Why do you think the following Grignard reaction is impossible to carry out without having a protecting group on the alcohol?
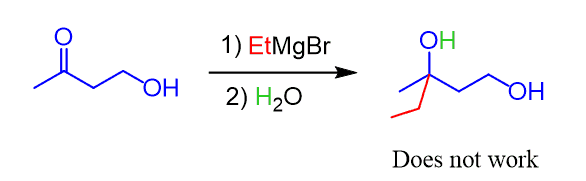
What is even a protecting group?
These are the questions we are going to address in today’s article.
Let’s, for a moment, forget about the protecting group and try to figure out what will happen if we mix the Grignard reagent with an alcohol:

Grignard reagents are excellent nucleophiles, but at the same time, they are also strong bases since there is an extremely polar C-Mg covalent bond, which leaves the carbon electron-rich.
Remember that acid-base reactions are among the fastest and will occur before substitution reactions. Now, with this said, can you identify a proton in the starting material that is acidic enough to be attacked by the ethylmagnesium bromide?

Looking at your pKa table (or better, without looking), you can see that the most acidic proton in this molecule is the alcohol (pKa = 16). And, even the α-hydrogens next to the carbonyl can be attacked by the Grignard reagent since they have a pKa of ~ 20. However, this reaction is slower, and the Grignard reagent first deprotonates the alcohol:
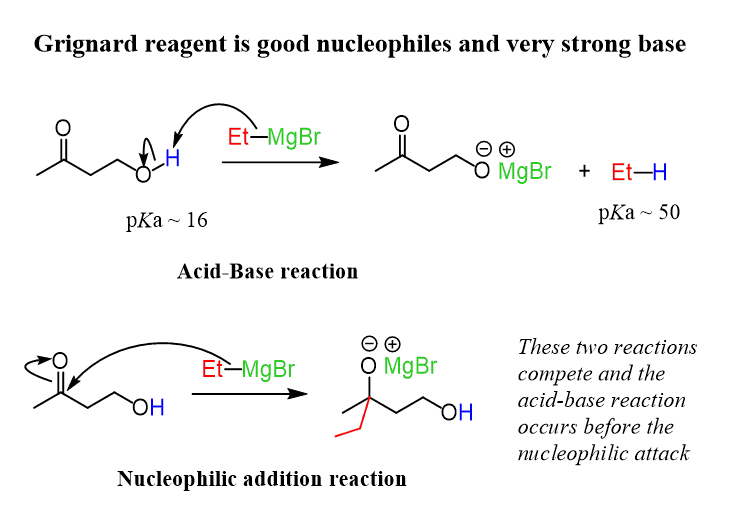
The acid-base reaction quenches the ethyl magnesium bromide, not allowing the desired Grignard reaction to take place. Therefore, remember that the Grignard reaction should be carried out in a “proton-free” environment, meaning no acidic functional groups and no protic solvents can be present.
And this is where the protecting groups become needed. A protecting group is a compound that temporarily converts a given functional group into another, allowing for performing reactions that are otherwise incompatible with that functional group.
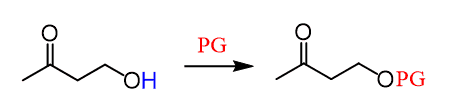
So, for our molecule, we need something to block the alcohol from the attack, perform the Grignard, and then remove this blocking unit at the end:
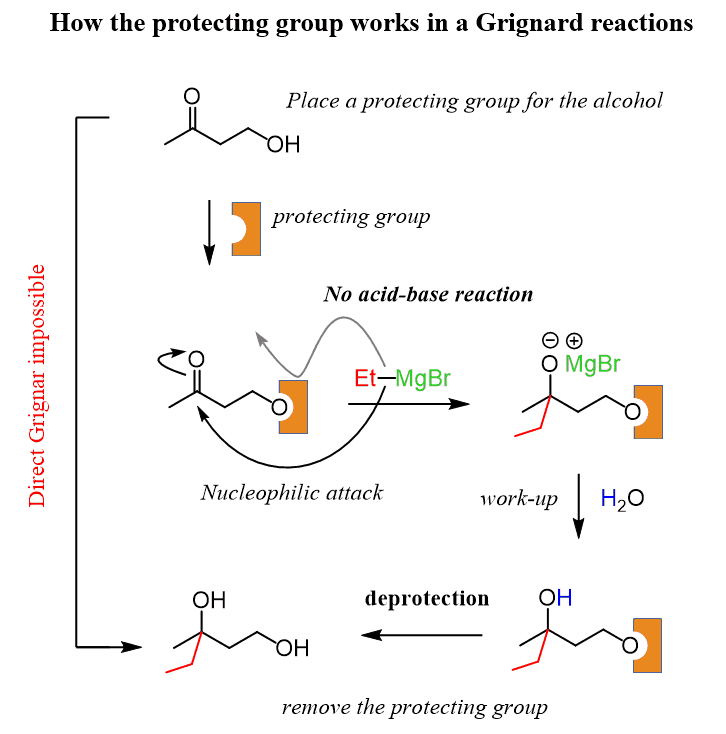
The term protecting group is usually abbreviated as “PG” in chemical structures:
The most common protecting groups for alcohols are the silyl ethers. Here is the idea behind it. We take a silyl chloride, do a substitution using the alcohol as a nucleophile, and then the alcohol converted into a silyl ether can be used in the presence of any strong base, including the Grignard reagent.
The reaction with alcohols is carried out in the presence of a base such as triethylamine (TEA) or Imidazole to deprotonate the oxygen:

Have you noticed the arrows indicating an SN2 mechanism in the first step of installing the protecting group? And do you remember that SN2 reactions are not possible with tertiary substrates? Well, that is true for the substrates with the leaving group on the carbon. However, silicon, being in the 3rd row of the periodic table, is larger, and the Si-C bond is longer than the C-C bond, which reduces the steric effects, making the SN2 mechanism possible on a tertiary atom:
![]()
The silyl ether protecting group is cleaved off with fluoride ion using tetrabutylammonium fluoride (TBAF) Bu4N+F–. The four butyl groups are installed to increase the solubility of the salt in organic solvents.

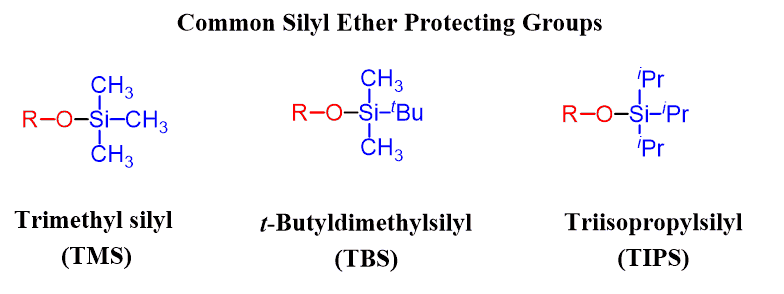
Different alkyl groups can be on the silicon, including the simplest Trimethylsilyl ether (TMS), tert-butyldimethylsilyl (TBS), and Triisopropylsilyl ether (TIPS):
Other Protecting Groups for Alcohols
You may wonder why not use a regular ether instead of a silyl ether. And that is a great question because alcohols can be converted into ether quite easily. The problem, however, is that it is difficult to cleave off the ether group. Ethers are quite stable, and in fact, they are used as a solvent for organolithium reactions. Even for the Grignard reaction lab, you will most likely use diethyl ether.
For example, in the following alkylation reaction of an acetylide ion, the OH groups must first be protected. In theory, we could use methyl iodide to convert the alcohol into a methyl ether. However, cleaving the ether later would require acidic conditions, which are not suitable for alkynes due to the risk of hydrohalogenation of the triple bond.

This is an important factor to consider since you don’t want your protecting group to be labile under certain conditions, but, on the other hand, you need to be able to get it off when the reaction is finished.
A good alternative to the silyl ethers with such qualities is the group called tetrahydropyranyl (THP). It forms a tetrahydropyranyl ether (an acetal), which is stable under basic conditions but can be cleaved with an acid.

The reaction starts by activating the dihydropyran ring, which is then attacked by the alcohol:
Acetals are common protecting groups for aldehydes and ketones as well. You can read more about them here.
Last but not least, benzyl (Bn) ethers represent another common way of protecting the alcohol functional group.
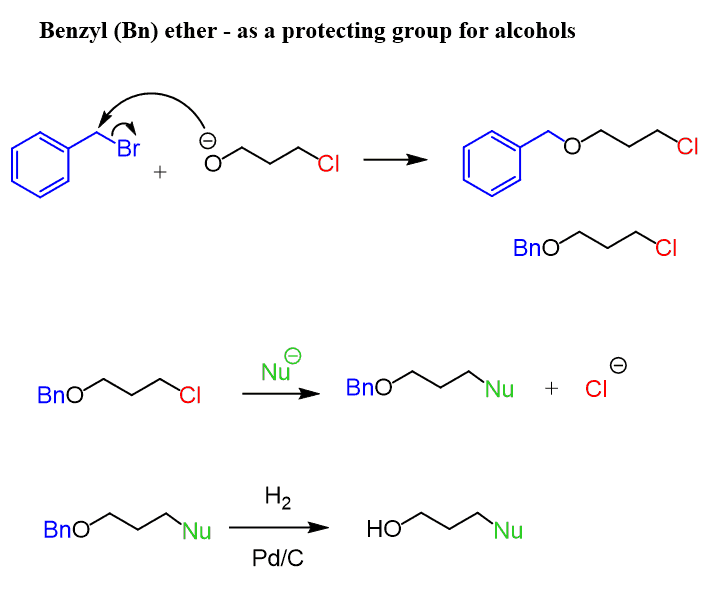
The benzyl group is usually stable under acidic and basic conditions and is cleaved by catalytic hydrogenation with H2 over Pd/C. And because of this, it is not suitable for many reactions that involve a double or a triple bond.

Check this 66-question, Multiple-Choice Quiz with a 2-hour Video Solution on the naming, preparation, and reactions of Alcohols.



Good