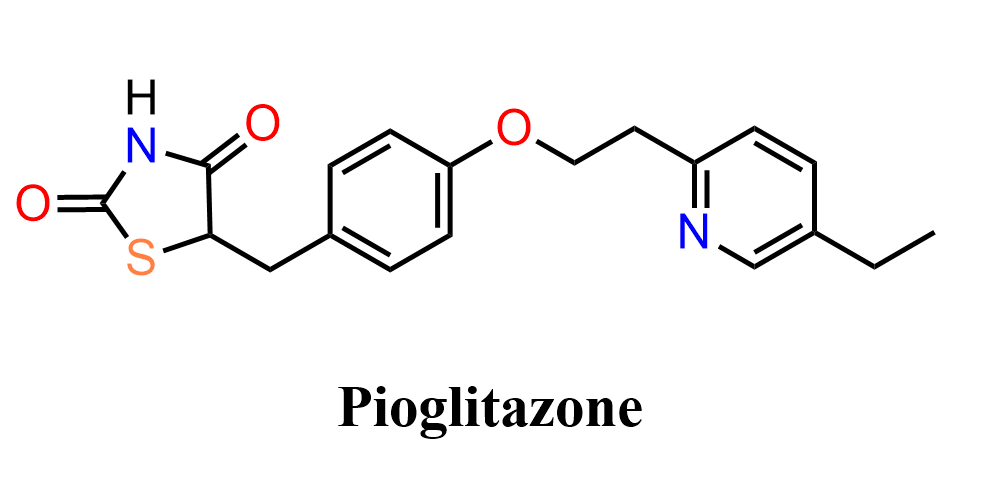
 Pioglitazone is an antidiabetic drug used in the treatment of type 2 diabetes. It belongs to the thiazolidinedione class and works by improving insulin sensitivity, helping to regulate blood glucose levels.
Pioglitazone is an antidiabetic drug used in the treatment of type 2 diabetes. It belongs to the thiazolidinedione class and works by improving insulin sensitivity, helping to regulate blood glucose levels.
Structurally, pioglitazone contains a thiazolidinedione core attached to an aromatic system bearing a pyridine substituent. The molecule features multiple functional groups, including an ether linkage and heterocyclic components, making it a useful target for studying a variety of transformations in organic synthesis.
Because of its structure, pioglitazone is often discussed in the context of nucleophilic aromatic substitution, nitro group reduction, diazotization chemistry, and key carbon–carbon bond-forming reactions involving activated intermediates.
In the following practice problem, we will focus on the organic synthesis of pioglitazone rather than its pharmacological properties. In particular, we will examine how a substituted aromatic system is constructed starting from 4-fluoronitrobenzene via nucleophilic aromatic substitution, followed by reduction to an aniline, conversion into a highly reactive diazonium intermediate, and subsequent transformations that install the required carbon framework, ultimately followed by cyclocondensation with thiourea to build the thiazolidinedione core and complete the synthesis.
As with all examples in this section, this discussion is presented solely for educational purposes and is not intended to represent a practical or real-world method of preparation.
Practice
Add the missing intermediates and reagents in the following synthesis of Pioglitazone. Draw a curved arrow mechanism for the last step of the synthesis.
This content is for registered users only.
Click here to Register!
By joining Chemistry Steps, you will gain instant access to the answers and solutions for all the Practice Problems, including over 40 hours of problem-solving videos, Multiple-Choice Quizzes, Puzzles, Reaction Maps, and the powerful set of Organic Chemistry 1 and 2 Summary Study Guides.

 Pioglitazone is an antidiabetic drug used in the treatment of type 2 diabetes. It belongs to the thiazolidinedione class and works by improving insulin sensitivity, helping to regulate blood glucose levels.
Pioglitazone is an antidiabetic drug used in the treatment of type 2 diabetes. It belongs to the thiazolidinedione class and works by improving insulin sensitivity, helping to regulate blood glucose levels.