Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons, giving them different atomic masses. The keyword here is “mass,” because we are discussing mass spectrometry.
In a practical sense, how are isotopes related to mass spectrometry, and what do we need to know when solving MS problems?
Although their role is crucial in mass spectrometry and its applications across different fields, we are mainly concerned with the isotopic patterns of two elements: Chlorine and Bromine. The reason is that their heavy isotopes are much more abundant than those of most other elements in organic chemistry. The ³⁷Cl isotope makes up 25% of all chlorine atoms, and ⁸¹Br constitutes 50% of bromine atoms. So the ratio with their lighter isotopes is: ³⁵Cl (75%) to ³⁷Cl (25%), and ⁷⁹Br (50%) to ⁸¹Br (50%), respectively.
This significant contribution means that the molecular ion peaks of compounds containing Cl and Br appear with approximate ratios of 3:1 and 1:1, respectively.
Consider, for example, the mass spectra of chlorobenzene and bromobenzene:
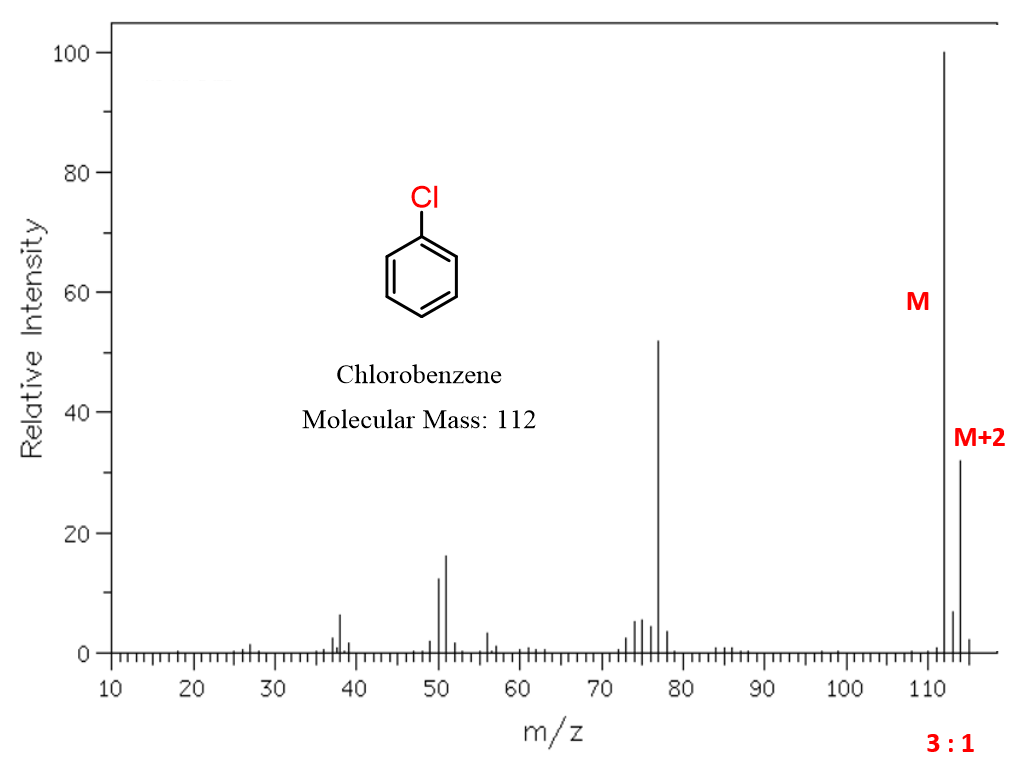

In the mass spectrum of chlorobenzene, we have two prominent molecular ion peaks separated by 2 m/z units: the M peak at m/z 112 corresponds to the molecule containing ³⁵Cl, while the M+2 peak at m/z 114 corresponds to the molecule containing ³⁷Cl. The relative intensities of these peaks are approximately 3:1, reflecting the natural abundance of the chlorine isotopes.
For bromobenzene, the molecular ion peaks are also separated by 2 m/z units, but here the M peak at m/z 156 (⁷⁹Br) and the M+2 peak at m/z 158 (⁸¹Br) appear with roughly equal intensity (1:1). This is a signature feature of bromine in mass spectra and makes it easy to identify.
Notice that the base peak in the mass spectrum of bromobenzene is not from the molecular ion. This is due to fragmentations, which we discuss extensively in the following two posts available to our registered students:
Compounds with 2 Cl and Br Atoms
Although not as often, you will also encounter mass spectra of molecules containing two or more atoms of chlorine and bromine. In these cases, the number of peaks and their ratios change from the usual 3:1 and 1:1 patterns.
Here is a small chart to remember the isotopic patterns of Cl and Br. Notice that when there are two Br atoms in the molecule, we get three peaks, and the intensity ratio changes to 1:2:1. This is less common, but you may definitely encounter it in mass spectrometry problems.

Summarizing the Isotopic Patterns in Mass Spectrometry
Isotopic patterns are extremely useful when working on practice problems to identify unknown compounds. In organic chemistry, we focus on the peak ratios of chlorine- and bromine-containing molecules. Remember: the presence of a 3:1 M/M+2 peak immediately suggests a chlorine atom, while a 1:1 pattern points to bromine.
Check also our detailed Guide on Solving Mass Spectrometry Problems that includes the key McLafferty fragmentations of aldehydes, ketones, esters, acids, and nitriles. Once you master the key fragmentation, work on the comprehensive Mass Spectrometry Problems.