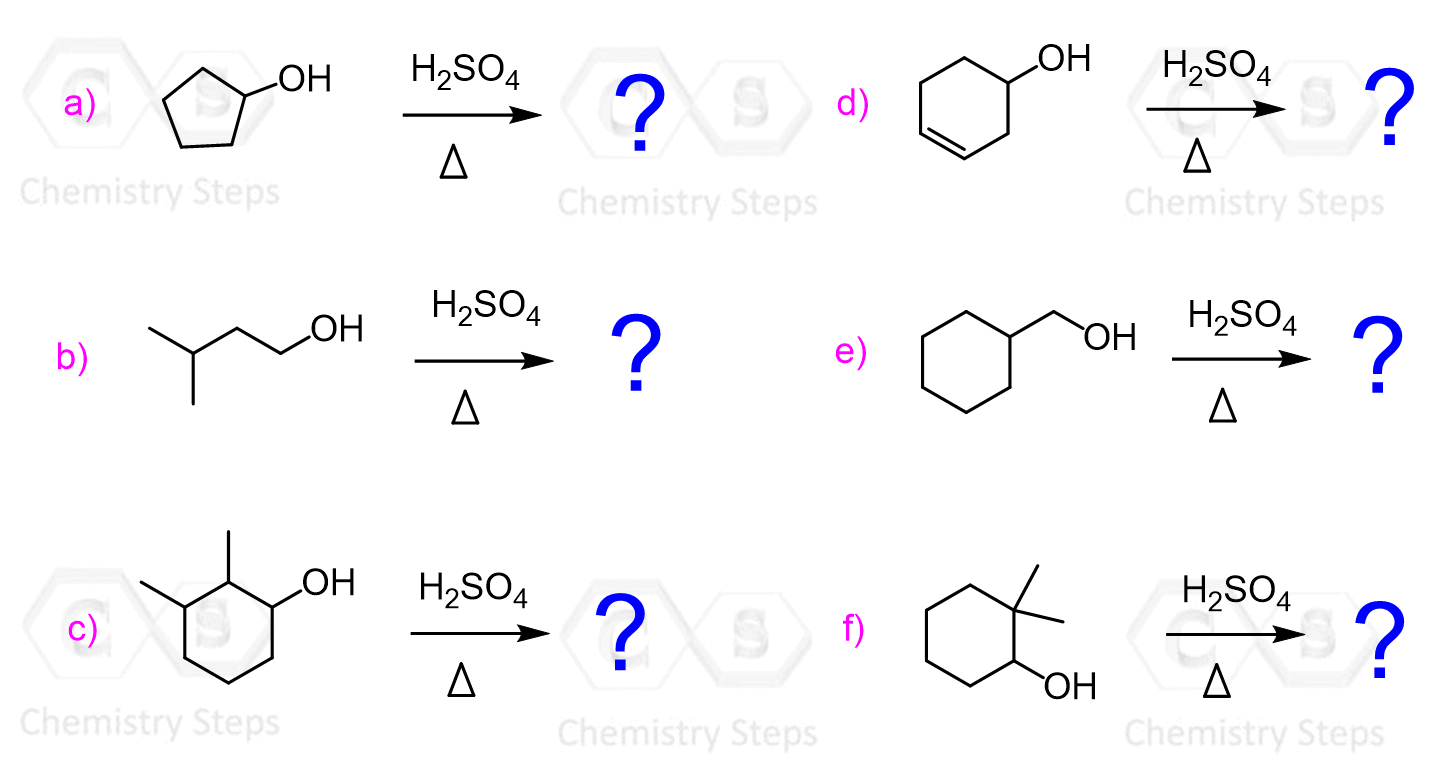
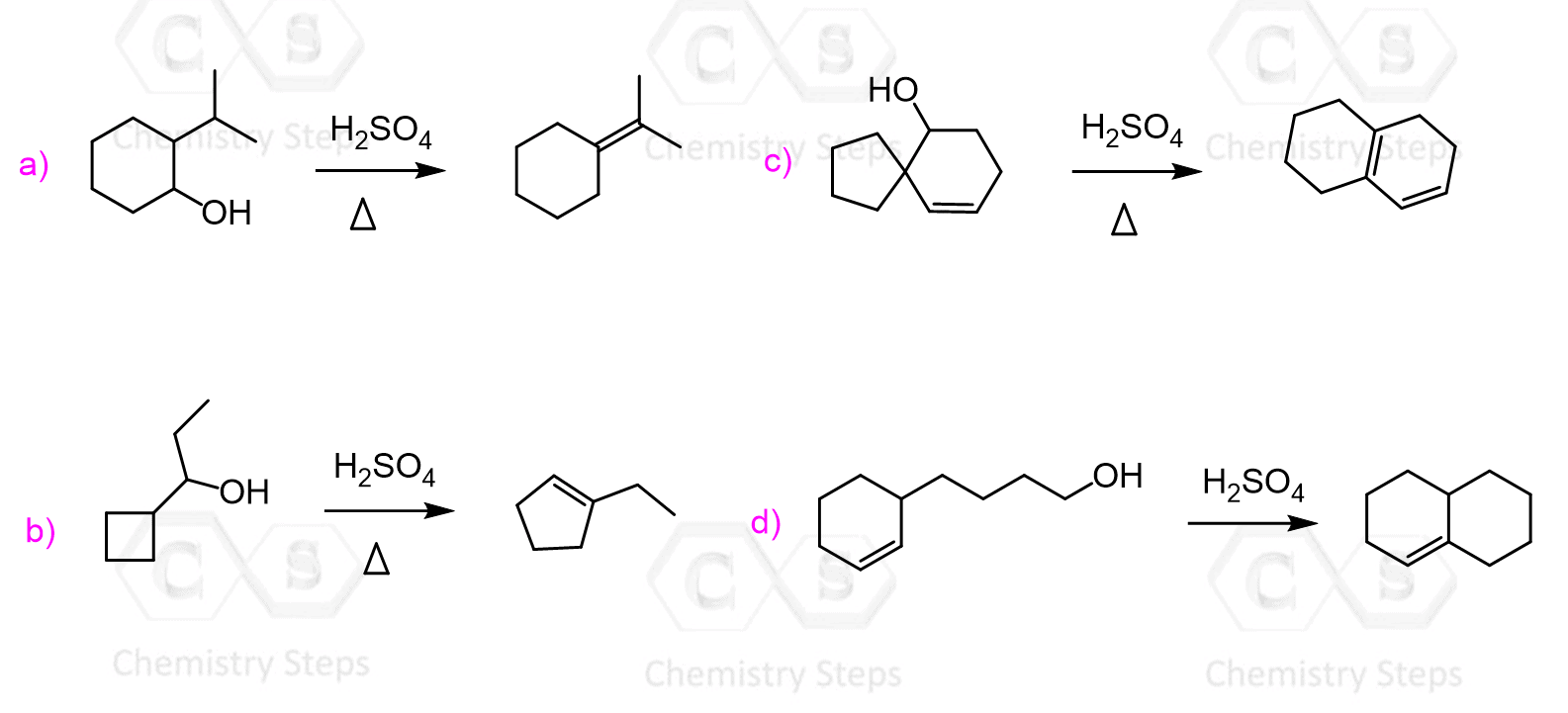
We have seen that carbocation rearrangements may lead to unexpected products in SN1 nucleophilic substitution reactions.
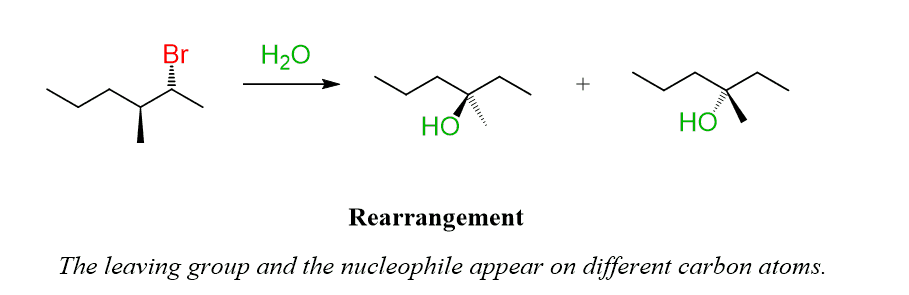
For example, when the following alkyl halide is reacted with water, the major product of the reaction is formed via a hydride shift, which transforms the initially formed secondary carbocation into a more stable tertiary carbocation.
Now, remember that the E1 mechanism is very similar to the SN1 mechanism in that both proceed through the formation of a carbocation intermediate. The only difference lies in what happens to that carbocation afterward:
- In SN1 reactions, a nucleophile attacks the carbocation, forming a substitution product.
- In E1 reactions, a base removes a β-hydrogen, leading to the formation of a double bond – the elimination product.

Because of this shared carbocation intermediate, E1 reactions can also undergo rearrangements just like SN1 reactions do. For example, when 2-chloro-3,4-dimethylpentane is reacted with water, an alkene and an alcohol are formed via a rearrangement of the intermediate secondary carbocation:

The SN1 and E1 reactions are always in competition, and the most common factor to favor the elimination path is heating up the reaction. You can read this article for more details on comparing the SN1 and E1 reactions and why heat favors elimination reactions.
Rearrangements in E1 Reactions of Alcohols
When an alcohol is protonated, the –OH group is converted into a better leaving group, water, and a carbocation is formed. Just like in the reactions of alkyl halides, a rearrangement can occur if the carbocation can be transformed into a more stable one.
For example, the following alcohol undergoes an acid-catalyzed dehydration; however, the major product of the reaction is not what we might expect at first glance:

The major product is a tetrasubstituted alkene, which is formed as a result of a hydride shift to transform the secondary carbocation into a more stable tertiary carbocation:

How to Avoid Rearrangements in E1 Reactions
The only way to avoid rearrangements in E1 reactions, of course, we’re talking about cases where rearrangements are possible is to change the reaction pathway to an E2 mechanism.
For example, the alcohol we mentioned earlier can first be converted to a mesylate or tosylate, which transforms the poor leaving group (–OH) into a much better one. This now allows the use of a strong base to promote an E2 elimination, forming the preferred regioisomer of the alkene without carbocation rearrangement.

Remember:
- Sterically unhindered bases such as ethoxide (EtO⁻) or methoxide (MeO⁻) generally give the Zaitsev product – the more substituted and more stable alkene.
- Bulky, sterically hindered bases such as tert-butoxide (t-BuO⁻) favor the Hofmann product, that is, the less substituted
The Use of POCl3 to Avoid Rearrangements
Another strategy for controlling the regiochemistry of an alcohol dehydration reaction is the use of phosphorus oxychloride (POCl₃) in the presence of a base, such as pyridine.
This method allows the elimination of water under milder, non-acidic conditions, which helps avoid carbocation rearrangements that commonly occur in acid-catalyzed E1 dehydrations.

In this reaction, POCl₃ first converts the alcohol into a good leaving group by forming a chlorophosphate intermediate. Then, the base (pyridine) abstracts a β-hydrogen in an E2-like elimination step, leading directly to the alkene without forming a carbocation.

Like with mesylates and tosylates, we are altering the transformation to an E2 mechanism, thereby avoiding the formation of carbocations and, consequently, any possible rearrangements.