There are several ways to prepare esters, and in this lesson, we’ll go over the most common ones you’ll see in organic chemistry. The most famous is the Fischer esterification, where a carboxylic acid is reacted with an alcohol under acidic conditions and elevated temperature. However, this reaction has some limitations, and for that reason, we also use other approaches for preparing esters.
Fischer Esterification
The Fischer esterification is the acid-catalyzed reaction of a carboxylic acid with an alcohol to make an ester and water:

The Mechanism of Fischer Esterification
Mechanistically, it’s an example of a nucleophilic addition-elimination reaction at the carbonyl carbon.
In the first step, the carbonyl oxygen is protonated, which makes the carbonyl carbon more electrophilic. The alcohol then attacks this carbon, and after a few proton transfers, a water molecule leaves, giving the ester.

One way of confirming this mechanism is the fact that the oxygen atom in the OR group of the ester comes from the alcohol. For this, an ¹⁸O-labeled alcohol is used, and this oxygen appears in the structure of the ester:

Reaction Conditions of Fischer Esterification
Every step in Fischer esterification is reversible, so we need to push the equilibrium toward the ester side. There are two common ways to do this:
- Use a large excess of the alcohol, or
- Remove the water as it forms (for example, using a Dean-Stark apparatus).
We normally run this reaction under reflux with a strong acid catalyst such as sulfuric or phosphoric acid. When dissolved in the alcohol, these acids form the conjugate acid of the alcohol, which is actually the true catalytic species.
Limitations of Fischer Esterification
The Fischer esterification works great in many cases, but not for all. There are a few important limitations to keep in mind.
- Some molecules simply cannot tolerate the strongly acidic or high-temperature conditions used in this reaction. For example, secondary and especially tertiary alcohols tend to dehydrate in acidic conditions rather than forming esters.

- Phenols (aryl alcohols) are poor nucleophiles because their lone pairs are delocalized into the aromatic ring, making them less reactive toward the carbonyl.

When you run into these limitations, there are alternative ways to make esters that work under milder or different conditions.
Alternative Methods for Making Esters
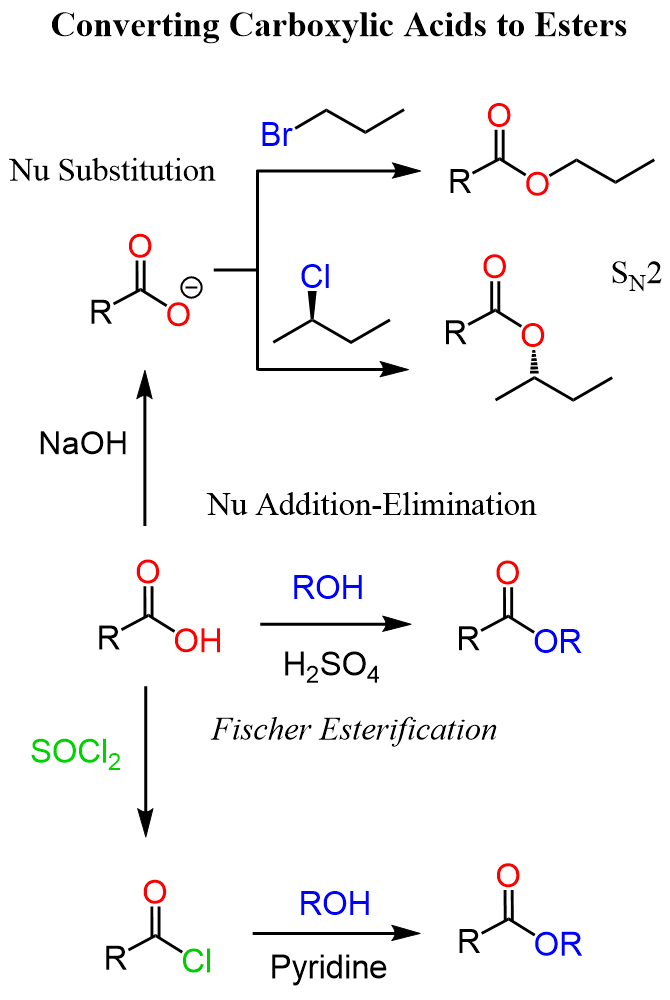
A good alternative to Fischer esterification is the use of acid chlorides, which can be prepared from carboxylic acids by chlorinating agents such as SOCl2, PCl3, and PCl5.
Remember, acid chlorides are the most reactive derivatives of carboxylic acids, and they require mild conditions for nucleophilic addition–elimination substitutions. In fact, most of these reactions are difficult to prevent, let alone apply forcing conditions.
For example, phenol can be reacted with an acid chloride to prepare the corresponding ester.

The reaction is carried out under basic conditions, and this may be more suitable for certain functional groups. This method is applicable to aromatic and aliphatic alcohols:

Ester from Carboxylate Salts (SN2 Reaction)
Another approach is to first deprotonate the carboxylic acid to make a carboxylate ion. This converts a poor nucleophile into a strong one.
The carboxylate then reacts with an alkyl halide in an SN2 substitution to give the ester:

Intramolecular Fischer Esterification – Forming Lactones
If the molecule has both a hydroxyl and a carboxylic acid group, it can undergo intramolecular Fischer esterification to form cyclic esters, also known as lactones.
Depending on the position of the –OH group relative to the –COOH, we can form:
- β-lactones – 4-membered rings
- γ-lactones – 5-membered rings
- δ-lactones – 6-membered rings
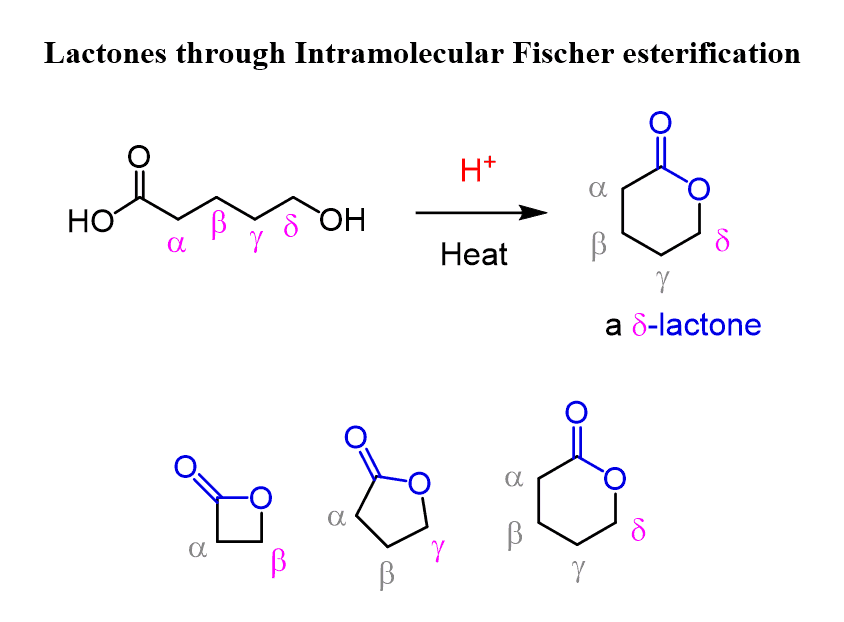
Five- and six-membered lactones form most readily because these ring sizes have the least strain.
Preparation of Esters: Summary
As we’ve seen, there are several ways to make esters depending on the structure of your starting materials and the reaction conditions you can work with. The Fischer esterification is the classic method, which works great for many carboxylic acids and alcohols, but it does have its limits, especially with bulky or acid-sensitive substrates.
When those conditions aren’t suitable, we can switch to more reactive acid chlorides, which form esters easily under milder, often basic, conditions. We can also make esters from carboxylate salts and alkyl halides via an SN2 reaction, or even form lactones through intramolecular esterification when both the –OH and –COOH groups are in the same molecule.
In the end, let’s summarize the strategies for converting alcohols to esters in one synthetic scheme:
Check Also
- Preparation of Carboxylic Acids
- Naming Carboxylic Acids
- Naming Nitriles
- Naming Esters
- Naming Carboxylic Acid Derivatives – Practice Problems
- The Addition-Elimination Mechanism
- Fischer Esterification
- Ester Hydrolysis by Acid and Base-Catalyzed Hydrolysis
- What is Transesterification?
- Esters Reaction with Amines – The Aminolysis Mechanism
- Ester Reactions Summary and Practice Problems
- Preparation of Acyl (Acid) Chlorides (ROCl)
- Reactions of Acid Chlorides (ROCl) with Nucleophiles
- R2CuLi Organocuprates – Gilman Reagent
- Reaction of Acyl Chlorides with Grignard and Gilman (Organocuprate) Reagents
- Reduction of Acyl Chlorides by LiAlH4, NaBH4, and LiAl(OtBu)3H
- Reduction of Carboxylic Acids and Their Derivatives
- Preparation and Reaction Mechanism of Carboxylic Anhydrides
- Amides – Structure and Reactivity
- Naming Amides
- Amides Hydrolysis: Acid and Base-Catalyzed Mechanism
- Amide Dehydration Mechanism by SOCl2, POCl3, and P2O5
- Amide Reduction Mechanism by LiAlH4
- Reduction of Amides to Amines and Aldehydes
- Amides Preparation and Reactions Summary
- Amides from Carboxylic Acids-DCC and EDC Coupling
- The Mechanism of Nitrile Hydrolysis To Carboxylic Acid
- Nitrile Reduction Mechanism with LiAlH4 and DIBAL to Amine or Aldehyde
- The Mechanism of Grignard and Organolithium Reactions with Nitriles
- The Reactions of Nitriles
- Converting Nitriles to Amides
- Carboxylic Acids to Ketones
- Esters to Ketones
- Carboxylic Acids and Their Derivatives Practice Problems
- Carboxylic Acids and Their Derivatives Quiz
- Reactions Map of Carboxylic Acid Derivatives
