We have seen that in the Diels-Alder reaction, the electrons flow in a cyclic, concerted manner. Therefore, the Diels-Alder reaction is classified as a pericyclic reaction, a class of reactions that proceed through a cyclic transition state with no intermediates.

Another important type of pericyclic reactions is sigmatropic rearrangements, where a σ bond shifts across the face of one or more π bonds.
For example, heating allyl phenyl ether at 200-250 °C leads to the formation of o-allylphenol via a sigmatropic rearrangement.
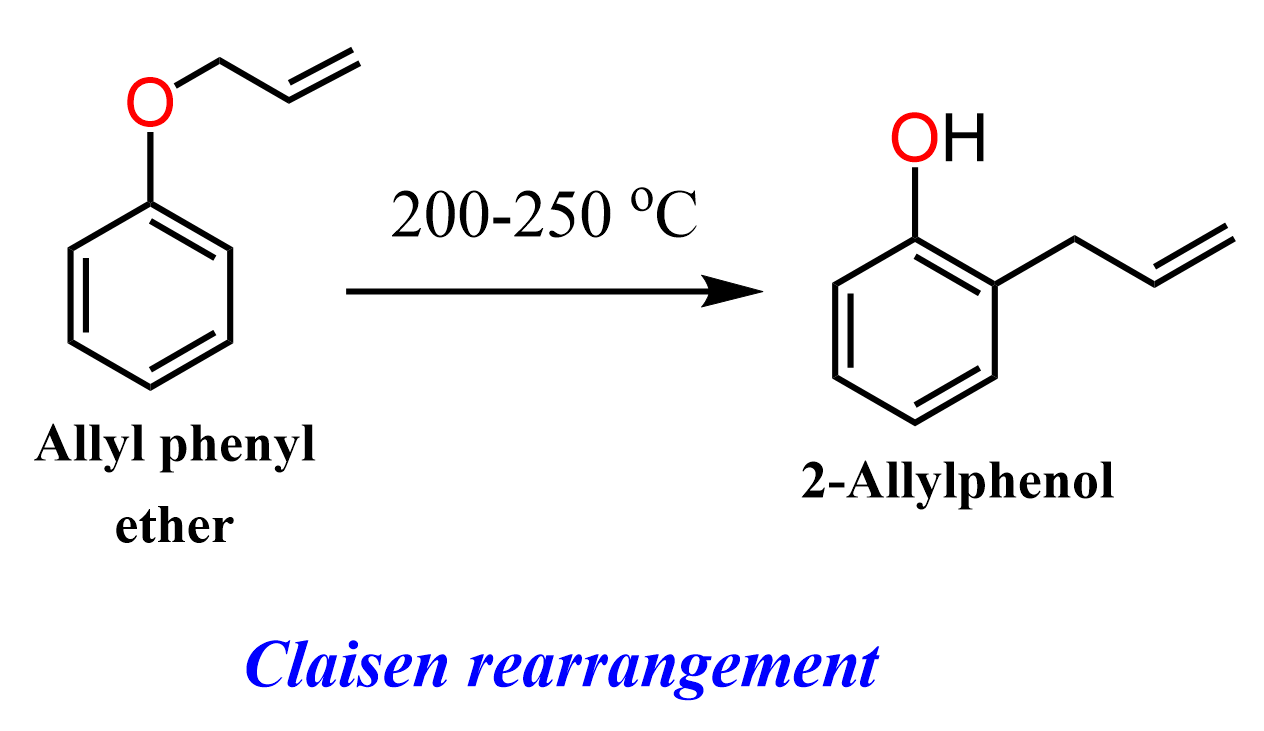
This transformation is known as the Claisen rearrangement, one of the most common and important pericyclic reactions encountered in organic chemistry.
The Mechanism of Claisen Rearrangement
Let’s see how this transformation happens. The reaction proceeds through a concerted, cyclic transition state involving six electrons. Upon heating, the π electrons of the allyl group and the aromatic ring, together with the σ bond between oxygen and the allyl group, reorganize in a single step: the O-allyl σ bond breaks while a new C–C bond forms at the ortho position, and the π bonds shift accordingly to give an enol intermediate. The enol then rapidly tautomerizes to the more stable O-allylphenol.
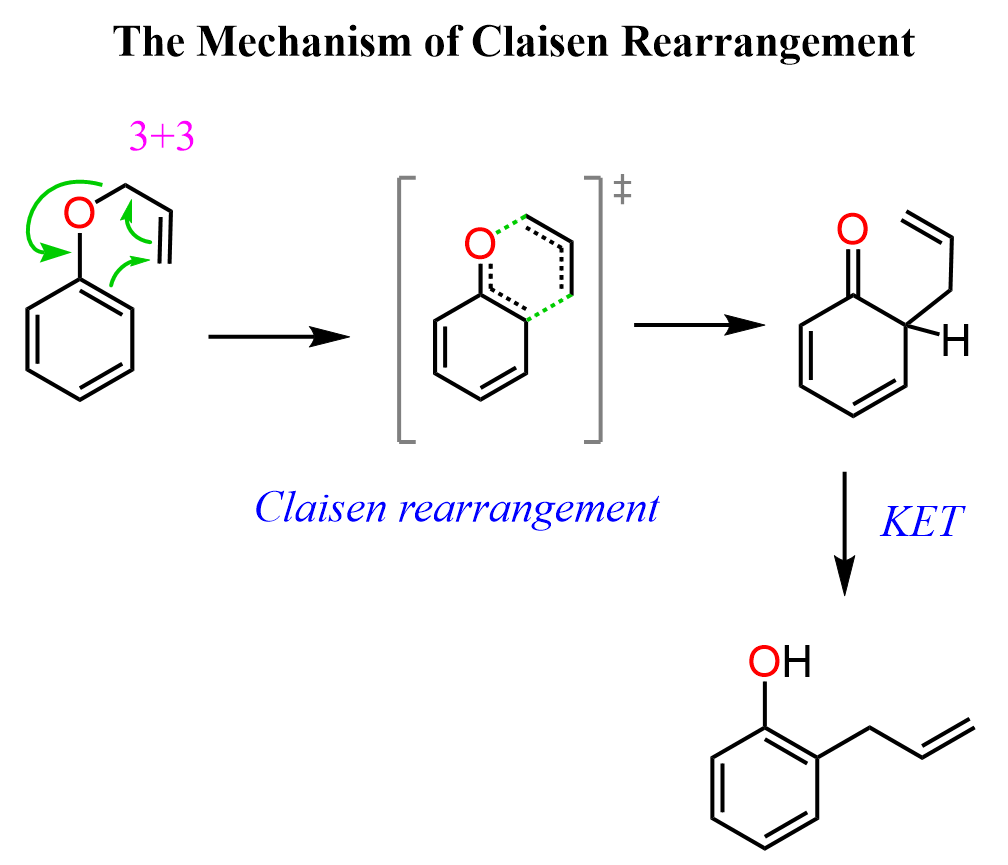
Notice that the “[3,3]” label indicates that a σ bond is migrating across two π systems, each containing three atoms, which together form the six-membered cyclic transition state.
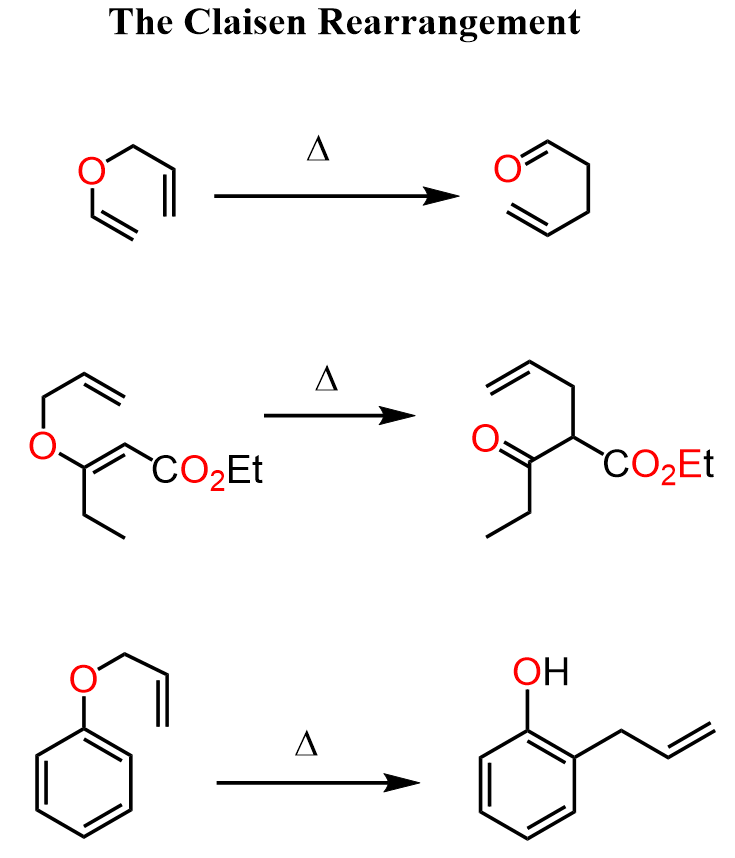
The Claisen rearrangement is not restricted to phenyl allyl ethers. In fact, it may not be the best example for explaining the driving force of the reaction, which is the formation of a more stable carbonyl compound.
It just happens that phenol is an exception where the enol form is more stable than the keto form because of aromatic stabilization. However, we know that in most systems, the keto form predominates in the equilibrium.
For example, here are some Claisen rearrangements where we can clearly see the final product containing the carbonyl group.
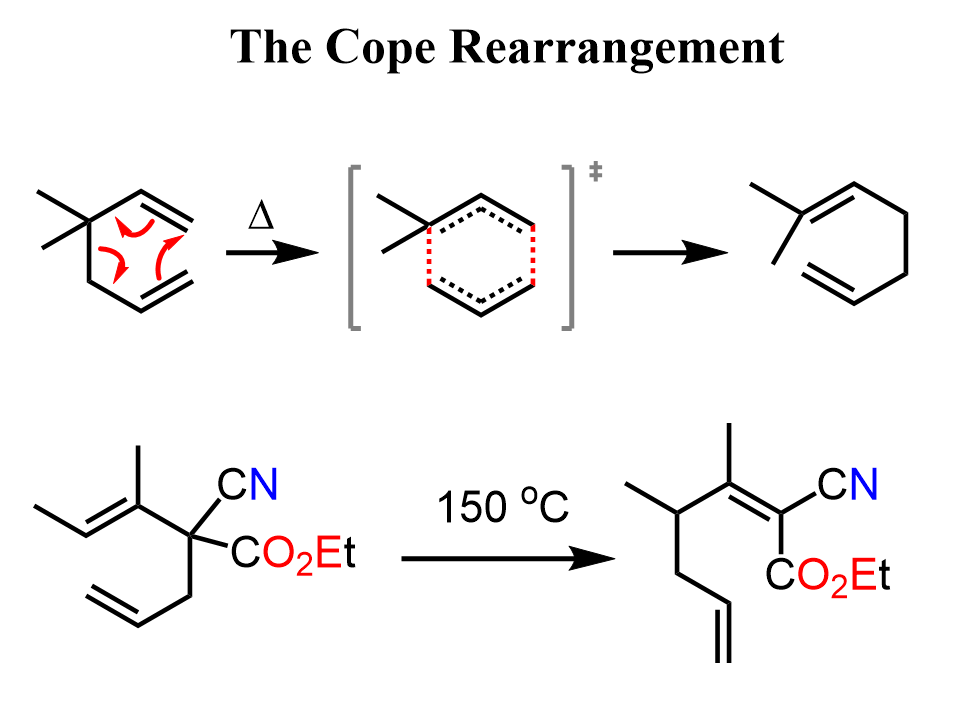
The Cope Rearrangement
A similar rearrangement happens with 1,5-dienes, known as the Cope rearrangement. Like the Claisen rearrangement, it is a concerted pericyclic process that proceeds through a six-membered cyclic transition state, where bonds are broken and formed simultaneously in a single step with no intermediates. In both cases, a σ bond migrates across a π system in a stereospecific fashion, following orbital symmetry rules. The key similarity is that both are [3,3]-sigmatropic rearrangements involving 6 π-electrons in a cyclic transition state.
Cascade of Sigmatropic Rearrangements
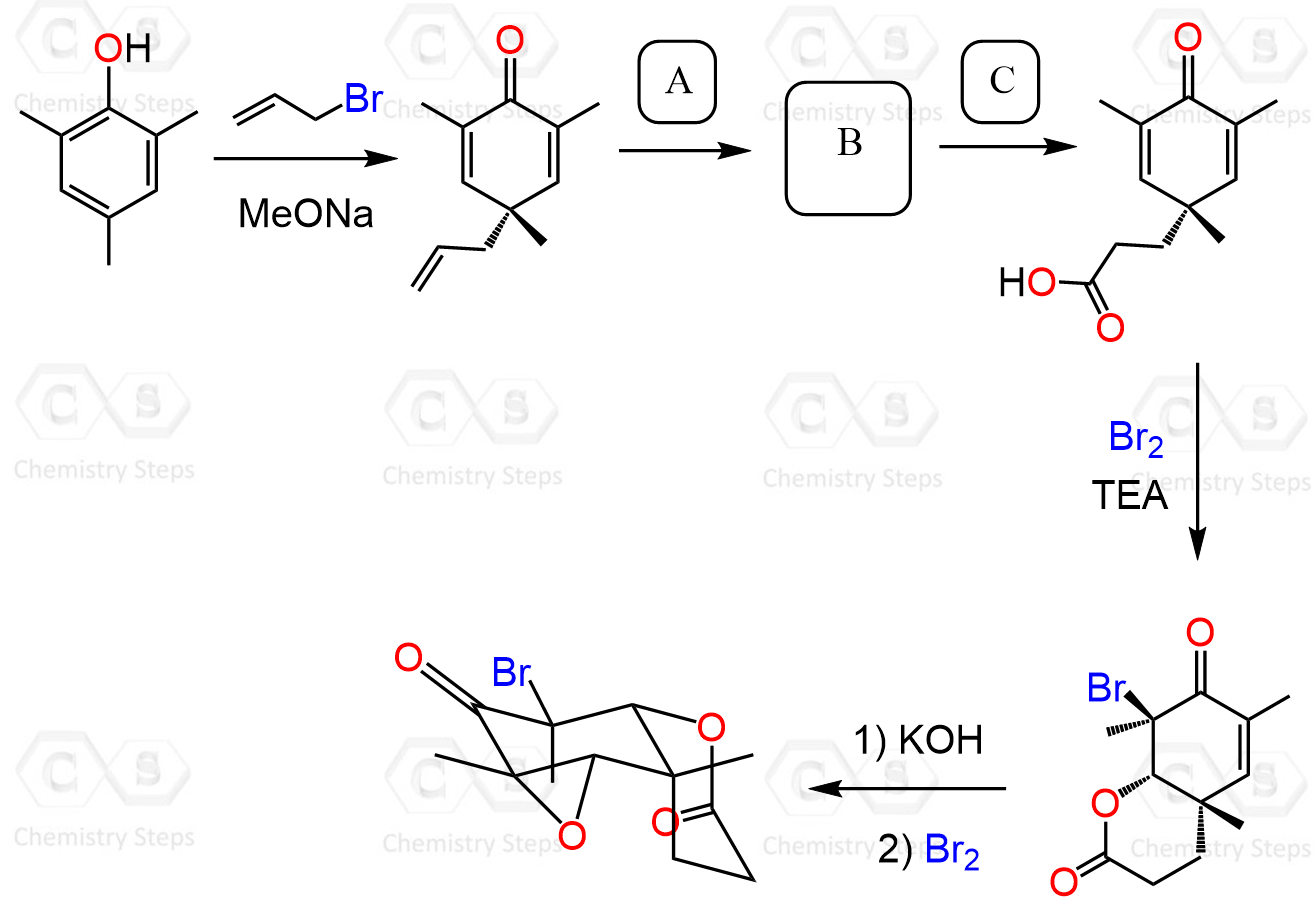
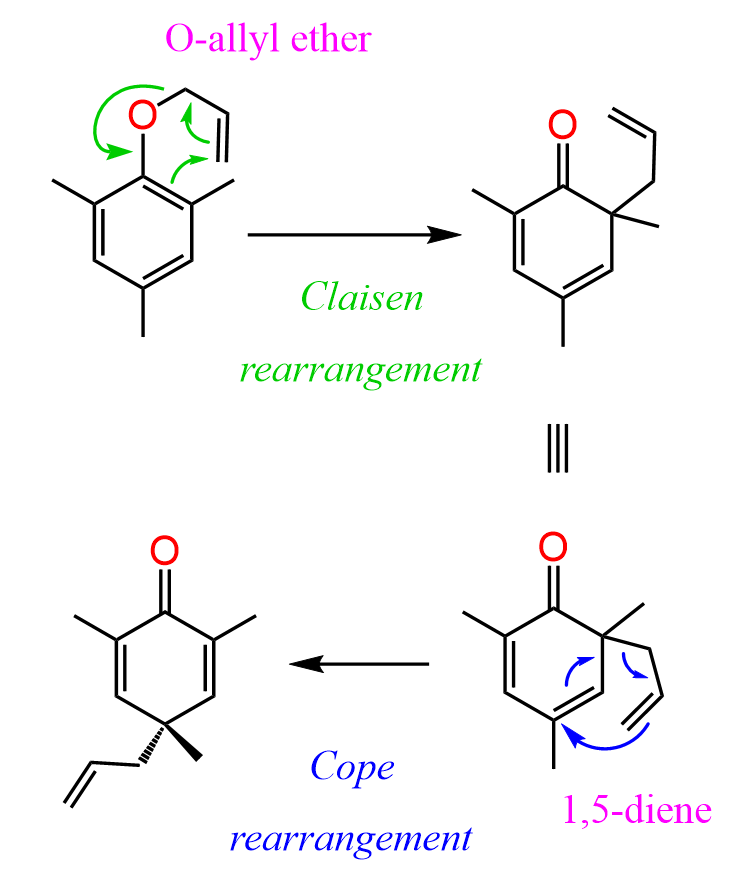
Sometimes a sequence of sigmatropic rearrangements can occur. For example, instead of phenyl allyl ether, if we take 2,4,6-trimethyl phenyl allyl ether, the allyl group ends up in the para position instead of the ortho position as in the earlier example.
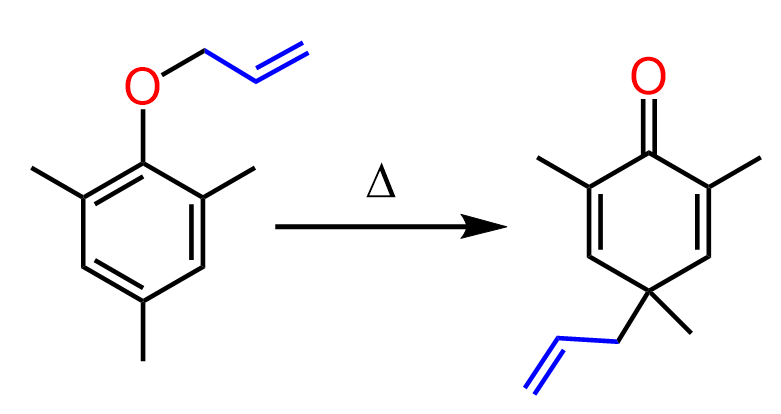
What happens here is that there is no available proton at the ortho position of the phenol, so the usual keto-enol tautomerization pathway is not possible. Instead, the intermediate undergoes a Cope rearrangement, involving the π electrons of the aromatic system, which allows the allyl group to migrate to the para position through a further pericyclic shift:
Reference: Comprehensive Chirality, Volume 2, 2012, Pages 625-647
Org. Chem. 2003, 68, 5493-5499