 In this review, we will try to summarize what topics and types of questions from organic chemistry are covered in the MCAT test. Organic chemistry is tested as part of the Biological and Biochemical Foundations of Living Systems Section. You will have 95 minutes to answer 59 questions in a multiple-choice format. This format might make it a little easier since the given options can also serve as hints to find the correct answer. The questions include topics from general chemistry, organic chemistry, biology, and biochemistry.
In this review, we will try to summarize what topics and types of questions from organic chemistry are covered in the MCAT test. Organic chemistry is tested as part of the Biological and Biochemical Foundations of Living Systems Section. You will have 95 minutes to answer 59 questions in a multiple-choice format. This format might make it a little easier since the given options can also serve as hints to find the correct answer. The questions include topics from general chemistry, organic chemistry, biology, and biochemistry.
The focus of our discussion is on organic chemistry, so let’s look at it in a little more detail.
Although percentwise only 5% of the questions are on organic chemistry, leaving much of those to biology (65%) and biochemistry (25%), the role of organic chemistry is not to be underestimated, at least for two main reasons. First, it is on the basis of biochemistry, and a lot of questions are interdisciplinary, requiring knowledge of related fields. Second, it is the importance of understanding organic chemistry which will likely be considered for your admission to a medical school, despite the number of questions in the test.
You will have a good idea of the topics listed below if you have already taken Organic Chemistry 1 and 2; however, if it’s been a while or you are still to take these courses, check the links to the blog post linked in this article. Chemistry Steps is focused on college-level organic chemistry, for which there are practice problems, multiple-choice quizzes, and study guides, which can definitely be a little help in preparing for MCAT. To access all of these, you will need a Prime membership. There are thousands of registered students from the US and all over the world, and this helps me write more content while working full-time and without any sponsors to pay for the hosting. The articles and practice problems are free to everyone! The content, however, is not created to specifically fit the official MCAT format and topics.
So, let’s put this together, and if you have taken or are planning to take the MCAT, let me know in the comments what should be changed or modified to better help everyone in this journey.
Structure and Bonding in MCAT
The first thing you need to have set in place for the MCAT organic chemistry is a solid foundation of structure and bonding from general chemistry. This includes understanding Lewis structures, bond-line structures, converting between those, and understanding formal charges. Formal charges are also important for being able to draw resonance structures and determine which is the most stable.
For example, when comparing resonance structures, keep in mind these two principles:
Principle 1. Atoms, in general, “don’t like” charges, so having no charge is better:

Sometimes, it is impossible to avoid charges, so if both resonance structures are charged, then the octet rule needs to be considered.
Principle 2. The resonance structure with a complete octet is more stable:

The conversion between resonance structures and most organic reactions is shown with curved arrows, so be sure to understand how they work. The main idea here is to remember that curved arrows show the movement of electrons and not show arrows starting from a neutral atom or a positive charge.

Note that resonance structures are not isomers, and they only differ in the electron distribution among the atoms. All the atoms are connected the same way, and therefore, resonance structures are shown with double-headed arrows rather than two arrows, which are used for equilibrium.
Another important concept is the types of bonding and the ideas behind classifying them. For example, how do we differentiate between polar, nonpolar, and ionic bonds? For this, we need to remember about electronegativity and how its difference affects the electron distribution between atoms. The imbalance of electron distribution results in a dipole moment, and you need to first be able to recognize dipole moments and know their basic characteristics:

Now, if there are two dipole moments in the molecule, we need to find the vector sum of these dipoles to determine the overall dipole of the molecule:

There may be questions where the molecule is not drawn, or the geometry is not given, and you are asked to determine whether the given molecule is polar or not.
To answer this question, you need to know how to determine the common molecular geometries and shapes of the molecule following the rules of the VSEPR theory. These are summarized in the following table, but are also covered in detail here:

Depending on the bonds in the molecule, MCAT questions may also require you to identify the types of intermolecular interactions, such as hydrogen-bonding, dipole-dipole interactions, and Van der Waals forces.
These interactions determine the physical properties like boiling and melting point and, in general, the stronger the intermolecular interactions, the higher the melting and boiling points are.
It will also be a good idea to remember the relative order of intermolecular interactions, in which ionic bonds > hydrogen bonding > dipole-dipole interactions > Van der Waals forces:

Bond Length and Bond Strength
The next step in mastering the concepts of chemical bonding is being able to predict the relative bond lengths and bond strengths. The key principle to remember is that the shorter the bond, the stronger it is.
For example, the bond strength of the C-H bond of alkane, alkene, and alkyne follows the pattern shown below:

The table suggests that the bonds between sp-hybridized carbon atoms are the shortest and therefore the strongest. This is explained by the s character of the bonding orbital, which, in turn, is explained by the hybridization state of the carbon atom.
IUPAC nomenclature and MCAT
You will likely get questions on determining the name based on the structure or coming up with a structure given the name. Even though the questions are multiple-choice, knowing the basic IUPAC rules for naming alkanes, alkenes, and alkynes is a must.
There might be questions on naming compounds with multiple functional groups and the first thing, of course, is to know the functional groups and then their priority in the IUPAC chart. Once this is done, you need to identify the functional group with the highest priority and add a suffix (ending) of that functional group. The other groups are treated as substituents and added to the name with prefixes:

Organic Acids and Bases in MCAT
First, be sure to know the definition of acids and bases so that you can recognize common organic acids and bases as well as conjugate acids and bases. Aside from recognizing acids and bases, you also need to learn about their qualitative and quantitative description by having a good understanding of pKa, its approximate values for common functional groups:

Another important skill set is being able to determine the direction of equilibrium of an acid-base reaction based on the pKa values of the acid and conjugate acid.
Some questions will require an understanding of the factors that determine the acid strength, such as the atom bonded to the acidic proton, the resonance effect of stabilizing the conjugate base, the inductive effect, and the hybridization of atomic orbitals. This is also known as the ARIO (atom, resonance, induction, orbital).
Alkanes and Cycloalkanes
Alkanes contain single bonds, and the free rotation about the sigma bonds allows for adopting different conformations. The conformation of alkanes can be shown in bond-line, Newman, Haworth, and Fischer projections.

You need to understand all these representations and be able to convert between them. Questions on Newman projections may also require identifying staggered, eclipsed, gauche, and anti-conformations, their relative stability, as well as rotating about σ-bonds to convert between these conformations.
For cycloalkanes, you need to be able to draw chair conformations correctly, assigning the substituents as axial and equatorial, recognize 1,3-diaxial interactions, perform a ring flip, and identify the more stable chair conformation.
Stereochemistry and Isomerism
There are different types of isomers divided into three main groups: constitutional/regioisomers (different connectivity of atoms), conformational (rotation about single bonds including ring-flips), which we discussed above, and stereoisomers (different spatial arrangement of atoms.
It is a very common question on MCAT and organic chemistry in genera,l asking you to identify the relationship between two compounds.
Here is a quick chart for determining the relationship between two molecules:

Keep in mind that isomers must have the same chemical formula, so when you are not certain, first check if this is the case. For large molecules, it might be tedious to count all the atoms, and to make it a little easier, count the carbons and heteroatoms, and determine the degree of unsaturation instead of also counting all the hydrogen atoms. Check this post for more details and examples, as understanding the degree of unsaturation or hydrogen deficiency index (HDI) is very important in IR and NMR spectroscopy problems as well.
Stereoisomers are divided into enantiomers and diastereomers. Remember, enantiomers are nonsuperimposable mirror images:

Any stereoisomers that are not mirror images are classified as diastereomers. You can always rely on determining the R and S configuration of chiral centers to determine whether the molecules are enantiomers or diastereomers. All the chiral centers are inverted in enantiomers, while diastereomers have at least one chiral center where the R, S configuration is the same and one where it is inverted. In other words, in diastereomers, some of the chiral centers are the same, and some are different:
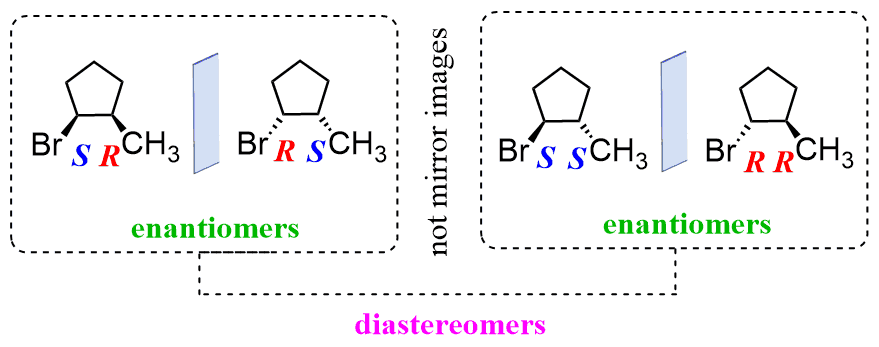
It is also important to be able to determine the R and S configuration on Newman and Fischer projections.
You also need to be able to relate the optical activity to chirality and enantiomers. Chiral molecules are optically active when it is not a mixture of enantiomers in a 1:1 ratio. These are called racemic mixtures, and they are optically inactive and therefore, achiral.
When one of the enantiomers is present in larger amounts, we quantify their difference by enantiomeric excess. This can be determined from the optical rotation of the solution.
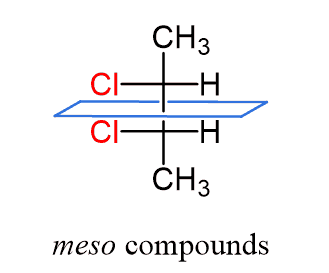
Meso Compounds
Keep in mind that not every compound with chiral centers is chiral. If the molecule has a plane of symmetry, it is a meso compound, and those are not chiral.
One more class of diastereomers is the cis and trans alkenes, as these are also stereoisomers that are not mirror images. Remember, cis and trans stereoisomerism depend on whether two identical alkyl groups on the c=c bond are on the same or opposite sides of that double bond:

If the two groups on the double bond are not identical, we cannot classify these as cis or trans, and the e and z notation is used:

Nucleophilic Substitution Reactions in MCAT
Together with acid-base chemistry, these are the first types of reactions in organic chemistry, and it is important to understand how the reactive species are classified. You need to be able to recognize the nucleophile, electrophile, and the leaving group in a substitution reaction. Read about polarity, partial charges, alkyl halides, and then start with the introduction to nucleophilic substitution reactions.

Generally speaking, weak bases are good leaving groups and strong bases are good nucleophiles. However, there are some important exceptions, and to get to all of them, you need to read the corresponding topics.
There are two main mechanisms in nucleophilic substitution reactions that you need to distinguish.
The first is the SN1 (substitution nucleophilic unimolecular) and the second is the SN2 (substitution nucleophilic bimolecular). The unimolecular and bimolecular refer to the order of these reactions, so SN1 is a first-order reaction, and SN2 is a second-order reaction.
A good understanding of these reactions implies being able to draw/determine the correct curved arrow mechanism, recognize the reaction from the transition state, an intermediate or its absence, from the product and especially the stereochemistry of it, and in general deciding if it is an SN1 or SN2 mechanism.
Some of the key points needed to distinguish them are:
- SN2 is a concerted mechanism – no intermediate carbocations.
- SN1 is a stepwise mechanism forming an intermediate carbocation before the nucleophilic attack.
- SN2 reactions proceed with inversion of configuration around this chirality center.
- SN1 reactions result in the racemization of the reacting center.
- Primary and methyl substrates favor the SN2 mechanism.
- Bulky substrates favor the SN1.
- Good nucleophiles favor the SN2.
- Weak nucleophiles favor the SN1.
- Polar aprotic solvents favor the SN2 mechanism.
- Polar protic solvents favor the SN1 mechanism.
Our Study Guides include a three-page summary of Nucleophilic Substitution and Elimination reactions you can use to prepare for the MCAT test.
Alcohols and MCAT
The first is to recognize the functional group and the specific prototiles, such as hydrogen bonding, that are associated with it. Next is the understanding of the nomenclature and important reactions of alcohols. This will mainly be the SN1 and SN2 substitution reactions, including conversion of alcohols to alkyl halides by HX acids, SOCl2, PBr3 as well as the preparation and use of mesylates and tosylates. Because of the relatively high acidity of the OH group, it often requires a protecting group, especially when strong bases and nucleophiles such as Grignard reagents are used.
Another important transformation of alcohols is their oxidation to aldehydes, ketones, and carboxylic acids. You will likely need to know the mild and strong oxidizing agents such as PCC, PDC, Dess Martin, Chromic acid, etc.
Aldehydes and Ketones in MCAT
The nomenclature and main reactions of aldehydes and ketones are tested on the MCAT exams. In general, aldehydes and ketones are the starting point of the very important class of nucleophilic addition reactions at the C=O bond. Practice these mechanisms and pay close attention to try to find a pattern by studying the formation and reactions of acetal, hemiacetals, imine, and enamines.
Another type of transformation is the oxidation and reduction reactions. Although ketones can no longer be oxidized like aldehydes, both can be reduced to alcohols by a variety if reagents such as LiAlH4, NaBH4, DIBAL, etc.
Other types of transformations, such as elongating the carbon chain of aldehydes and ketones, become possible by their reaction with cyanides, forming cyanohydrins. The nitrile group can then be reduced or hydrolyzed and thus converted to new functional groups such as amines and carboxylic acid,s which we will discuss next.
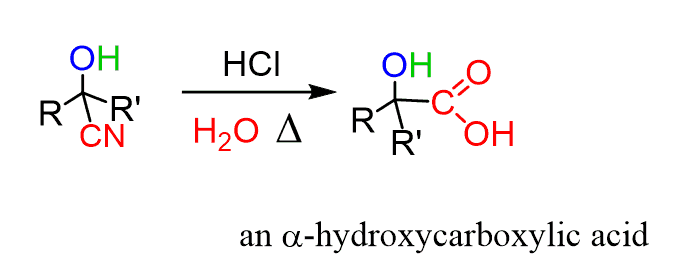
Carboxylic Acids and their Derivatives in MCAT
The questions on carboxylic acids and their derivatives, such as esters, amides, anhydrides, and acid halides, are focused on the description, nomenclature, physical properties, and the main reactions. This will require knowing the Fischer esterification, including lactones, transesterification, formation of amides, including lactams, the reduction of the carboxyl group to an alcohol, and decarboxylation of β-keto esters.
Be sure to go over the isotope effect in the esterification, transesterification, and hydrolysis reactions mechanisms, as it helps to grasp the concept of the addition-elimination to the carbonyl group.

It is also important to understand the pattern of nucleophilic addition-elimination reactions and the relative reactivities based on the leaving group ability. For example, esters can be converted to amides more easily than the reverse reaction since the conjugate base of an ester is a weaker base than that of an amine.
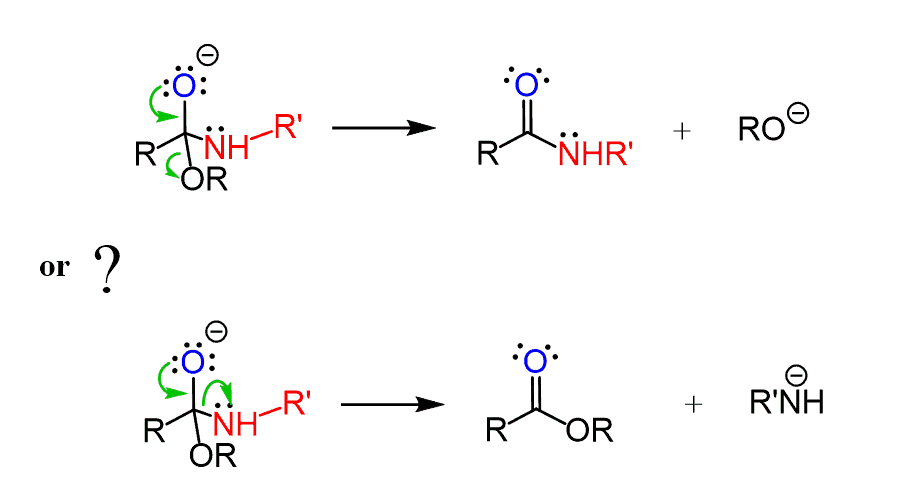
Comparing the pKa values of alcohols (16) and amines (38), we know that alkoxy groups are much weaker bases and therefore better leaving groups than a conjugate base of an amine. So, when the lone pairs on the oxygen move down to restore the C=O bond, the alkoxy group is kicked out, producing an amide.
Although I couldn’t find specifically in the AAMC 2020 official guide of the MCAT exam content, some books do mention that the reaction of Grignard reagents with esters and acid chlorides does occur in the tests. You can share your experience or what you know about this in the comments below.
Enolates and Alpha Carbon Chemistry in MCAT
First, you need to be able to recognize the alpha carbon and its acidity caused by the electron-withdrawing carbonyl group. The alpha carbon chemistry is different from the chemistry of the C=O group, which is the electrophile in the nucleophilic addition, while here the deprotonated alpha carbon is the nucleophile.
This deprotonation forms an ion called an enolate, which is a good nucleophile capable of halogenation, alkylation, and a number of aldol reactions.
Unsymmetrical ketones can be alkylated on both sides, depending on the base and the temperature. Alkylation of the less substituted carbon is achieved by using a sterically hindered base, and LDA is, by far, the most common base you are going to see being used for this purpose. The more substituted carbon is usually deprotonated by sodium hydride:
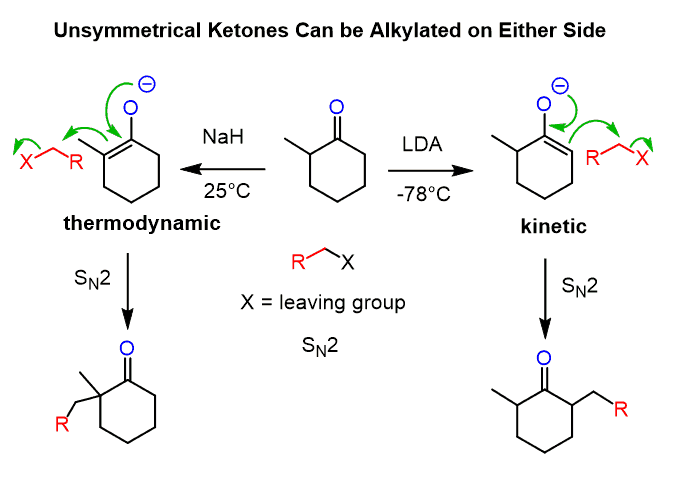
The less substituted enolate is the kinetic product (red pathway below) as seen from the lower activation energy (Ea) and therefore occurs faster.
The thermodynamic enolate (blue pathway) is the more stable enolate because of the more substituted C=C double bond.
A few common reactions of enolates for forming a new C-C bond are the aldol reaction, crossed aldol reaction, Claisen condensation, acetoacetic ester synthesis, malonic ester synthesis, etc.
Carbohydrates in MCAT
Some of the key concepts and reactions of carbohydrates on MCAT.
The Structure and Classification of Carbohydrates
The first thing here would be going over the structure and classification of common monosaccharides in order to recognize and draw them. There is a lot, and knowing all of them is an overwhelming task; however, try to focus on the most important ones, such as glucose, ribose, fructose, mannose, and galactose.
In a more general scope, remember that carbohydrates are divided into aldoses and ketoses depending on whether the C-1 carbon is an aldehyde or a ketone group.

In addition to monosaccharides, there are some important disaccharides, including sucrose, lactose, and maltose, that you will need to know. Remember that the single sugar units in di- and trisaccharides are linked by the glycosidic linkage
The Stereochemistry of Carbohydrates
You also need to understand the stereochemistry of cyclic and linear carbohydrates with a major emphasis on the D and L notation. While the D and L notations of the same sugar represent enantiomers since all the stereogenic centers are inverted, many common carbohydrates are also epimers. You may also be asked directly or in the context of different questions to distinguish epimers and anomers.
In short,
- Epimers are diastereomers that differ in the configuration of only one chiral center.
- Anomers are epimers specifically applied to characterize cyclic carbohydrates.
For example, D and L-Glucose are enantiomers while D-Glucose and D-mannose are epimers, and to specify, we can also say that they are epimeric at carbon-2.

Another important skill for the carbohydrates in MCAT tests is being able to convert between Fischer and Haworth Projections to a chair conformation and vice versa. In this operation, it is crucial to keep the stereochemistry of the chiral centers correct. The anomeric carbon can be formed in two configurations, alpha and beta, depending on the direction of the OH attack on the carbonyl group.
Common reactions of carbohydrate derivatives
The key reactions you will need to know are the oxidation-reduction reactions, the Kiliani-Fischer synthesis, and the Wolf degradation.
Oxidation of carbohydrates can be achieved using bromine and water to distinguish aldoses from ketoses, Tollens’ reagent (Ag+ NH3, HO−), Fehling’s reagent (Cu2+ in aqueous sodium tartrate), and Benedict’s reagent (Cu2+ in aqueous sodium citrate) for detecting carbohydrates, as well as the nitric acid oxidation, which converts the carbohydrate to an aldaric acid.

The presence of an aldehyde and ketone group allows for the reduction of carbohydrates using NaBH4, which produces alditols.
The Kiliani-Fischer synthesis and Wolf degradation are overall reverse transformations. The former is for increasing the carbohydrate chain, and the latter is for decreasing it by one carbon atom.

Spectroscopy and MCAT
You will need to have a good understanding of the basics in spectroscopic characterization, such as UV/vis, Infrared (IR), NMR spectroscopy, and Mass spectrometry. For the UV/vis, it is important to recognize the correlation between the absorption region and the color of the substance. Remember that the more conjugated systems absorb higher wavelengths and therefore, lower-energy light.
IR spectroscopy is a quick way of determining the functional groups present in the molecule. IR spectra are usually crowded with peaks, but the good news is that you don’t need to analyze them all. There are some important absorption regions, such as the 1700 cm-1 C=O region, the 3000-3300 region for the sp3, sp2, and sp C-H bonds, and knowing them will allow you to solve most IR problems of an undergraduate standardbred.
NMR spectroscopy is the most comprehensive technique for organic structure determination and can go from easy to grasp to as difficult as it gets for even professional chemists. Fortunately, there is nothing insane on MCAT regarding NMR spectroscopy; however, you need to know its basics, such as the chemical shift regions of the protons in common functional groups, integration, equivalent and nonequivalent protons, and spin-spin splitting based on the n+1 rule. Learning some tactics and common patterns for solving NMR problems is definitely a good time investment.
What else is on MCAT Organic Chemistry
This review of the topics on MCAT exams is not comprehensive and should not be used as an official guide. A few of the additional concepts are the laboratory techniques of separation and purification, such as extraction, distillation, chromatography, including the basic principles of paper chromatography, thin-layer chromatography (TLC), column chromatography, high-pressure liquid chromatography (HPLC), and electrophoresis.
Some concepts go beyond the material covered in standard organic chemistry courses, and you will need to bring up your skills in biology and biochemistry.
Let us know in the comments what to add. What advice do you have for those taking the MCAT test?

Please try to include materials and Practice problems on UV-Vis Spectroscopy and Mass spectrometry Sir.
Mass spectrometry problems have been added: https://www.chemistrysteps.com/solving-mass-spectrometry-practice-problems/, https://www.chemistrysteps.com/mass-spectrometry-practice-problems/