In the previous post, we discussed some strategies for converting aldehydes to ketones. So, let’s now see how we can go the other way around to convert ketones to aldehydes.
Ketones to Aldehydes via Reduction of Esters
One way to convert a ketone to an aldehyde is first oxidizing it to an ester and reducing the latter to an aldehyde using DIBAL.

The reduction of festers to aldehydes is sometimes more challenging than it looks like, so if that does not work, we can reduce the ester to alcohol and oxidize it back to the aldehyde using a mild oxidizing agent such as PCC.
We can oxidize the ketone to ester by Baeyer–Villiger oxidation also known as Baeyer–Villiger rearrangement.

Ketones to Aldehydes via Oxidative Cleavage of Alkenes
Another strategy for converting ketones to aldehydes can be their conversion to akenes followed by an oxidative cleavage of the double bond. We can first reduce the ketone to a secondary alcohol, and dehydrate the alcohol to the Zaitsev or Hofmann alkene. The alkene can be cleaved by ozonolysis or KMnO4 to an aldehyde and a ketone depending on the structure of the double bond:

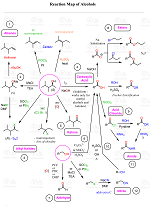
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Nomenclature of Aldehydes and Ketones
- How to Name a Compound with Multiple Functional Groups
- Preparation of Aldehydes and Ketones
- Nucleophilic Addition to Carbonyl Groups
- The Addition-Elimination Mechanism
- Reduction of Carbonyl Compounds by Hydride Ion
- Reactions of Aldehydes and Ketones with Water
- Reactions of Aldehydes and Ketones with Alcohols: Acetals and Hemiacetals
- Acetals as Protecting Groups for Aldehydes and Ketones
- Imines from Aldehydes and Ketones with Primary Amines
- Enamines from Aldehydes and Ketones with Secondary Amines
- Reactions of Aldehydes and Ketones with Amines-Practice Problems
- Acetal Hydrolysis Mechanism
- Imine and Enamine Hydrolysis Mechanism
- Reaction of Aldehydes and Ketones with CN Cyanohydrin Formation
- Hydrolysis of Acetals, Imines and Enamines-Practice Problems
- The Wittig Reaction: Examples and Mechanism
- The Wittig Reaction-Practice Problems
- Aldehydes and Ketones Reactions Practice Quiz