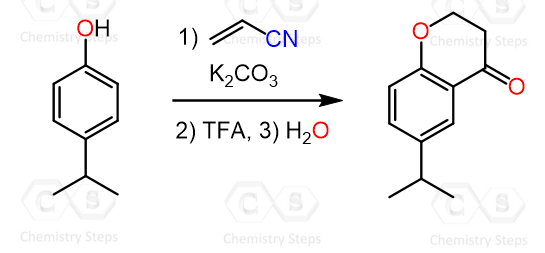
We have seen how aryl ketones are prepared by the Friedel-Crafts acylation when benzene or other aromatic compound derivatives are reacted with acid chlorides in the presence of Lewis acids such as AlCl3.
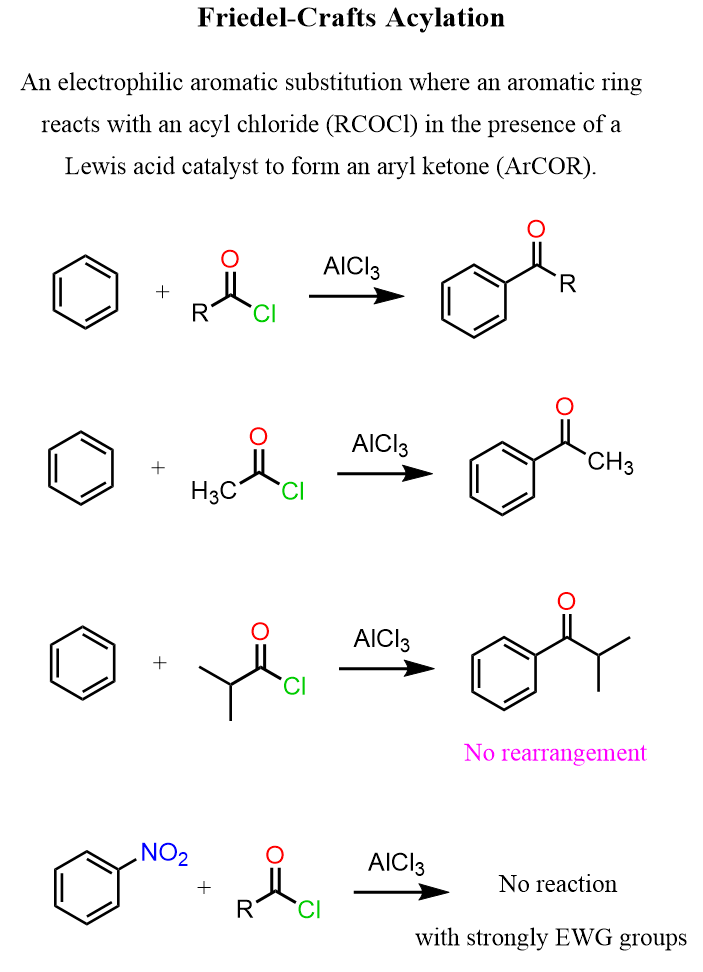
Now, when highly activated benzene rings such as those of phenol and aniline are used, there are a few issues, such as O- and N-alkylation as well as polyacylation of the aromatic ring.
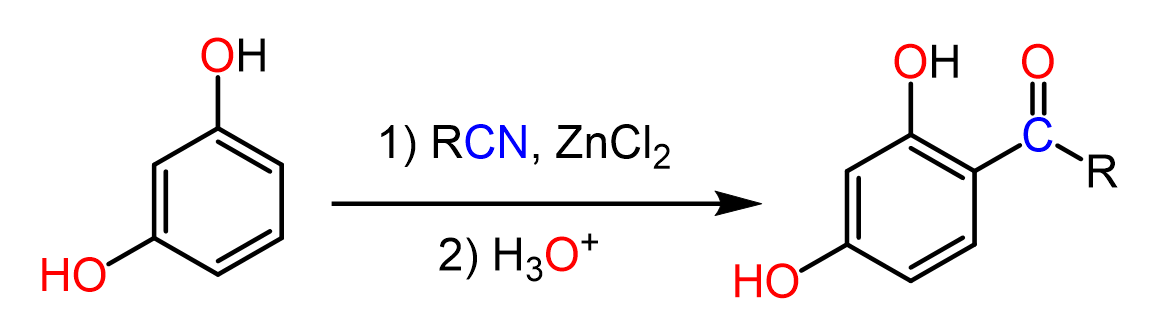
To overcome this, in 1915, Karl Hoesch developed a method for monoacylation of highly activated aromatic rings such as polyphenols and their ester derivatives. In this approach, nitriles replace the acyl chlorides, and instead of harsh Lewis acids like AlCl3, milder acids such as ZnCl2, often in the presence of HCl, are used.
The idea is to use a less electrophilic reactive species for incorporating the ketone group, and it turned out nitriles are great candidates for this because they contain an electrophilic carbon atom, but it is not as reactive because instead of an oxygen, it is connected to a less electronegative nitrogen:

So, if we activate this carbon by either protonating or coordinating a Lewis acid, we can direct the π electrons of an aromatic system to make a new C-C bond:
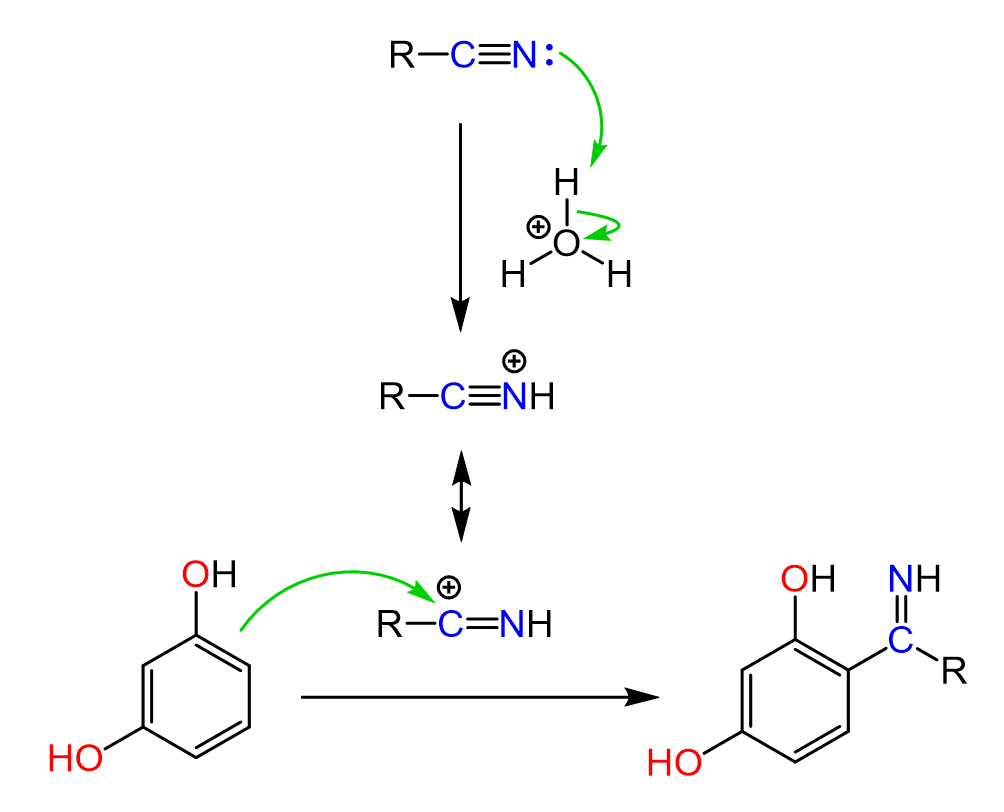
Notice that we are showing the nucleophilic attack of the aromatic ring on the positively charged carbon atom, but it can also occur on the other resonance structure by pushing the pi bonds to the nitrogen. Resonance structures are not real after all, and we do this to emphasize the increased electrophilicity of the carbon atom when the nitrogen is protonated.
So far so good, but the next question you may be wondering is how that nitrogen is replaced with an oxygen – how do we transform the C=N to a C=O so that we get our target aryl ketone?
Recall from the functional groups that the intermediate that we showed above is an imine, and imines can be hydrolyzed to carbonyls under acidic conditions.

We also cover the entire hydrolysis of nitriles to ketones, which involves the hydrolysis intermediate imines here.
This is a topic that belongs in the reactions of carboxylic acids and their derivatives, and if you have not covered that in class yet, you can skip it for now.
So, in the Houben-Hoesch reaction, we use a nitrile as a source of the carbonyl, as opposed to what we are used to seeing in the Friedel-Crafts acylation. The C=N group is then hydrolyzed to the corresponding ketone, which allows an effective acylation of activated aromatic systems such as polyphenols and their ether derivatives.
The Mechanism of the Houben-Hoesch Reaction
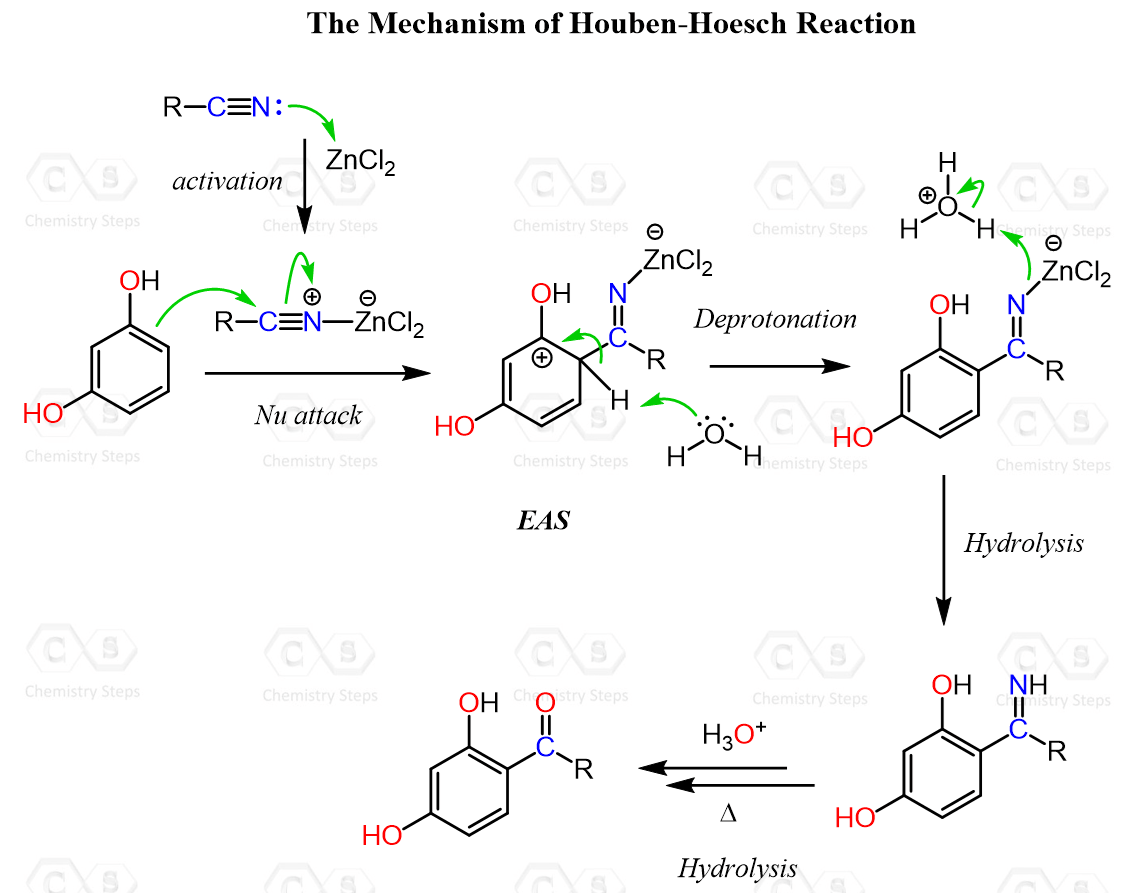
We have just seen the principle of the reaction and key parts of the mechanism using protonation of the nitrile, so let’s also write a complete mechanism using ZnCl2, since both types of catalysts can be used.
The reaction starts with the activation of the nitrile by coordination of the nitrogen to the Lewis acid. This makes the nitrogen positively charged and increases the electrophilicity of the nitrile carbon, allowing for nucleophilic attack of the aromatic ring. The resulting sigma complex is then deprotonated to restore aromaticity, giving a ketimine intermediate. Finally, the ketimine is hydrolyzed to the corresponding ketone.
This is by far not the only reaction of nitriles where they undergo nucleophilic addition. Reactions such as reduction, hydrolysis, and reactions with Grignard and organolithium reagents are also common, so check out this article on the reactions of nitriles for more details.
Let’s also summarize and list some important features of the Houben-Hoesch reaction:
- The Houben-Hoesch reaction is used for monoacylation of activated aromatic systems
- Only highly activated aromatic compounds undergo the transformation, typically those bearing hydroxy or alkoxy groups, and at least one is required.
- The reaction works best for disubstituted systems such as polyphenols, where controlled monoacylation is needed.
- Heterocyclic compounds such as pyrroles, indoles, and furans can also participate.
- The reaction proceeds through a less reactive electrophile, which helps avoid polyacylation.