldehydes, on the other hand, are problematic for alkylation because of the competing aldol condensation.
The Regiochemistry of Enolate Alkylation
Unsymmetrical ketones can be alkylated on both sides, depending on the base and the temperature. Alkylation of the less substituted carbon is achieved by using a sterically hindered base, and, as mentioned earlier, LDA is, by far, the most common base you are going to see being used for this purpose. The more substituted carbon is usually deprotonated by sodium hydride:
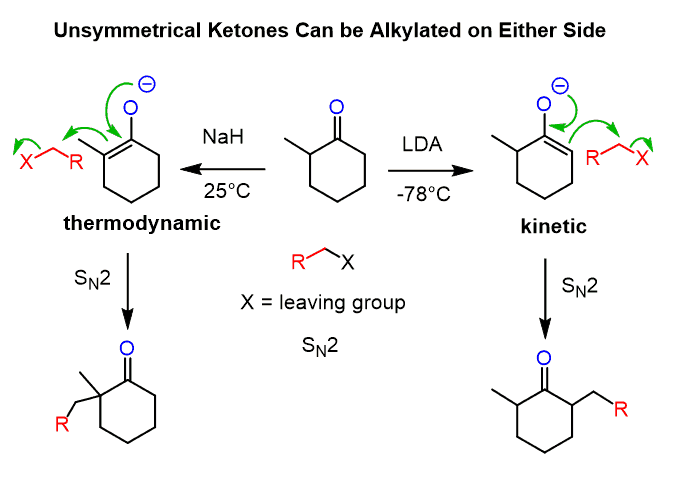
The less substituted enolate is the kinetic product (red pathway below), as seen from the lower activation energy (Ea) and therefore occurs faster.
The thermodynamic enolate (blue pathway) is the more stable enolate because of the more substituted C=C double bond.
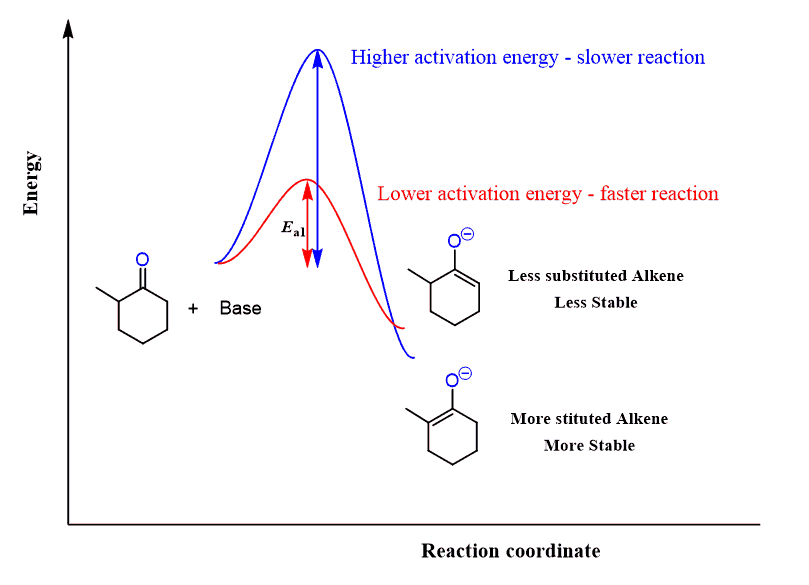
For a symmetrical ketone, it wouldn’t matter which of these bases is used.
The only thing is to make sure a strong base is used. Sodium hydroxide and ethoxides don’t work here because the enolate is not formed irreversibly, and self-condensation reactions can occur because there is still a lot of carbonyl present in the equilibrium mixture. This is especially true for aldehydes, which tend to undergo aldol condensation a lot faster than ketones do.
And, as always, if you are ready to test your skills, here is a good set of comprehensive problems on alpha carbon chemistry and also a separate one for the alkylation of enolates:
Enolate Alkylation Practice Problems
Enolates in Organic Synthesis – a Comprehensive Practice Problem
Check Also
- Alpha Halogenation of Enols and Enolates
- The Haloform and Iodoform Reactions
- Alpha Halogenation of Carboxylic Acids
- Alpha Halogenation of Enols and Enolates Practice Problems
- Aldol Reaction – Principles and Mechanism
- Aldol Condensation – Dehydration of Aldol Addition Product
- Intramolecular Aldol Reactions
- Aldol Addition and Condensation Reactions – Practice Problems
- Crossed Aldol And Directed Aldol Reactions
- Crossed Aldol Condensation Practice Problems
- Alkylation of Enolates Alpha Position
- Enolate Alkylation Practice Problems
- Acetoacetic Ester Synthesis
- Acetoacetic Ester Enolates Practice Problems
- Malonic Ester Synthesis
- Michael Reaction: The Conjugate Addition of Enolates
- Robinson Annulation, Shortcut, and Retrosynthesis
- Claisen Condensation
- Dieckmann Condensation – An Intramolecular Claisen Reaction
- Crossed Claisen and Claisen Variation Reactions
- Claisen Condensation Practice Problems
- Stork Enamine Synthesis
- Enolates in Organic Synthesis – a Comprehensive Practice Problem
