The McLafferty rearrangement is one of the most important named fragmentations you’ll come across in mass spectrometry. It is a rearrangement fragmentation that occurs in compounds containing a carbonyl group (C=O) and a hydrogen atom located three carbons away (γ-hydrogen). The fragmentation takes place via a six-membered cyclic transition state, generating a neutral alkene and a stabilized radical cation containing the carbonyl group. Remember, the instrument detects only positively charged species, so the neutral alkene is not detected. Let’s see how, for example, octan-2-one undergoes the McLafferty rearrangement. The γ-hydrogen from the C-5 carbon is transferred to the carbonyl oxygen, and cleavage between C-2 and C-3 occurs. This produces a strong fragment ion at m/z 58, which is the enolic carbocation.

Here is another example of McLafferty rearrangement forming a peak in the mass spectrum of Isovaleric acid:
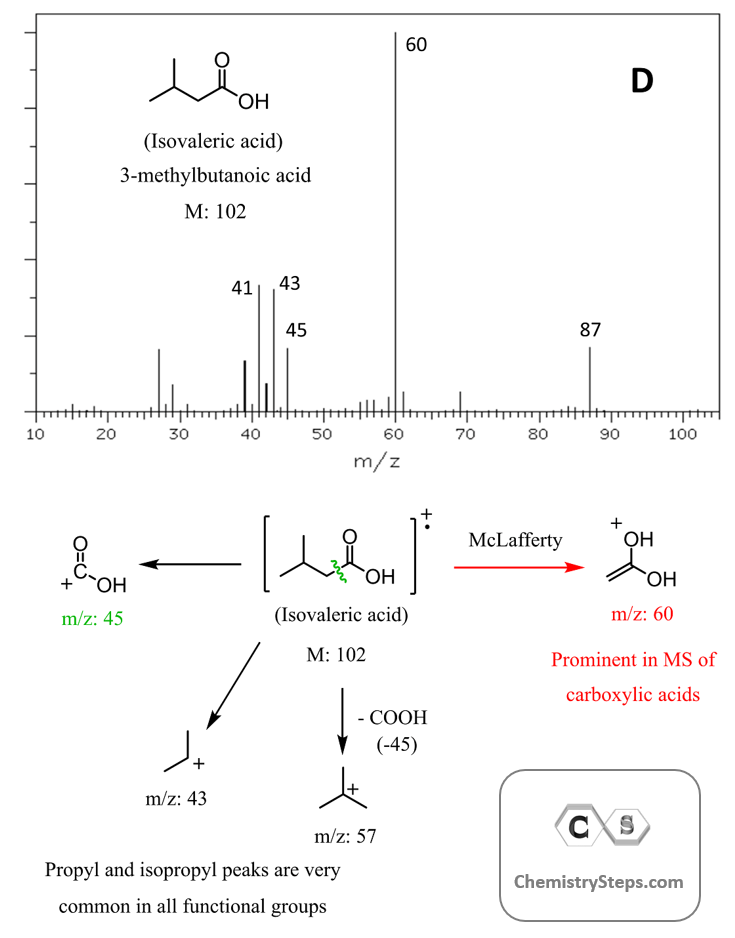
It’s important to remember that not all carbonyl compounds undergo McLafferty rearrangement because it requires the presence of a γ-hydrogen. If that hydrogen is missing, the rearrangement can’t happen.
Alpha Cleavage
Although the McLafferty fragment is the base peak, there is another prominent peak at m/z 43. This comes from a very common species known as acylium ion (CH₃–C≡O⁺), which is formed via α-cleavage:

The alpha cleavage occurs by breaking the bond between the alpha and beta carbons. It is a highly favored process because the resulting carbocation is stabilized by the lone pair of electrons on the heteroatom.
This includes a large number of functional groups, such as aldehydes, ketones, esters, alcohols, and amines. The role of the heteroatom is the stabilization of the carbocation by its lone pair of electrons.
For example, in the mass spectrum of 2-pentanol, the molecular ion peak is barely visible, while the base peak appears at m/z 45.

Notice that the alpha cleavage occurs on both sides of the carbonyl, and two resonance-stabilized carbocations are formed.
Summarizing McLafferty Rearmament
In summary, the McLafferty rearrangement is a key fragmentation in mass spectrometry that occurs mainly in carbonyl-containing compounds with a γ-hydrogen. It proceeds through a six-membered cyclic transition state, producing a neutral alkene and a stabilized radical cation, which is detected by the instrument.
Understanding both McLafferty and α-cleavage patterns is essential for interpreting mass spectra accurately, so keep these patterns in mind:
- Requires a carbonyl group and a γ-hydrogen (three carbons away).
- Proceeds through a six-membered cyclic transition state.
- Produces a neutral alkene (not detected) and a stabilized radical cation (detected).
- The fragment ion mass (e.g., m/z 58 in octan-2-one) depends on what is attached to the α-carbon.
- If no γ-hydrogen is available, McLafferty cannot occur.
Check for α-cleavage fragments, as this is also characteristic of oxygen- and nitrogen-containing molecules.
Check out our detailed Guide on Solving Mass Spectrometry Problems that includes the key McLafferty fragmentations of aldehydes, ketones, esters, acids, and nitriles. Once you master the key fragmentation, work on the comprehensive Mass Spectrometry Problems.
 IR, MS, and NMR Combined Problems
IR, MS, and NMR Combined Problems

Draw the McLafferty rearrangement mechanisms for the molecular ion of hexyl hexanoate that will result in the formation of different fragment ions