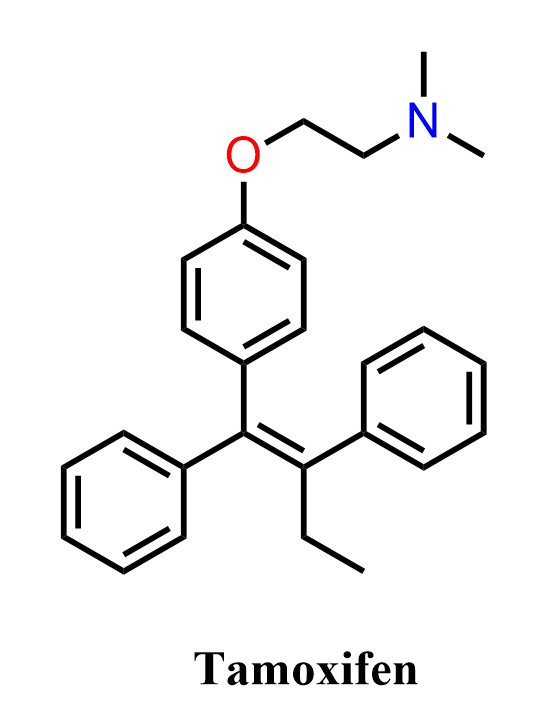
Tamoxifen is a selective estrogen receptor modulator (SERM) widely used in the treatment and prevention of estrogen receptor–positive breast cancer. Structurally, it is a triphenylethylene derivative, consisting of a substituted alkene framework connecting three aromatic rings, along with a basic side chain. Its biological activity arises from binding to estrogen receptors and modulating their activity, making it a classic example of how relatively simple aromatic building blocks can be assembled into a biologically active molecule with significant therapeutic impact. Because of its structure, tamoxifen is often discussed in organic chemistry in the context of alkene synthesis, stereochemistry (E/Z isomerism), and functional group interconversions.
In the following practice problem, we will focus on the organic synthesis of tamoxifen rather than its pharmacological properties. Specifically, we will examine how the triphenylethylene core can be constructed starting from simple aromatic precursors such as benzene, using key transformations including electrophilic aromatic substitution, Friedel-Crafts acylation/alkylation, carbonyl additions, and elimination reactions to form the central alkene. The synthesis also highlights the installation of the dimethylaminoethoxy side chain, illustrating nucleophilic substitution and functional group manipulation. The goal is to practice multistep synthesis design while understanding how these reactions are combined to build the highly substituted alkene framework of tamoxifen.
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
Add the missing intermediates and reagents in the following synthesis of Tamoxifen.
Draw a plausible mechanism for the conversion of intermediate 2 to 3.
This content is for registered users only.
Click here to Register!
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.

