Direct Conversion of Acid Chlorides to Ketones
Acid chlorides also known as acyl chlorides (ROCl) can be converted to ketones in direct and indirect transformations. The direct conversation is achieved by their reaction with organocuprates (Gilman reagent – R2CuLi).
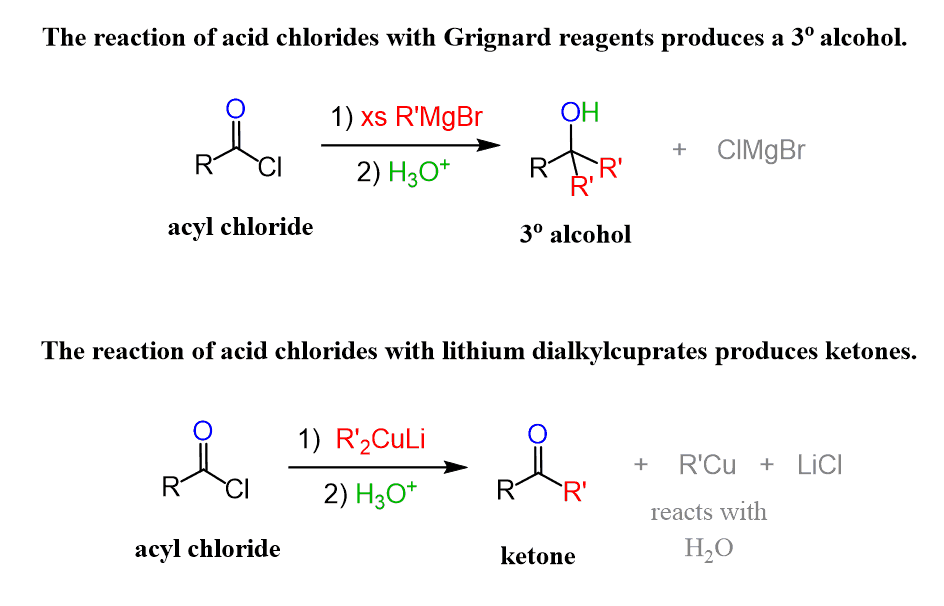
Notice that Grignard and organolithium reagents make additions to the carbonyl group of acid chlorides thus converting them to tertiary alcohols. We have also seen this pattern in the reaction of organometallics with esters and acid anhydrides. The reason for this is the formation of a ketone after the first addition which is further attacked by the nucleophile.
What makes organocuprates special is they are unreactive towards aldehydes, ketones, and derivatives of carboxylic acids except for acid chlorides. Therefore, the ketone that is formed after the nucleophilic addition can be isolated as the final product of the reaction:

The mechanism of organocuprates is not as simple as it is shown here, and it is believed to involve radicals. It is beyond the scope of this post to go into more detail, so ask your instructor if this would be an acceptable mechanism for the nucleophilic addition of organocuprates.
Acid Chlorides to Ketones via Carboxylic Acids
If using an organocuprate is not an option, then we can hydrolyze the acid chloride to a carboxylic acid and react the latter with an organolithium:

Notice the contrast of using a soft organometallic R2CuLi with a very strong organolithium when switching from an acid chloride to carboxylic acid. Carboxylic acids do not undergo other nucleophilic additions because they are first deprotonated to carboxylate ions which are not as electron-deficient to undergo a nucleophilic attack.
Organolithiums, however, are extremely powerful bases and nucleophiles capable of adding to the carboxylate ion. This addition forms a tetrahedral dianion intermediate which lacks a relatively good leaving group and stays in the solution until water is added to hydrolyze it the corresponding ketone:

Acid Chlorides to Ketones via Amides
Another approach for indirect conversion of acid chlorides to ketones is the use of amides. Preparing amides from acid chlorides is easily achieved by reacting them with ammonia or amines. If it is a primary amide, we can convert it to a nitrile by dehydrating it with SOCl2 or P2O5, and reacting the latter with a Grignard or organolithium reagent. So, this approach allows for using a Grignard reagent if organocuprates and organolithiums are not available:

Notice from the synthesis scheme that there are other approaches for converting amides to ketones such as the use of Weinreb and N-Boc amides, but that would deviate from today’s topic too much, so feel free to check the article “Converting Amides to Ketones”.
Organic Chemistry Reaction Maps
Never struggle again to figure out how to convert an alkyl halide to an alcohol, an alkene to an alkyne, a nitrile to a ketone, a ketone to an aldehyde, and more! The comprehensive powerfull Reaction Maps of organic functional group transformations are here!
Check Also
- Preparation of Carboxylic Acids
- Naming Carboxylic Acids
- Naming Nitriles
- Naming Esters
- Naming Carboxylic Acid Derivatives – Practice Problems
- Fischer Esterification
- Ester Hydrolysis by Acid and Base-Catalyzed Hydrolysis
- What is Transesterification?
- Esters Reaction with Amines – The Aminolysis Mechanism
- Ester Reactions Summary and Practice Problems
- Preparation of Acyl (Acid) Chlorides (ROCl)
- Reactions of Acid Chlorides (ROCl) with Nucleophiles
- Reaction of Acyl Chlorides with Grignard and Gilman (Organocuprate) Reagents
- Reduction of Acyl Chlorides by LiAlH4, NaBH4, and LiAl(OtBu)3H
- Preparation and Reaction Mechanism of Carboxylic Anhydrides
- Amides – Structure and Reactivity
- Naming Amides
- Amides Hydrolysis: Acid and Base-Catalyzed Mechanism
- Amide Dehydration Mechanism by SOCl2, POCl3, and P2O5
- Amide Reduction Mechanism by LiAlH4
- Amides Preparation and Reactions Summary
- Amides from Carboxylic Acids-DCC and EDC Coupling
- The Mechanism of Nitrile Hydrolysis To Carboxylic Acid
- Nitrile Reduction Mechanism with LiAlH4 and DIBAL to Amine or Aldehyde
- The Mechanism of Grignard and Organolithium Reactions with Nitriles
- Carboxylic Acids to Ketones
- Esters to Ketones
- Carboxylic Acids and Their Derivatives Practice Problems
- Carboxylic Acids and Their Derivatives Quiz