A very common exam question in the chapter of Acids and Bases is to ask for the most acidic proton in a molecule or when comparing two molecules.
So, how do we do this?
If you have already covered and learned the key principles of acids and bases, such as the pKa value, its relationship to acids, and the effects of hybridization and resonance on acidity, you know that it’s going to be a long discussion touching on ARIO and stuff like that.
However, as always, let’s try to make it simple, at least for starters, and go from there.
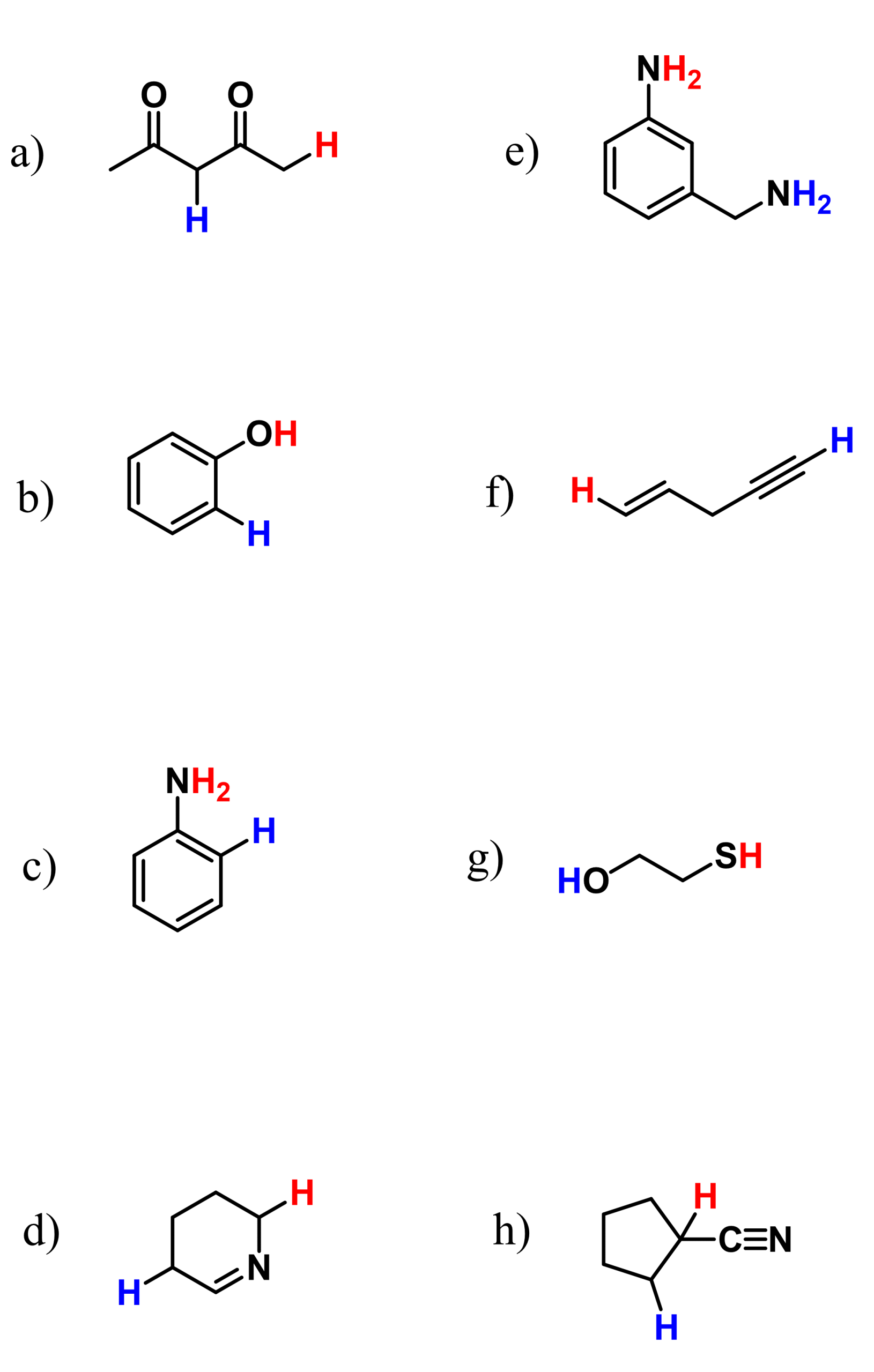
The first thing you should do when choosing the most acidic protons is identify those that are connected to heteroatoms (any atoms other than carbon). The good news is that it’s going to be either N, O, or S, as halogens make only one bond in organic chemistry, and there is not much left that is so common in organic chemistry.
Let’s look at these examples – they should be(come) very easy for you in the course of your organic chemistry class:
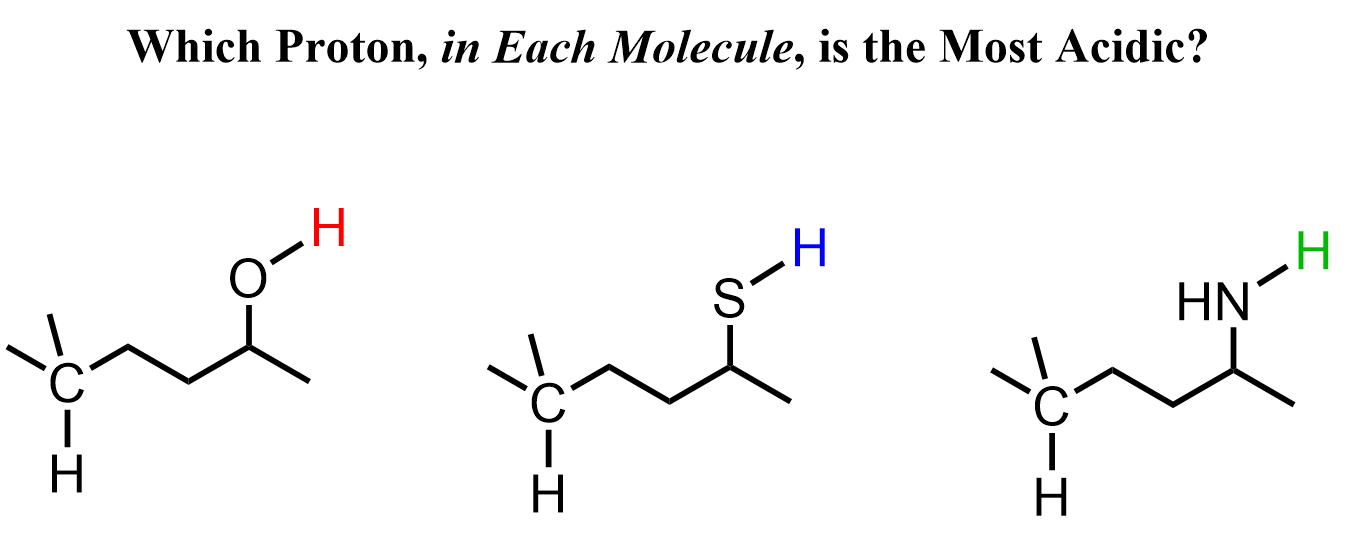
You should be like, “Well, obviously all the colored protons are more acidic.” And yes, a lot of the time, this is going to be correct because hydrogens on electronegative atoms such as O and N are usually more acidic.

Let’s recall why that is the case. Remember, a strong acid is one that “has no problem losing a proton” because it can successfully stabilize the negative charge formed as a result of this loss.
Now, what is a negative charge? A negative charge is an excess of electron density such that it exceeds the standard bonding number of the atom. We know from general chemistry that electronegative atoms like electrons and therefore, they are generally good at stabilizing the negative charge.
This is essentially the “A” of “ARIO” – the atom that handles the negative charge of the conjugate base.
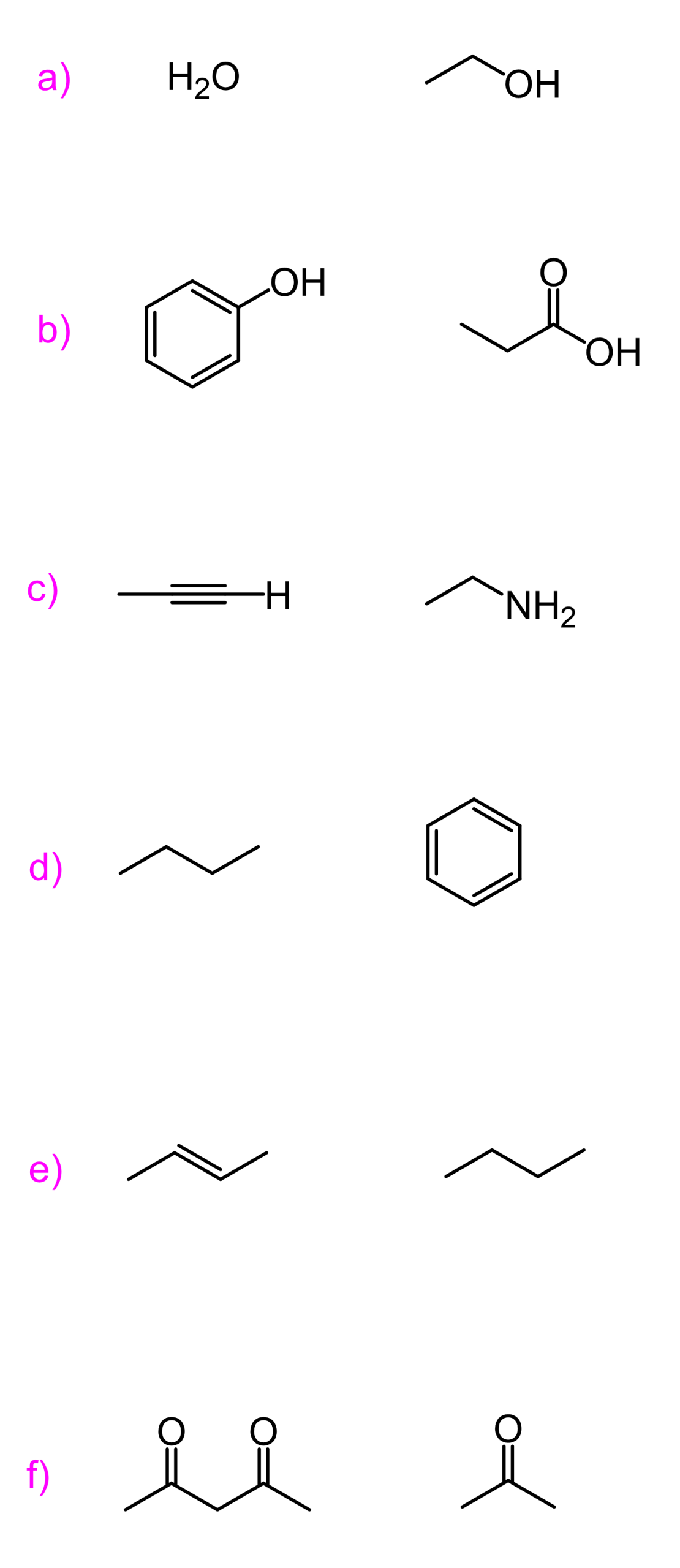
For example, if we compare the acidity of an alcohol with an amine, that would be a less obvious comparison; however, the idea is the same:

Oxygen is more electronegative than nitrogen, and it stabilizes the negative charge better; thus, alcohols are stronger acids than amines.
So, as a general trend, let’s remember that the acidity of H–A increases as the electronegativity of A increases, going from left to right in the periodic table:

Charge and Acidity
Most or a significant portion of the examples we discuss for understanding the acidity of protons are on neutral molecules. However, there are common protonated species in organic chemistry that play important roles in many chemical and biological processes. For example, the protonated forms of amines in amino acids and elsewhere, alcohols, and last but not least, water. We all know the hydronium ion (H3O+) and how scientifically more acidic it is than water:
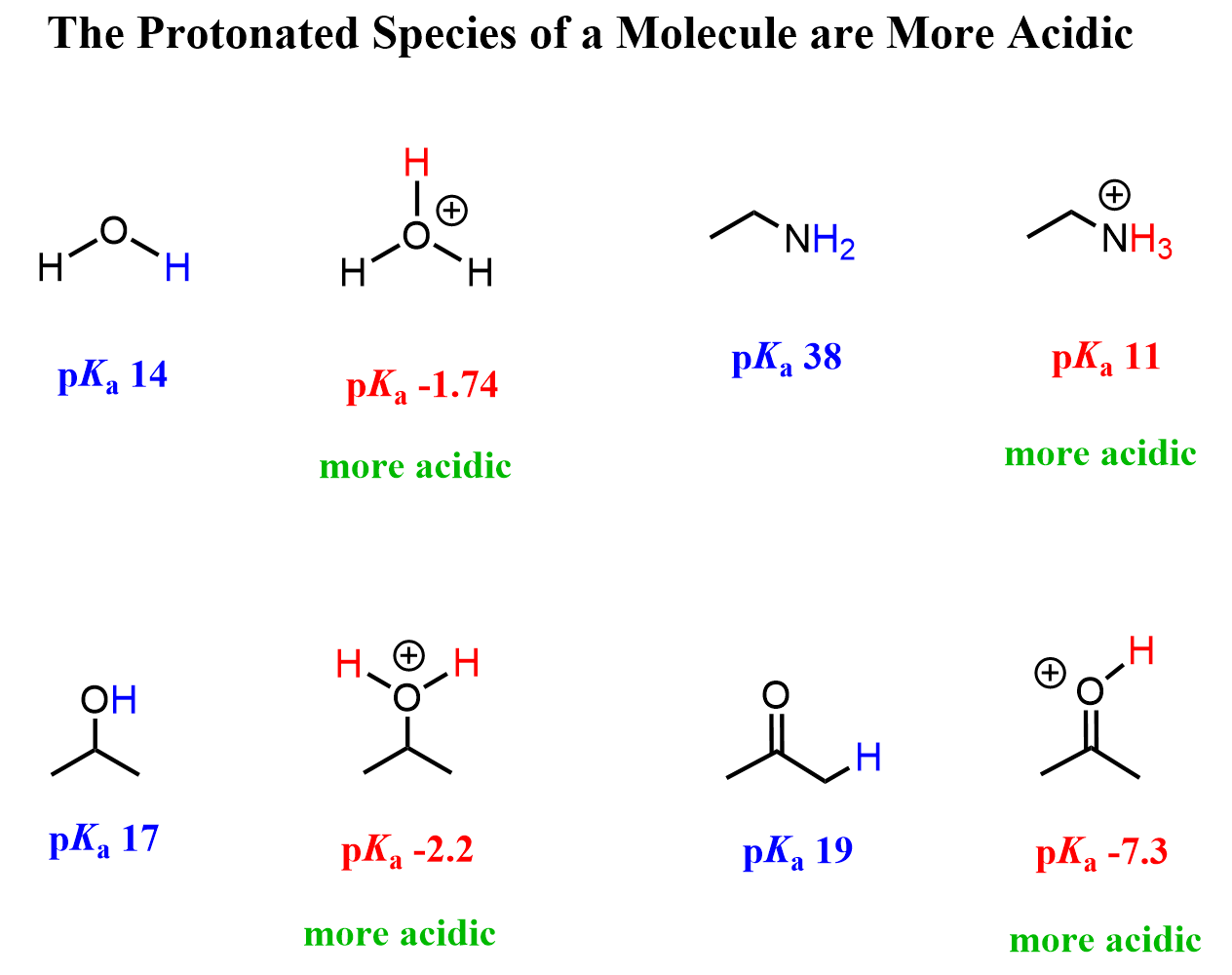
So, the general trend is that protonated species tend to be a lot more acidic than their neutral counterparts. This is intuitive, isn’t it? Why would a molecule want to be protonated? Remember, when discussing major and minor resonance contributors, we said that neutral species are more stable.
Because of this, the term CARIO, where C stands for charge, instead of ARIO, is used.
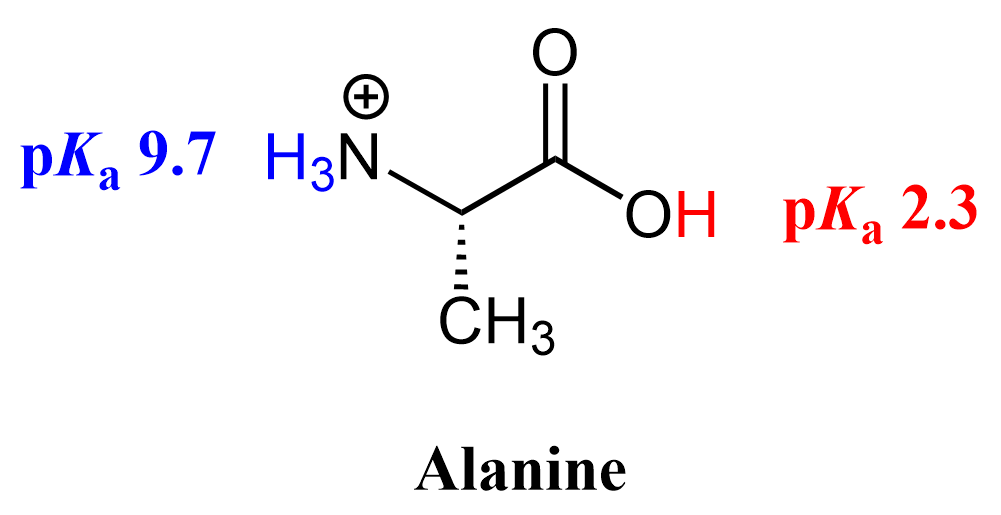
Now, just because “Charge” stands before “Atom” in this abbreviation, it does not mean any protonated atom is always going to be more acidic than a neutral atom. To share a secret with you, you’re going to see that anytime your professor says “always,” there’s going to be an exception (or a few). Hopefully in the lecture, and not on the test 😉.
Here is an example where the proton on a neutral atom is more acidic than the one on a protonated atom.
Atomic Size and Acidity
We mentioned in the previous sentence that the acidity increases for the atoms going from left to right in the periodic table because that’s how electronegativity increases, too.
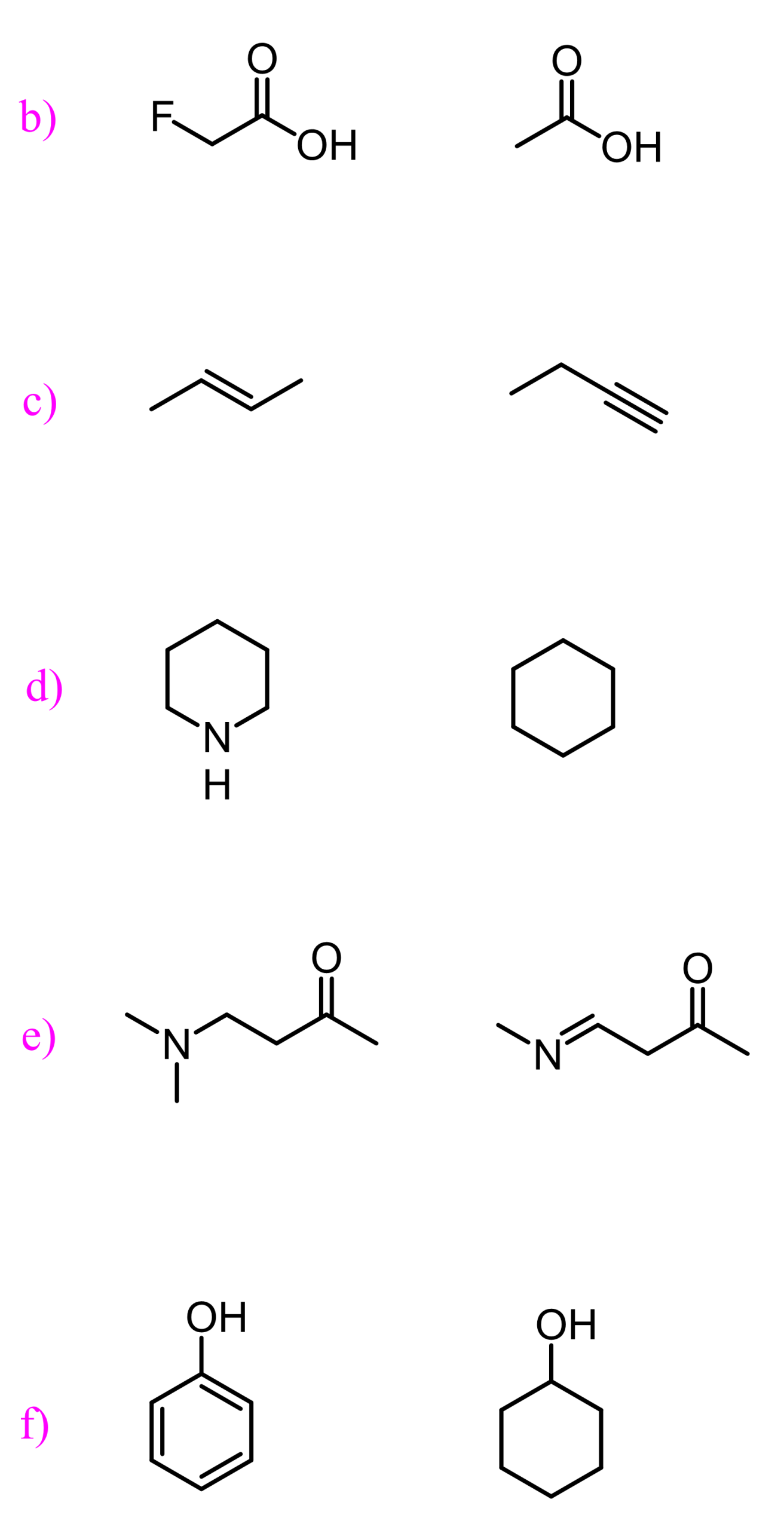
A tricky situation arises when we go down a group in the periodic table. Remember, the atomic radius gets larger, and helps handle the negative charge better despite the atom being less electronegative. As an example, compare the acidity of alcohols and thiols.

The pKa values suggest that thiols are about 1000 times more acidic than alcohols.
The reason for this is the ability of larger atoms to better stabilize the negative charge:

You can think about it this way: the negative charge is spread around a larger surface/volume, thus it is better stabilized.
Therefore, down the periodic table, the atomic size determines the acidity and not the electronegativity.
Inductive Effect and Acid Strength
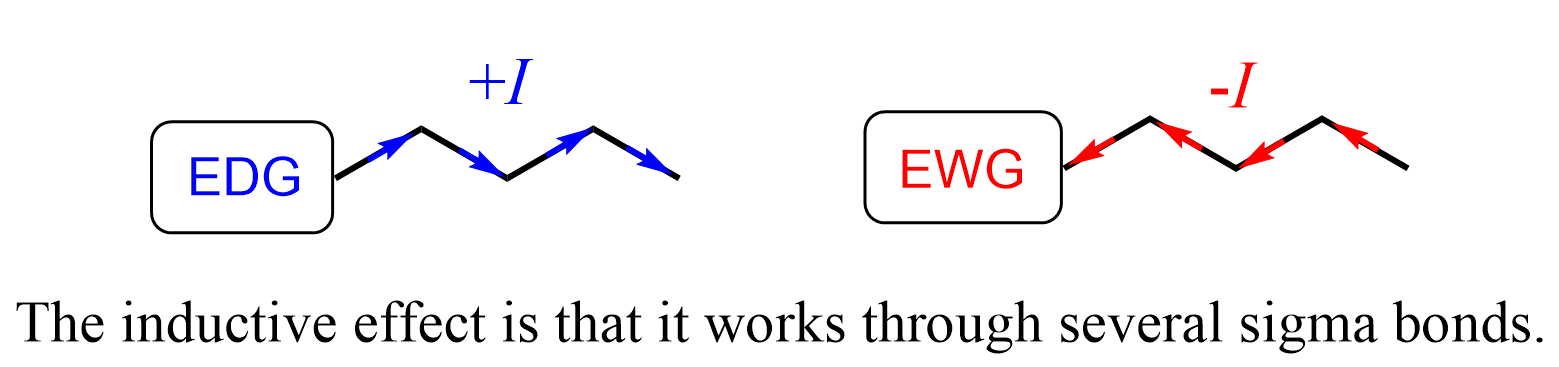
Inductive effect is an electronic effect that occurs through sigma (σ) bonds. If the atom or the group donates electron density, it is said to have a positive inductive effect (+I). Atoms or groups that pull electron density from another atom or a group through sigma bond(s), are electron-withdrawing and denoted as –I groups.
Perhaps the most important feature of the inductive effect is that it works through several sigma bonds.
Let’s see how the inductive effect influences the acidic and basic properties of molecules. For example, 2-fluoroethanol is a stronger acid than ethanol, as seen from their pKa values:
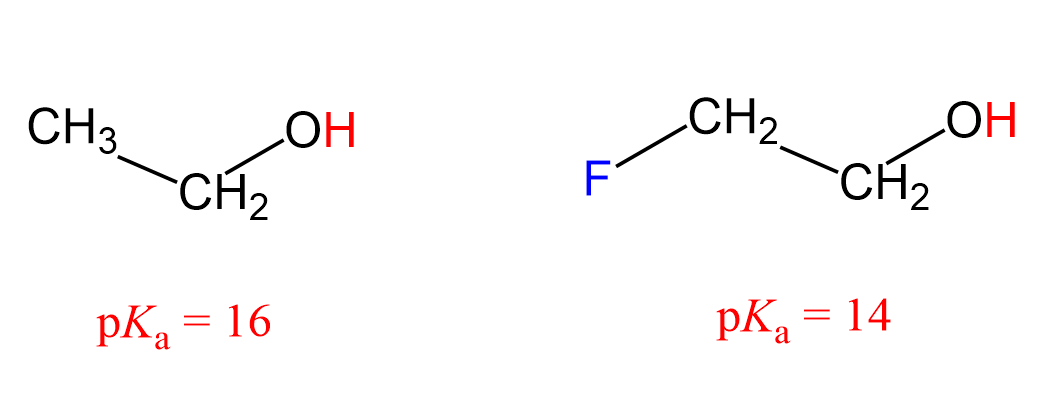
What makes 2-fluoroethanol a stronger acid? The acid strength increases with better stabilization of the negative charge of its conjugate base, so what happens here is that fluorine pulls the electron density from the negatively charged oxygen, thus stabilizing the conjugate base of the alcohol.
The way it works is that the F-C bond reduces the electron density of the carbon, which in turn does the same to the other carbon, and finally, the carbon connected to the oxygen helps it handle the negative charge by reducing its electron density:

This pattern is very common and very well-studied for carboxylic acids. The presence of a halogen increases their acidity through the electron-withdrawing inductive effect. The greater the electronegativity of the halogen, the greater the inductive effect and thus the stronger the acid.
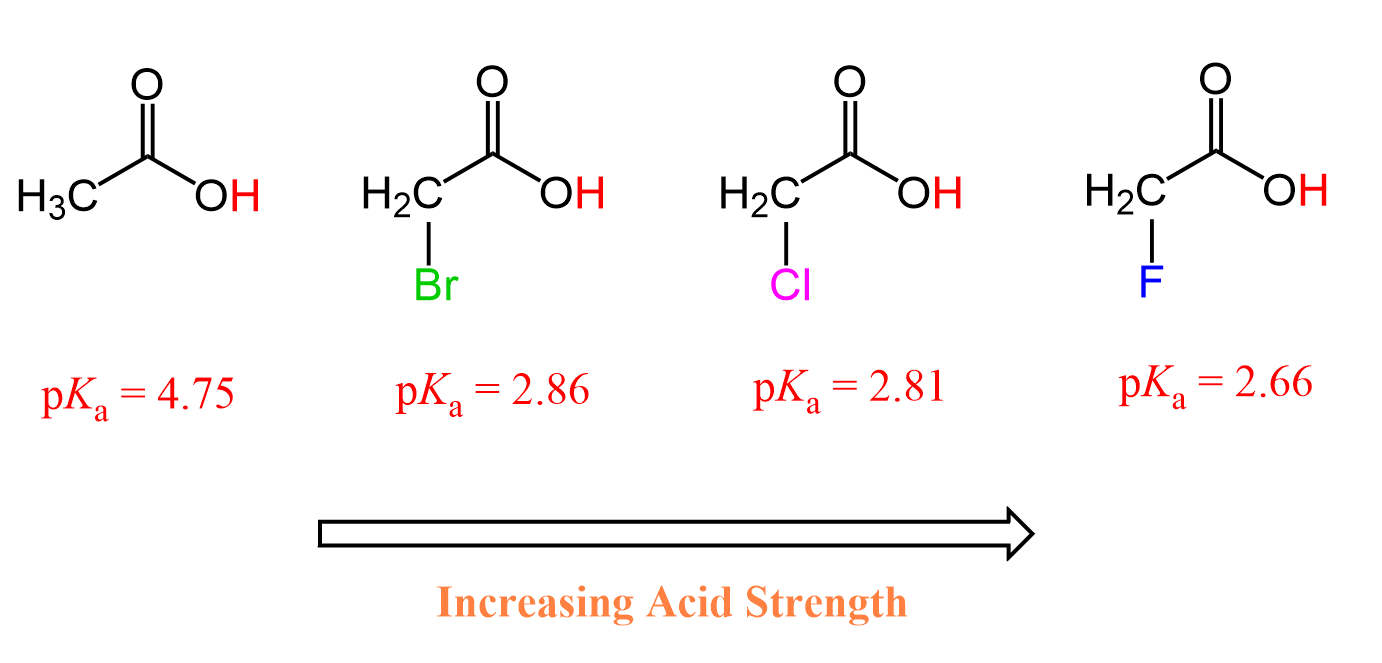
Fluoroacetic acid is stronger than chloroacetic acid, which is stronger than bromoacetic acid, which, in turn, is stronger than acetic acid by itself because the electronegativity of these halogens decreases in this order: F > Cl > Br.
Interestingly, it is not only the nature of the electron-withdrawing atom that affects the charge stabilization but also its distance from the carboxylate group. This is expected because the inductive effect gets weaker with the increasing number of sigma bonds between the two centers:
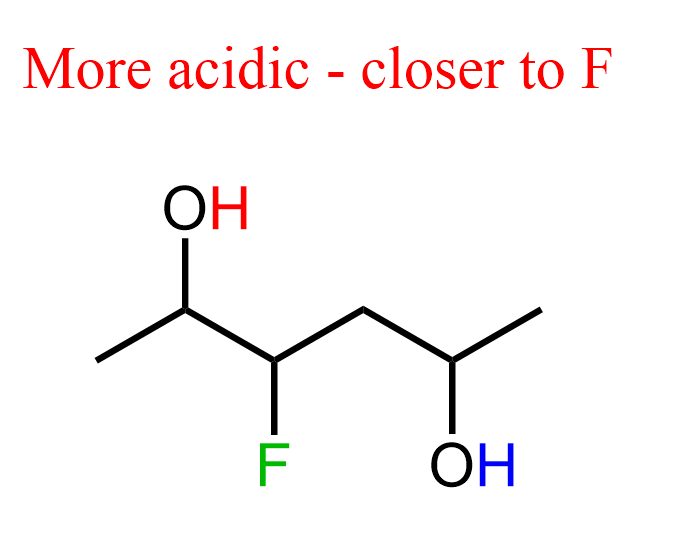
For example, compare the acidity of butanoic acid with 2, 3, and 4-chlorobuatanoic acid:

2-chlorobutanoic acid has the lowest pKa because the chlorine is the closest to the carboxylic group, and thus the inductive effect is the most profound.
So, if you have two protons connected to the same atom, the one closer to an electron-withdrawing group is going to be more acidic:
Acidity and Resonance
Let’s compare the acidity of ethanol and acetic acid. We can see, from the pKa values, that acetic acid is a much stronger acid than ethanol, even though upon dissociation, the negative charge ends up on the same element, which is oxygen:
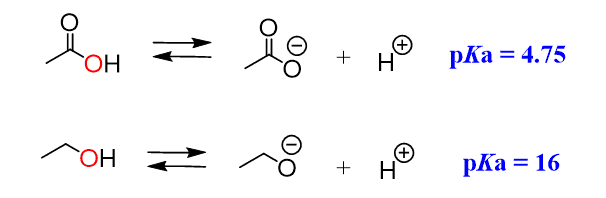
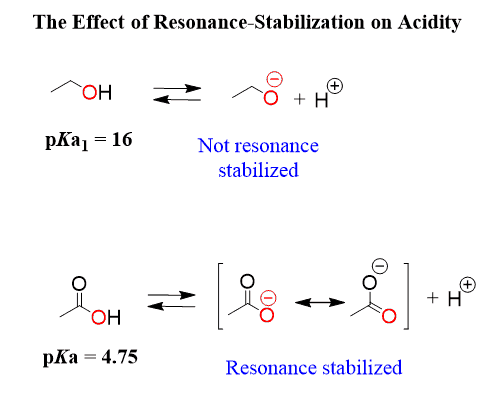
This is because the electrons on the oxygens of carboxylic acid are delocalized (in resonance with the other oxygen) and the negative charge is handled by both atoms, while the oxygen in the alcohol handles the negative charge alone:
Another example of the resonance stabilization of the conjugate base, thus making the acid stronger, is phenol. Compared to cyclohexanol, it is again about a million times more acidic, which is again because of resonance stabilization of the conjugate base. Although this is a bit more complicated, as we also say that the lone pairs on phenol are not delocalized over the benzene ring, as that would disturb the aromaticity, but that is another topic, and a lot of you have probably not talked about aromatic compounds yet.

To keep this fair, another factor contributing to the increased acidity of phenol is the hybridization of the carbon atoms in the aromatic ring. These are sp2 hybridized and sp2 hybridized carbon atoms are more electronegative than sp3 carbons because they have more s character.
As a result, they have an electron-withdrawing inductive effect while the aliphatic carbons are electron donors. So, the sp2 carbon is more helpful in stabilizing the negative charge on the oxygen via an electron-withdrawing inductive effect (Recall the ARIO).
In general, the more s character, the more electronegative the atom is. These are the percentages of the s orbital (s-character) in each hybrid orbital:

To have all these parameters identical and see what would be the effect of resonance electron withdrawing, we can also compare the acidity of phenol with nitrophenols. The nitro group contains a N=O pi bond, and it is a resonance-withdrawing group like, for example, different carbonyl derivatives, such as aldehydes, ketones, carboxylic acids, etc. This pi bond allows for a resonance stabilization of the negative charge that is formed upon the deprotonation of the OH group.
Both 3-nitrophenol (m-nitrophenol) and 4-nitrophenol (p-nitrophenol) are more acidic than phenol.
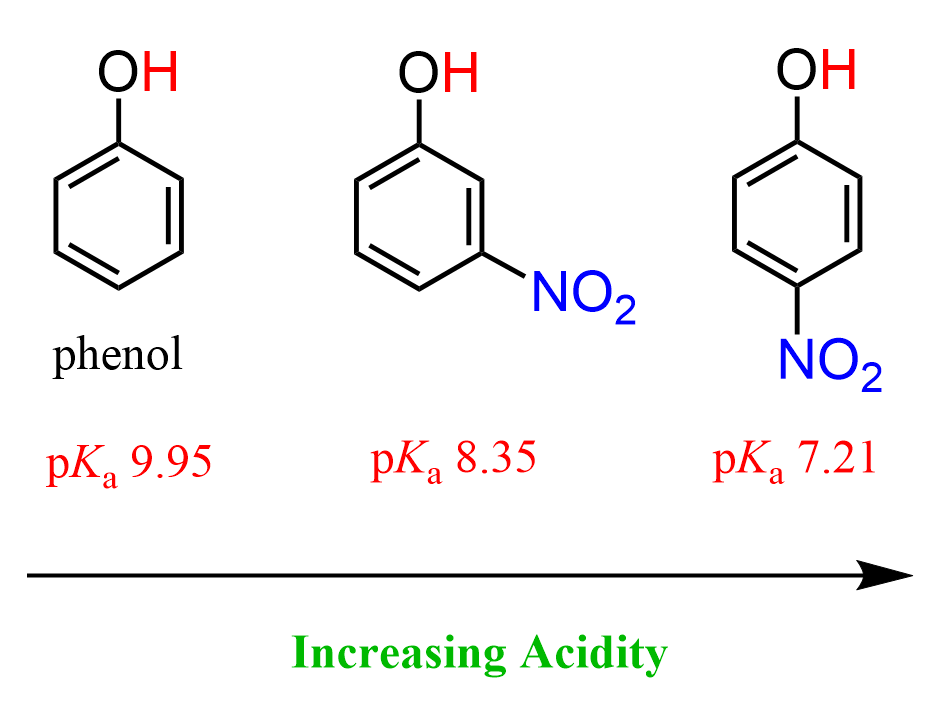
Notice that p-nitrophenol is also more acidic than m-nitrophenol, and that has to do with the fact it the nitro group in the meta position can only stabilize the negative charge via inductive electron-withdrawing effect, whereas in p-nitrophenol, the negative charge is stabilized by both inductive and resonance effects.
You do not need to worry about the terms “meta”, para, “aromatic etc., if you have not reached the chapter of aromatic chemistry, which is an organic 2 topic.
The take-home message for you here is that resonance-withdrawing groups can stabilize the conjugate base by delocalizing its lone pairs, thus increasing the acidity of the compound. Once again, recall that the weaker/more stable the conjugate base, the stronger the acid.
Watch out for Resonance Rules
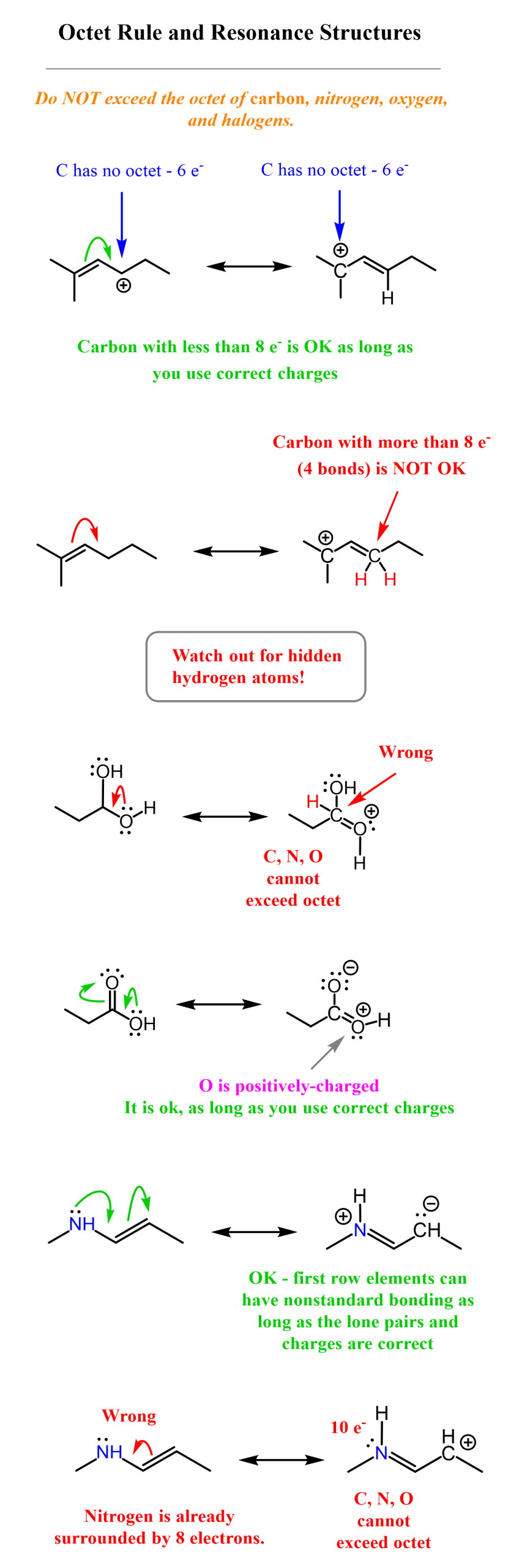
Do not fall for resonance-stabilization anytime you see a double bond near the proton. Not every lone pair (negative charge) can be resonance delocalized. You need to follow the rules for drawing resonance structures. This is mainly about not exceeding the octet on carbon and other second-row elements, and not breaking or making single bonds when drawing resonance forms. Here are some examples of correct and incorrect resonance transformations, and you can check the linked article for more details.
Resonance vs Electronegativity
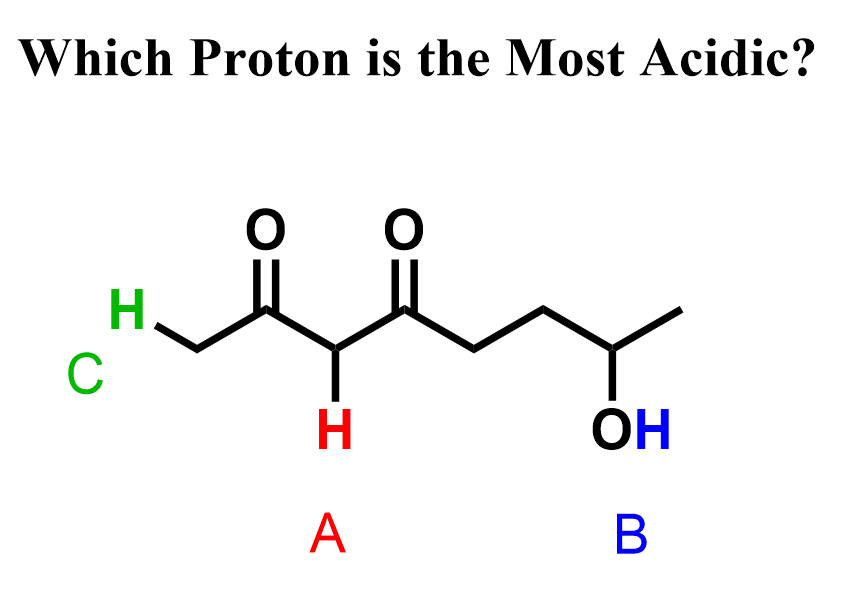
The resonance effect can sometimes make a proton connected to carbon, or, in general, a less electronegative atom, more acidic. For example, the red proton in molecule A is between two resonance-withdrawing carbonyl groups, and it is more acidic than the OH proton. This is despite the fact that it is connected to a carbon, which is less electronegative than oxygen.
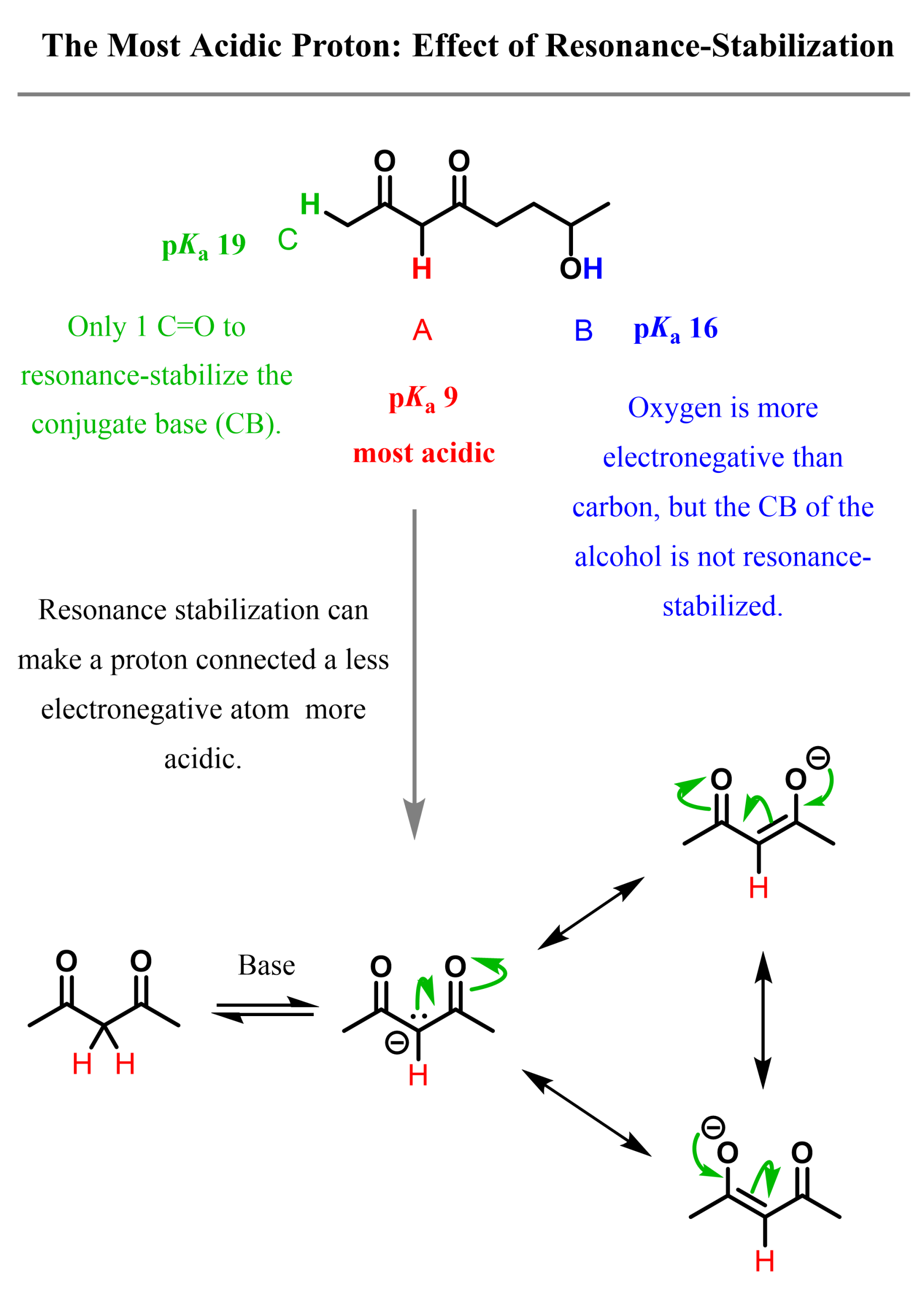
So, we have seen how the resonance stabilization by two carbonyl groups makes proton A more acidic than proton B. Notice that proton C is connected to a carbon, but it only has one adjacent carbonyl group to stabilize the conjugate base. It turns out this is not enough to make it more acidic than the OH proton. In fact, it is about 1000 times weaker than typical alcohols.
At the end, I’ll include a simplified table of pKa values for common functional groups, so you can use it to practice. You’ll “always” be allowed to use a pKa table on the test, but make it a goal to identify acidic protons using the principles we discussed.

You don’t need to memorize every pKa value, but being able to recognize when one proton is significantly more acidic than another is a valuable skill to have.