α-Cleavage is a common fragmentation in mass spectrometry that occurs in molecules containing a heteroatom, such as oxygen or nitrogen. In this process, the bond between the α-carbon (the carbon directly attached to the heteroatom) and the β-carbon breaks, producing a carbocation stabilized by the lone pair of electrons on the heteroatom and a neutral radical. This fragmentation is especially common in aldehydes, ketones, esters, alcohols, and amines, and often gives strong, diagnostic peaks in the mass spectrum.
For example, in the mass spectrum of 2-pentanol, the molecular ion peak is missing, while the base peak appears at m/z 45. What happens is that the radical cation of the molecule undergoes α-cleavage, breaking the bond between the carbon bearing the OH group and the α-carbon (the adjacent carbon):

Notice that the α-cleavage occurs on both sides of the carbonyl, forming two resonance-stabilized carbocations.
As mentioned earlier, α-cleavage is characteristic of oxygen- and nitrogen-containing compounds, including carbonyl functional groups. For example, the mass spectrum of isovaleric acid shows an α-cleavage peak at m/z 45:
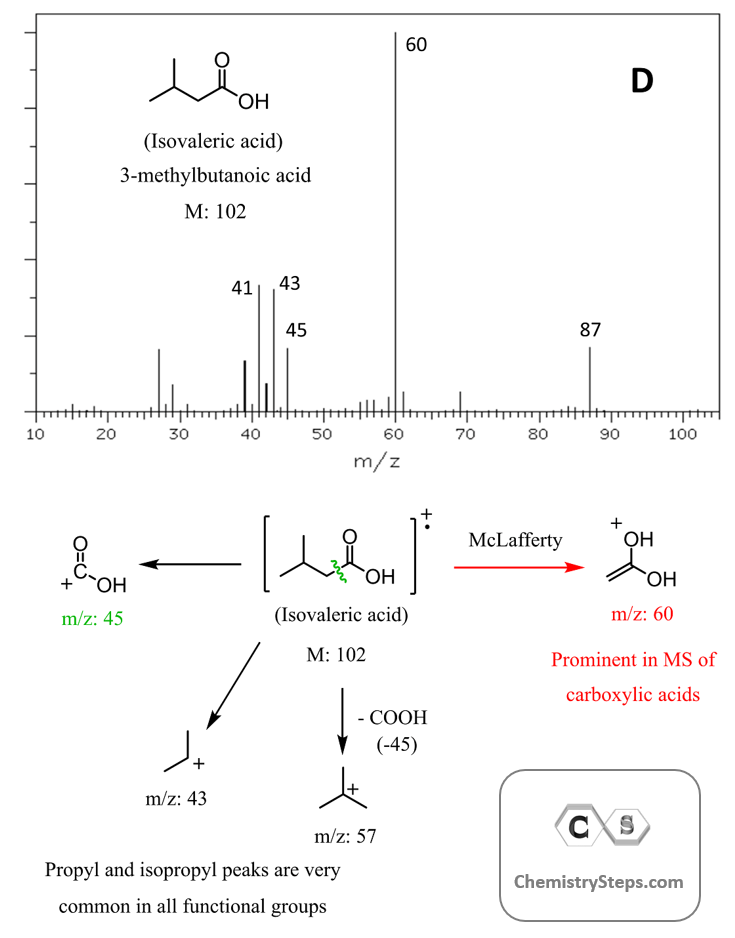
Notice that the base peak appears at m/z 60, and it originates from McLafferty rearrangement, which generates a resonance-stabilized enolic carbocation. McLafferty rearrangement is very common in the mass spectra of carbonyl-containing compounds, and we have a separate post on it, so feel free to check that out as well.
Check also our detailed Guide on Solving Mass Spectrometry Problems that includes the key McLafferty fragmentations of aldehydes, ketones, esters, acids, and nitriles. Once you master the key fragmentation, work on the comprehensive Mass Spectrometry Problems.